Global Journal of Medical and Clinical Case Reports
Myxoid Leiomyosarcoma of the Uterus in a Fertile Woman
Vito Leanza1, Alfio D’Agati1, Gianluca Leanza1, Carlo Pafumi1*, Mayada Chammas2 and Antonio Carbonaro1
2American University of Beirut - Lebanon
Cite this as
Leanza V, D’Agati A, Leanza G, Pafumi C, Chammas M (2017) Myxoid Leiomyosarcoma of the Uterus in a Fertile Woman. Glob J Medical Clin Case Rep 4(1): 004-007. DOI: 10.17352/2455-5282.000033A 37 year old woman (G1P1 spontaneous delivery) was admitted to our university hospital for menometrorrhagia. The case history showed the patient had menometrorrhagia for six months; moreover, during the abdominal examination we found a mass occupying the hypogastric and mesogastric area. The consistence was hard and the mass reached the level of the umbilicus. The combined vaginal-abdominal examination confirmed the presence of the mass on the anterior wall and, in addition, multiple myomata were felt; the uterus was found enlarged in size (equivalent to 18 pregnancy weeks); both ovaries and Fallopian tubes were regular. During surgery multiple myomata were visualized. The largest, 10 centimeter diameter, was soft in consistence with a gelatinous structure. Total abdominal hysterectomy with preservation of adnexa was performed.
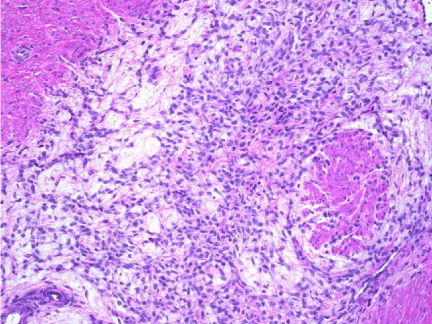
Histopathological result gave evidence of myxoid leiomyosarcoma for the largest myoma, whereas the other nodes were benign.
Introduction
The most common uterine mass during the fertile age are myomata [1,2]. The malignant degeneration of a myoma is a rare event.
Uterine sarcomas account for 3% of uterine cancers. Uterine sarcomas comprise leiomyosarcoma, endometrial stromal sarcoma, adenosarcoma, and their variants [3]. The rarity of these tumors has prevented the performance of large epidemiological studies to identify risk factors. Data regarding parity, onset of menarche, or age at menopause as risk factors are inconclusive [4].
Myxoid leiomyosarcoma (MLMSs) is an unusual tumor, in most cases recognized only after surgery. Rubin at al reported 18 cases of soft tissue leiomyosarcomata in which myxoid stroma occupied >50% of the tissue examined. In their study patients ranged from 22 to 84 years old (median, 57.5 yrs) and female patients outnumbered male patients for 14 to 4 [5].
When MLMSs is associated with multiple myomata, the gross appearance of the malignant mass differs from others for a soft, necrotic and gelatinous aspect. In the past, the presence of 10 or more mitoses per 10 high-power fields (HPFs) was considered right key to establish the diagnosis of leiomyosarcoma. Even though mitotic index is an important feature in the assessment of malignancy, several studies based on large series of uterine smooth muscle tumors (U-SMTs) have shown that mitotic activity alone is not predictive of poor outcome [6,7,8]. Current criteria for diagnosis of classic leiomyosarcomas require assessment of the number of Mitotic Figures (MFs) ⁄ 10 HPFs, nuclear atypia, and tumor cell necrosis (TCN), in recognition of the fact that a variety of exogenous and endogenous hormones can influence both the mitotic index and the development of TCN [9].
With ultrasound examination topographic limits are normal, whereas the internal structure is inhomogeneous and hypoechogic compared to either the fibroid node or uterine wall.
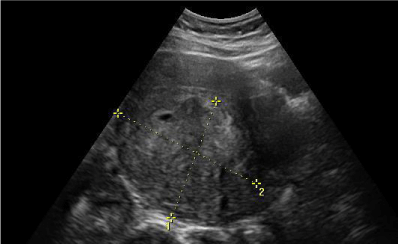
The doppler flow shows that intratumoral resistance index (RI) is significantly lower for uterine sarcomas comparison with leiomyomata, even though the result is not concordant in all cases. When a cut-off value of 0.5 for the RI is considered, the detection rate for uterine malignant mass is 67% [10].
In this study we report a case of uterine myxoid leiomyosarcoma associated with multiple myomata in a fertile woman.
Case Report
A 37 year old female, gravida 1, para 1 (spontaneous delivery), was admitted to our university hospital complaining of a medical history of abdominal and pelvic pain, associated with menometrorrhagia lasting for 6 months. During this period the patient had been undergoing intensive therapy with iron drugs and folates.
At the admission blood tests showed: haemoglobin 11.5 g/dl; iron 31 µg/dl; Red Blood Cells 4.320.000/µL; White Blood Cells 7.900/µL; Platelets 285.000/µL.
Upon abdominal examination a mass occupying the hypogastric and mesogastric was felt. The consistence was hard and it reached the level of the umbilicus. On vaginal examination, uterus was 18 weeks size and a mass was palpated on the anterior wall, multiple myomata were felt as well, both ovaries and Fallopian tubes were regular.
Speculum examination showed a regular cervix and vagina with clots coming from the uterine orifice. With ultrasound examination uterus appeared increased in size (longitudinal: 180 mm; transversal: 107 mm; sagittal diameter: 108 mm). Four fibroid masses were detected, three were subserous myomata belonging to the right (42 mm) and the left (34 mm) angle of the uterus, and the third (59 mm) to the left uterine wall. The fourth and greatest mass (100 mm), localized on the fundus, was irregular in structure and inhomogeneous.
The Doppler (Figure 1) flow study showed that the vascularization of the sick tissue was spread everywhere, mainly inside the tumor; on the contrary, the remaining three myomata showed a predominant peripheral vascularization. Resistance index was regular for all the masses (RI: 0.8). The ultrasound aspect of the largest mass was hypoechoic compared with the fibroid nodes.
The “Pfannestiel” Transversal laparotomy under general anesthesia was performed.
During surgery, as previously observed at ultrasonographic examination, uterus was found increased in size and multiple myomata were detected. The largest myoma had a grayish color and a gelatinous consistence; whereas the smaller ones were whitish and hard. Adnexa were regular in size and shape. Total hysterectomy with preservation of ovaries and Fallopian tubes was carried out. Post-operative course was uneventful. No transfusions were requested. After four days the patient was discharged.
The histopathological result (Figure 2) found a malignant neoplasm (myxoid leiomyosarcoma) infiltrating the surrounding myometrium; besides, areas of cotyleidoned leiomyoma were observed. The other fibroid nodes were typical for myomata. Endometrium was proliferative. A micropolypoid cervicitis was observed too.
Discussion
The limit between benign and malignant tumors is not always defined. Even benign nodes can be associated with malignant ones.
Histologically many types of leiomyomata have been recognized, although the aspect is not always indicative macroscopically. The great cellularity of myomata may lead to confusion with leiomyosarcomata.
Among various subtypes, the following are considered [11]:
1. Atypical leiomyomata are characterized by moderately to severely pleomorphic multinucleated tumor cells with low mitotic counts and absence of coagulative tumor cell necrosis. If multinucleated cells are numerous, the terms “bizarre” or “symplastic” leiomyoma have been applied. These tumors are also confused with leiomyosarcoma, which have a higher mitotic count [12].
2. Epitheliod leiomyoma include leiomyoblastoma, clear cell leiomyoma, and plexiform leiomyoma [13]. The clinical behaviour of the epitheliod leiomyomas is varied. Small tumors without cytologic atypia and with circumscribed margins, extensive hyalinization, low mitotic activity, and clear cells are considered benign.
3. Lipoleiomyoma contains large areas of fat. These areas may be well circumscribed or diffused. A pure lipoma is uncommon. These tumors should not be confused with mixed mesodermal sarcoma.
4. Leiomyoma with tubules is uncommon. Histologically, epithelium-lined tubules are noted. Mesothelial differentiation also occurs.
5. Myxoid leiomyoma contains an amorphous myxoid substance producing a soft translucent appearance. The margins are well circumscribed and mitotic figures are absent.
6. Cotyledoined leiomyoma or Sternberg Tumor is a very rare variant of uterine smooth muscle tumor characterized by the presence of congestal exophytic placental tissue. Cotyledonoid leiomyoma has a peculiar gross appearance which usually raises the suspicion of sarcoma. To avoid overtreatment of such cases, intraoperative frozen section is a mandatory and helpful procedure [14].
Uterine smooth muscle tumors range from the very common benign leiomyoma to the uncommon, but frequently lethal, leiomyosarcoma [15].
Usually, uterine smooth muscle neoplasms with fewer than 5 mitoses/10 high power fields are considered benign, whereas those with greater than 10 mitoses/10 high power field will often pursue a malignant course [16]. Other leiomyosarcomatous changes occur as the myoma outgrows its blood supply and are termed hyaline, myxomatous, calcific, cystic, fatty, carneous (red), and sarcomatous. The mildest change is hyaline degeneration, found in 65% of myomas. Its appearance is homogeneous, with loss of the whorled pattern and cellular detail as the smooth muscle cells are replaced by fibrous connective tissue. Myxomatous change occurs in 15% of tumors, and calcific degeneration occurs in 4% to 10%, more commonly in older women. Cystic degeneration is found in 4% of leiomyomata and results from coalescence of hyalinised areas with liquefaction.
Fatty degeneration occurs rarely, but may result from late stage hyaline degeneration.
Immunohistochemical examination of p53, p16 and Ki-67 is considered a very useful tool for identification of malignant focus [17].
Patients with a leiomyosarcoma with a leiomyomata component usually have favorable prognosis.
Conclusion
This case report is very interesting due to the rarity of this pathology occuring in women of fertile age. Uterine MLMSs are often difficult to recognize, owing to their mild cytologic atypia and a low mitotic index.
When many nodes are discovered and alterations of aspect found, the malignancy must be suspected and a surgical treatment must be performed. A correct diagnosis and treatment may save the woman and lead to a favorable prognosis and good quality of life.
These results suggest that the intratumoral RI detected by color and pulsed doppler ultrasonography by themselves could be poor for the preoperative differential diagnosis of uterine sarcoma.
This case report offers to consider mainly two aspects related to the diagnosis and the treatment. Regarding diagnosis, we have to consider three stages: the pre-operative, the intra-operative and the final result. Pre-operative clinical diagnosis is difficult to perform and vaginal bimanual examination cannot disclose the malignancy of the mass.
Ultrasounds are useful for the diagnosis of the mass, but in many cases the difference between leiomyoma with benign degeneration and leiomyosarcoma is very tricky.
Doppler flowmetry is helpful when there is a suspicion of malignancy; moreover, in many cases it is not discriminating.
Intra-operatively, the macroscopic appearance of gelatinous aspect and its infiltrating characteristic tend towards malignancy.
The surgical treatment of gynecological masses complains both laparoscopic [18-21] and laparotomic approaches. Usually laparoscopy is preferred when masses are supposed benign or in some cases to define the diagnosis. In case of malignancy laparotomy is preferred.
Histologic result is considered the final diagnosis; nevertheless, in such a case the doubts remain and it confirms the old phrase: “natura non facit saltus” (nature doesn’t make steps), meaning that changes are gradual and limits are sometimes difficult to find.
Valentina Pafumi has carried out English language editing for this article.
- Carlo P, Vito L, Antonio C, Gianluca L, Grazia SM, et al. (2011) Efficacy evaluation of a test CINtec® p16INK4a in screening for cervical HPV infection. Open Journal of Preventive Medicine1: 154-163. Link: https://goo.gl/zstbRH
- Pafumi C, Iraci SM, Abbate G, Clemente CM, La Rosa I, et al. (2010) Protection of ovarian tissue from radiotherapy. Bratisl Lek Listy 111: 443-448. Link: https://goo.gl/UW0fhv
- Zarbo G, Giunta MR, Giannone TT, Pafumi C, Leanza V (2014) Correlazione tra HPV e adenocarcinoma dell’endocervice. Gazzetta Medica Italiana. Archivio Per Le Scienze Mediche 173: 259-264. Link: https://goo.gl/LnYIwk
- Leanza V, Fichera S, Leanza G, Cannizzaro MA (2011) Huge fibroid (g. 3.000) removed during cesarean section with uterus preservation. A case report. Ann Ital Chir 82: 75-76. Link: https://goo.gl/9LiIhM
- Leanza V, D’Agati A, Accardi M, Russo ER (2009) Multiple myomectomy performed during cesarean section: a case report. Minerva Ginecologica 61: 245. Link: https://goo.gl/Cpnawy
- Tse KY, Crawford R, Ngan HY (2011) Staging of uterine sarcomas. Best Pract Res Clin Obstet Gynecol 25(6): 733-749. Link: https://goo.gl/AMJQfn
- Christopherson WM, Williamson EO, Gray LA (1972) Leiomyosarcoma of the uterus. Cancer 29: 1512-1517. Link: https://goo.gl/aRrvFE
- Rubin BP, Fletcher CD (2000) Myxoid leiomyosarcoma of soft tissue, an underrecognized variant. Am J Surg Pathol 24: 927-936. Link: https://goo.gl/6fTMZj
- Chang E, Shim SI (1998) Myxoid leiomyosarcoma of the uterus: a case report and review of the literature. J Korean Med Sci 13: 559-562. Link: https://goo.gl/4aQIxj
- Hendrickson MR, Kempson RL (2000) A diagnostic approach to smooth muscle tumours of the uterus. Curr Diagn Pathol 6: 21–30 Link: https://goo.gl/1APRvc
- Oliva E, Clement PB, Young RH (2002) Mesenchymal tumours of the uterus: selected topics emphasizing diagnostic pitfalls. Curr Diagn Pathol 8: 268–282 Link: https://goo.gl/6qapGz
- Burch DM, Tavassoli AF (2011) Myxoid leiomyosarcoma of the uterus. Hystopathology 59: 1144-1155 Link: https://goo.gl/osoQS1
- Szabó I, Szánthó A, Csabay L, Csapó Z, Szirmai K, et al. (2002) Color Doppler ultrasonography in the differentiation of uterine sarcomas from uterine leiomyomas. Eur J Gynaecol Oncol 23: 29-34. Link: https://goo.gl/vyLMb5
- Toledo G, Oliva E (2008) Smooth muscle tumors of the uterus: a practical approach. Arch Pathol Lab Med 132: 595-605 Link: https://goo.gl/CcbG6H
- Sung CO, Ahn G, Song SY, Choi YL, Bae DS (2009) Atypical Leiomyomas of the uterus with long-term follow-up after myomectomy with immunohistochemical analysis for p16INK4A, p53, Ki-67, estrogen receptors, and progesterone receptors. Int J Gynecol Pathol 28: 529-534. Link: https://goo.gl/lV7Bap
- Kurman RJ, Norris HJ (1976) Mesenchymal tumors of the uterus. VI. Epithelioid smooth muscle tumors including leiomyoblastoma and clear-cell leiomyoma: a clinical and pathologic analysis of 26 cases. Cancer 37: 1853-1865 Link: https://goo.gl/mp273v
- Seed AS, Hanaa B, Faisal AS, Naila AM (2006) Cotyledonoid dissecting leiomyoma of the uterus: a case report of a benign uterine tumor with sarcoma like gross appearance and review of literature. Int J Gynecol Pathol 25: 262-267 Link: https://goo.gl/vmY6Le
- Quade BJ, Wang TY, Sornberger K, Dal Cin P, Mutter GL, et al. (2004) Molecular pathogenesis of uterine smooth muscle tumors form transcriptional profiling. Genes Chromosomes Cancer 40: 97-108 Link: https://goo.gl/67Ao7l
- Fletcher JA, Morton CC, Pavelka K, Lage JM (1990) Chromosome aberrations in uterine smooth muscle tumors: potential diagnostic relevance of cytogenetic instability. Cancer Res 50: 4092-4097 Link: https://goo.gl/gGm61n
- Yanai H, Wani Y, Notohara K, Takada S, Yoshino T (2010) Uterine leiomyosarcoma arising in leiomyoma: Clinicopathological study of four cases and literature review. Pathol Int 60: 506-509. Link: https://goo.gl/z1sAoW
- Vecchio R, Leanza V, Genovese F, Accardi M, Gelardi V, et al. (2009) Conservative laparoscopic treatment of a benign giant ovarian cyst in a young woman. J Laparoendosc Adv Surg Tech A 19: 647-648. Link: https://goo.gl/b2oxd8

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Save to Mendeley
Save to Mendeley
