Archives of Clinical Gastroenterology
Duodenal Eosinophilia and Gastroparesis: Is there a role?
Sharareh Moraveji1*, Mohammad Bashashati1, Ben Alvarado1, Irene Sarosiek1, Alireza Torabi2 and Richard McCallum1
2Department of Internal Medicine and Pathology, Presented in Poster Form at Digestive Disease Week (DDW) 2017, May 6-9 in Chicago, IL at McCormick Place, USA
Duodenal eosinophils are not increased in idiopathic gastroparesis patients compared to gastroparesis of diabetic etiology and control patients with normal gastric emptying.
This separates the entities of functional dyspepsia and gastroparesis as far as proposing a spectrum linking functional dyspepsia to idiopathic gastroparesis and hence treatment options would be different for the two entities.
Cite this as
Moraveji S, Bashashati M, Alvarado B, Sarosiek I, Torabi A, et al. (2018) Duodenal Eosinophilia and Gastroparesis: Is there a role? Arch Clin Gastroenterol 4(1): 001-004. DOI: 10.17352/2455-2283.000046Gastroparesis (GP) is a disorder that affects the motility of the stomach resulting in delayed gastric emptying (GE) without mechanical obstruction and has accompanying symptoms that include nausea, vomiting, early satiety, bloating, and epigastric pain. It was reported that increased eosinophils were identified in the first part of the duodenum in a subset of patients with the entity of functional dyspepsia (FD) and the main symptom of early satiety. It is recognized that approximately 40% of FD patients may also have delayed gastric emptying, termed Idiopathic GP (ID-GP). Therefore, to investigate the hypothesis that duodenal eosinophilia may be present in patients diagnosed with ID-GP we investigated whether there is any evidence of increased numbers of duodenal eosinophils in patients with the diagnosis of ID-GP as compared to diabetic GP (DM-GP) patients and controls who had a normal GE.
Methods: Endoscopic biopsies were obtained from the first part of the duodenum in 15 ID-GP and 15 DM-GP patients, all symptomatic and with documented GP based on scintigraphic evidence of >10% retention of a radiolabeled meal at 4 hours. In addition, we studied 18 patients with a normal gastric emptying undergoing endoscopy for miscellaneous indications. None of the GP and controls had any organic GI abnormality. The biopsies were stained with H&E and the eosinophils in intact duodenal villi were counted and averaged in 5 high power fields (HPF) by 2 independent experts who were blind to the clinical diagnosis.
Results: None of the ID-GP, DM-GP and control patients had any duodenal inflammation or pathological features of celiac disease. No gastric pathology was detected and the gastric biopsies were negative for H. Pylori. No significant differences were observed in duodenal eosinophil counts among the 3 groups- specifically the mean number of eosinophils in the first part of duodenum of control patients was 8.8/HPF (SD 3.9), while this was 11.7/HPF (SD 9.9) in DM-GP and 10.8/HPF (SD 7.3) in ID-GP.
Conclusion: Duodenal eosinophils are not increased in ID-GP patients, which would indicate that duodenal eosinophilia does not seem to play a role in the pathophysiology of this disorder nor indicate that there may be a spectrum linking some functional dyspepsia patients to gastroparesis.
Background
Gastroparesis (GP) is characterized by the presence of chronic and often debilitating upper gastrointestinal (GI) symptoms in the setting of delayed emptying of the stomach in the absence of mechanical obstruction [1]. It is a relatively common GI disorder affecting more than 10 million individuals in the United States [2]. The major etiologies of GP are longstanding Type I or II Diabetes Mellitus (DM), idiopathic GP and post-vagotomy. The pathophysiology of idiopathic gastroparesis (ID-GP) is not well understood. ID-GP has clinical overlaps with functional dyspepsia (FD) and since gastric emptying (GE) is not routinely measured in FD patients, some of these patients may actually have gastroparesis which would be classified as idiopathic gastroparesis (ID-GP) [3-5]. Earlier studies have reported increased eosinophils in the duodenum of some FD patients who presented with the dominant symptoms of both early satiety and post-prandial fullness [6,7]. To pursue the hypothesis that duodenal eosinophilia could also be present in GP patients, specifically those with gastroparesis of idiopathic (ID) etiology, we examined whether the number of duodenal eosinophils was increased in patients with typical GP symptoms and ID-GP compared to patients with Diabetic (DM) GP and patients undergoing upper gastrointestinal endoscopy for non-gastroparetic indications and who had a normal gastric emptying.
Methods
Biopsies were obtained during upper gastrointestinal (GI) endoscopy from the first part of the duodenum in symptomatic GP patients: 15 of ID etiology, 15 with DM-GP, all based on abnormal GE scintigraphy, as well as in 18 patients without GP (control group) who underwent upper endoscopy for symptoms not related to gastroparesis, e.g., iron deficiency anemia, persistent gastroesophageal reflux symptoms, occult gastrointestinal bleeding, surveillance for malignancy and dysphagia/odynophagia.
Symptomatic DM-GP and ID-GP patients were diagnosed based on gastric emptying (GE) scintigraphy. GE scintigraphy was performed in the morning between 8 and 9 AM in patients who had fasted overnight. The meal was prepared just before the test and consumed within 20 minutes. The meal consisted of a scramble egg substitute (120g Eggbeater, 60kcal), 2 slices of bread (120kcal), strawberry jam (30 grams, 75 kcal), and water (120mL). First, 1 mCi of 99Tc- labeled sulfur colloid was mixed with the egg substitute; then this mixture was cooked in a microwave oven for 2 min, and stirred during cooking. For the scintigraphy, anterior & posterior images were taken within 1 min of the completion of the meal. Subsequent images were obtained at 60, 120, and 240 min in all patients [8].
Delayed GE was diagnosed if there was > 10% retention of the isotope labeled meal after 4 hours.
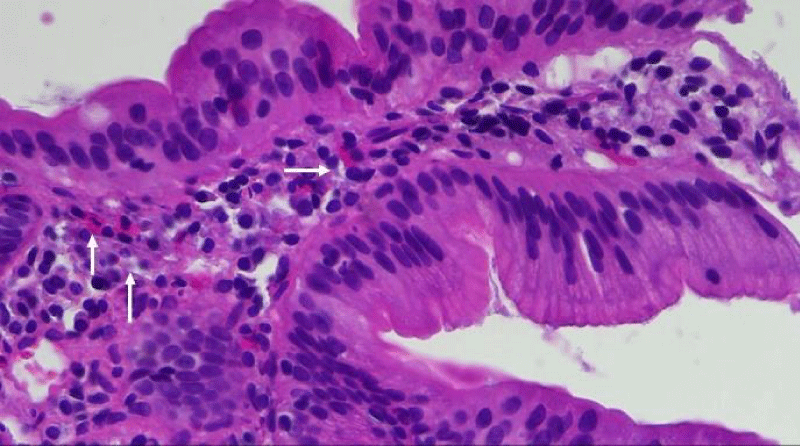
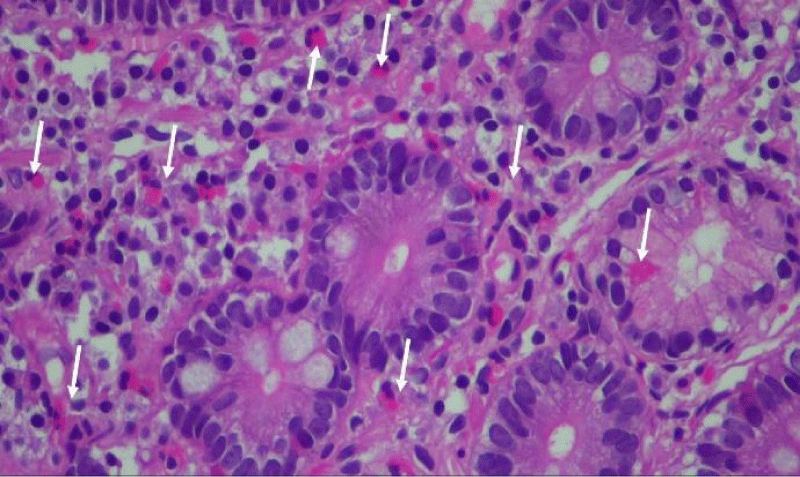
The 18 control patients had normal gastric emptying (<10% retention at 4 hours). None of the GP or control patients had any organic GI abnormality. The endoscopic biopsies were stained with H & E and the eosinophils identified in intact duodenal villi (Figures 1a,b), were counted by 2 independent experts who were blinded to the clinical diagnosis. The number of duodenal eosinophils/high power field (HPF) was calculated by averaging 5 high power fields in both GP and control groups. The data are presented as mean plus or minus standard deviation in addition to the absolute eosinophil numbers for ID-GP, DM-GP groups and the control patients.
Results
In ID-GP, DM-GP and control subjects, 13/15, 13/15 and 9/18 were female, respectively. Mean age of the ID-GP, DM-GP and control patients was 51.5 (SD 17.1), 43.2 (SD 12.2), and 48.2 (SD 16.3) years old, respectively. None of the ID-GP, DM-GP and control patients had any duodenal inflammation or pathological features of celiac disease by duodenal biopsy. Routine gastric biopsies were obtained from the antrum and body and were negative for H. Pylori. There was no statistically significant difference in the mean number of eosinophils identified from biopsies in the first part of duodenum of control patients: 8.8/HPF (SD 3.9), as compared to DM-GP 11.7/HPF (SD 9.9) and ID-GP patients 10.8/HPF (SD 7.3) (Figure 2). Duodenal eosinophil counts in normal control subjects ranged from 3 to 16 eosinophils/HPF. In 15% of both ID-GP and DM-GP patients the upper limit of this range was exceeded to similar degrees. In ID patients, the highest number counted was 40 eosinophils per high power field (HPF) while in diabetic gastroparesis the most eosinophils counted was 35 eosinophils/ HPF.
Discussion
Based upon recent research in functional dyspepsia (FD) patients up to 73% have been identified with increased numbers of duodenal eosinophils on endoscopic biopsies [7]. This group of FD patients had predominant symptoms of early satiety and post-prandial fullness. In that report, there was an increased mean number of duodenal eosinophils in FD patients (p<0.01) as compared to controls: 33/HPF as compared to 18.4/HPF in controls. These “control” numbers of eosinophils were derived from a previous study in 1001 subjects undergoing upper gastrointestinal endoscopy and biopsy to assess for pathology to explain their symptoms. A duodenal eosinophil count of <22 eosinophils/HPF was regarded as the normal value although counts ranged from 8-55/HPF [7]. These observations about duodenal eosinophils in FD were confirmed by Walker et al. in 2010, demonstrating that 47% of FD patients with symptoms of postprandial distress, most notably with symptoms of early satiety, also had increased duodenal eosinophils. A further report by these authors in 2014, also confirmed this association between increased eosinophils in the duodenum in FD patients with early satiety where their mean eosinophil count was 49 compared to 8 in their controls [6].
Dyspepsia in the setting of chronic/recurrent discomfort or pain in the upper abdomen and a negative upper endoscopy leads to a diagnosis of functional dyspepsia (FD). According to Rome IV criteria, functional dyspepsia (FD) is subdivided into 1) epigastric pain syndrome (EPS) and 2) postprandial distress syndrome (PDS). PDS encompasses symptoms of postprandial fullness and early satiety. On the other hand, epigastric pain syndrome (EPS) has predominant symptoms of epigastric pain or burning [9]. The subset of FD patients reported by Tally and Walker would meet criteria for PDS.
GP symptomatically overlaps functional dyspepsia (FD) and since gastric emptying (GE) is not routinely measured in FD patients, some of these patients may have GP. It has been reported that approximately 40% of patients with functional dyspepsia have documentation of delayed GE and these patients with slow gastric emptying would be termed idiopathic in origin [10,11].
Other studies have shown that 86% of patients with ID-GP also meet the criteria for the diagnosis of functional dyspepsia [12]. When it comes to symptoms, upper abdominal pain has been identified as more prevalent in functional dyspepsia, while nausea is more prevalent in GP patients.
Gastroparesis (GP) is characterized by chronic symptoms related to delayed gastric emptying (GE) in the absence of any mechanical obstruction. Symptoms of GP include: nausea, vomiting, early satiety, postprandial fullness, bloating and abdominal pain and/or discomfort. It is mainly observed in females (75%). McCallum et al demonstrated in a study that the etiology of gastroparesis was diabetes in 29%, idiopathic in 36% and postsurgical in 13% [13]. A multicenter database investigation of patients with gastroparesis by Parkman et al., revealed that 64% had idiopathic gastroparesis and 31% had diabetic gastroparesis; therefore, making idiopathic gastroparesis (ID-GP) the most common form [14].
Our study examined the number of duodenal eosinophils in patients with GP symptoms and documented delayed GE. The GP patients were of both ID and DM etiologies and their results were compared to a “control” group of patients with normal gastric emptying who underwent upper endoscopy for miscellaneous indications. In our control group, the duodenal eosinophil counts ranged from 3-16 eosinophils/HPF. Based on analyzing mean eosinophil data, we found similar numbers of duodenal eosinophils in ID or DM GP patients. We did observe that 15% of each subtype (ID and DM) did exceed the upper limits of our normal subjects - >22/HPF.
However, the range of up to 40 eosinophils/HPF in ID and 35 in DM respectively was similar. These numbers are well below the upper limit of the range for the control subjects (55/HPF) in the previous reports by Tally et al.
One of the weaknesses of our study could be judged as the need for a larger population of both patients and “controls” to confirm our conclusions. Our strengths are the documentation of gastroparesis was carefully confirmed at an academic gastroparesis referral center, in addition to the careful pathological examination for the counting of duodenal eosinophils by two experienced pathologists blinded to the patient diagnoses as well as the inclusion of a control group of patients with normal gastric emptying.
We conclude that duodenal eosinophils do not appear to be increased in ID-GP patients when compared to DM-GP and controls, indicating that duodenal eosinophilia does not play a role in the pathophysiology of ID-GP nor is there evidence for a spectrum or link connecting FD with ID-GP.
Acknowledgments
Funding and Disclosures
MB, RMC, SM performed the research and data gathering. MB, RMC designed the research study. AT contributed essential reagents or tools. AT, MB analyzed the data. MB, RMC, SM wrote the paper. (MB = Mohammad Bashashati; RMC= Richard McCallum; SM= Sharareh Moraveji).
- Feldman M (2016) Gastric neuromuscular function and neuromuscular disorders. Sleisenger and Fordtran's Gastrointestinal and Liver Disease: Pathophysiology, Diagnosis, Management. 10th ed. Philadelphia, Pa.: Saunders Elsevier; 2016. Link: https://goo.gl/vLsCnG
- Bharucha AE (2015) Epidemiology and natural history of gastroparesis. Gastroenterology Clinics of North America 44: 9. Link: https://goo.gl/XVtRfN
- Stanghellini V, Tosetti C, Paternico A (1996) Risk indicators of delayed gastric emptying of solids in patients with functional dyspepsia. Gastroenterology 110: 1036–1042. Link: https://goo.gl/L9ufmX
- Sarnelli G, Caenepeel P, Geypens B (2003) Symptoms associated with impaired gastric emptying of solids and liquids in functional dyspepsia. Am J Gastroenterology 98: 783–788. Link: https://goo.gl/sVuuJC
- Maes BD, Ghoos YF, Hiele MI (1997) Gastric emptying rate of solids in patients with non-ulcer dyspepsia. Dig Dis Sci 42: 1158–1162. Link: https://goo.gl/Gk6AGD
- Walker MM, Aggarwal KR, Shim LS (2014) Duodenal eosinophilia and early satiety in functional dyspepsia: confirmation of a positive association in an Australian cohort. J Gastroenterology Hepatology 29: 474–479. Link: https://goo.gl/ynqD7D
- Talley NJ, Walker MM, Aro P (2007) Non-ulcer dyspepsia and duodenal eosinophilia: an adult endoscopic population-based case-control study. Clinical Gastroenterology Hepatology 5: 1175–1183. Link: https://goo.gl/NLfzSe
- Tougas G (2000) Assessment of Gastric Emptying Using a Low Fat Meal: Establishment of International Control Values. The American Journal of Gastroenterology 95: 1456-1462. Link: https://goo.gl/yypyrs
- Tack J, Talley NJ, Camilleri M (2006) Functional gastroduodenal disorders. Gastroenterology 130: 1466–1479. Link: https://goo.gl/1q3jdy
- Quartero AO, de Wit NJ, Lodder AC (1998) Disturbed solid-phase gastric emptying in functional dyspepsia: a meta-analysis. Dig Dis Sci 43: 2028–2033. Link: https://goo.gl/ZUA2X1
- Parkman HP (2010) Gastroparesis and functional dyspepsia: excerpts from the AGA/ANMS meeting. Neurogastroenterology & Motility 22: 113–133. Link: https://goo.gl/2yB3us
- Yarandi SS, Christie J (2013) Functional Dyspepsia in Review: Pathophysiology and Challenges in the Diagnosis and Management due to Coexisting Gastroesophageal Reflux Disease and Irritable Bowel Syndrome. Gastroenterology Research and Practice 2013. Link: https://goo.gl/UK3SfP
- McCallum RW, Ricci DA, Rakatansky H (1983) A multicenter placebo-controlled clinical trial of oral metoclopramide in diabetic gastroparesis. Diabetes Care 6: 463–467. Link: https://goo.gl/fvbU34
- Parkman HP (2011) Clinical features of idiopathic gastroparesis vary with sex, body mass, symptom onset, delay in gastric emptying, and gastroparesis severity. Gastroenterology 140: 101-115. Link: https://goo.gl/4GFbez

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
