Open Journal of Plant Science
Comparative ultrastructure of caryopsis and leaf surface anatomy in wild rice Oryza coarctata and O. rufipogon through Scanning Electron Microscope (SEM)
Subhas Chandra Roy1* and Anurag Chowdhury2
2Sundarban Biosphere Reserve, 24 Pargana South Division, Pin-700027, WB, India
Cite this as
Roy SC, Chowdhury A (2021) Comparative ultrastructure of caryopsis and leaf surface anatomy in wild rice Oryza coarctata and O. rufipogon through Scanning Electron Microscope (SEM). Open J Plant Sci 6(1): 030-041. DOI: 10.17352/ojps.000030The wild rice Oryza coarctata (Roxb.) is an herbaceous halophytic plant belongs to the grass family poaceae prevalent to the coastal regions of Southern Asia. The O. coarctata is the only hydro-halophytic rice germplasm under the genus Oryza and shows high salinity. Caryopsis ultrastructure of O. coarctata was compared with another wild rice O. rufipogon through Scanning Electron Microscopy (SEM) including leaf surface anatomy to unveil the differentiation between the two species of Oryza. In O. rufipogon, embryo is small and orthodox type with long viability, in O. coarcata, embryo is large size and recalcitrant type. In O. coarctata, lower part of the spikelet has callus and expanded disc-like structure, without any globose rachilla, lemma devoid of tubercles, prickles and microhairs. Leaves of O. coarctata contain salt hairs and salt glands to secrete excessive salt, during high salt concentration which is a most important characteristic of this halophytic wild rice. Caryopsis endosperm contains starch granule of spherical shape with protein bodies in O. coarctata, whereas polygonal or hexagonal with moderate angularity starch granule in O. rufipogon. Aleurone layer is not so distinct in O. coarctata in compared to O. rufipogon, where it is clear and distinct. Protein profiling was studied through SDSPAGE for banding pattern variation analysis. This study of rice caryopsis ultrastructure and leaf surface anatomy including salt-hairs will contribute to the knowledge about the conservation of such precious germplasm of Sudarban mangrove region for the improvement of climate resilient rice varieties in future through pre-breeding and transgenic system.
Abbreviations
AG: Aleurone Grain; AL: Aleurone Layer; ALC: Aleurone Layer Cell; CSG: Compound Starch Granule; DUS: Distinctive Uniformity Stability Test; EDAX: Energy Dispersive X-Ray Analysis; IRRI: International Rice Research Institute; kDa: Kilo Dalton; kV: Kilo Volt; Mb: Million Base Pair; mM: Millimolar; Nacl: Sodium Chloride; PB: Protein Body; PPV&FR: Protection Of Plant Varieties & Farmers Right Acts; QTL: Quantitative Trait Loci; Saltol: Salt Tolerance; SDS-PAGE: Sodium Dodecyl Sulfate –Polyacrilamide Gel Electrophoresis; SEM: Scanning Electron Microscopy; SG: Starch Granule; SSG: Single Starch Granule
Introduction
The tribe oryzeae consisting of 12 genera, among these only 2 are cultivated species and remaining 22 are wild species. Total 24 species are available under this tribe Oryzeae and spread worldwide [1,2]. Among the two cultivated species, one species Oryza sativa L. grows throughout the world and O. glaberrima Steud. only grows in West Africa [3,4]3. Species of this genus Oryza are distributed through the world (Asia, Africa, Australia, and the Americas) ranges from driest desert to sea level to an altitude of 3000 m above sea level. These species are growing in such diverse agro-climatic conditions and show wide range of genetic diversity because they have not undergone any human selection or any kind of domestications [5]. Naturally they grow and adapted in this fluctuating environmental conditions and many genes have been accumulated slowly through the natural selection. Different species of rice have different genome sets such as diploid genome sets are AA, BB, CC, EE, FF, GG genome, tetraploid genome sets are BBCC, CCDD, KKLL, HHJJ, and HHKK indicating 11 genome categories in the genus Oryza [2,6]. Therefore, wild rice species are the source of many important genes/ QTLs responsible for biotic and abiotic stress tolerances. Many traits such as resistance to diseases and tolerance to submergence, drought, salinity, and cold have been successfully transferred from wild species into cultivated rice varieties for yield increase and better performance in the different ecoagroclimatic conditions. It is uncertain whether the current agricultural production system will be able to feed the expected nine billion people in this world by 2050 [7-10], whereas we need to produce double amount of rice. Because, productivity of rice affected several biotic and abiotic stresses (drought, cold, flood, salinity, heat) due to climate changes [11,12]. Plant has evolved some kind of mechanism to protect from such type of abiotic stresses by a series of adaptation leading to morphological, physiological and biochemical changes [13]. Wild relatives of the cultivated species are the natural source of ‘tolerance genes’ against these stresses (Abiotic) [14]. Salinity stress may cause 20% yield decrease in rice production [15] and may cause many physiological changes ( Na+/K+ ratio balance) in plant growth and development [16-19]. Abiotic stresses such as flooding, drought, salty soil and other climate change has created more burdens to food security [20].
The Oryza sativa gene, OsHKT1;5 is the causative genetic component of The Saltol is a major major Quantitative Trait Locus (QTL) containing gene OsHKT1;5 and is necessary genetic component for salt tolerance in rice [21]. It has identified as a plasma membrane transporter which controls the partitioning of Na+ between roots and shoots through efflux of Na+ from the xylem to neighbouring cells of parenchyma [22]. Local rice cultivar indica type, Nona Bokra has high salt tolerant potentiality due to increased Na+ efflux activity in presence of OsHKT1;5 transporter gene with four amino acid changes in compared to other salt sensitive rice cultivar [22,23]. Improved rice varieties can be developed by exploring and proper utilizing the rice germplasm for desirable abiotic stress tolerant traits [24]. Salinity of the soil is increasing globally day by day due to surface irrigation, high cropping intensity with high yielding varieties resulted in reduced yield potentiality [25]. Genetic potential of stress tolerance (salt) trait is existing among the germplasm of primary gene pool are utilized for the development of salt tolerance crops. Wild species of rice is a good source of many biotic and abiotic tolerance genes /QTLs which are not selected during rice domestication [26]. It has been reported earlier that Oryza coarctata, a wild rice species of mangrove ecology has high adaptability to saline stress [27], whereas other wild species of rice such as O. ridleyi and O. schlechteri are known for the trait of submergence tolerance [6,17,28]. The O. coarctata is considered as hydrohalophytic because they can tolerate high soil salinity and naturally growing in mangrove region of the world carrying allotetraploid genome set KKLL (2n=4X=48) with self-fertility behaviour and also has submergence tolerance trait [4,29-31]. Genome size is approximately 665 Mb (Mandal, et al. 2019). Prevalence of this wild rice O. coarctata mainly found in coastal region of India, Pakistan, Myanmar, Malyasia, and Bangladesh (regions of South Asian countries) with high submergence and salt tolerance characteristics [17,32]. This wild rice can be a good source of germplasm to develop high salinity tolerance improved varieties [32]. Hybridization was made between O. sativa and O. coarctata to develop hybrid F1 to establish their cross compatibility with cultivated rice [33] and others (www.irri.org). Salt tolerant rice cultivars with introgressed O. coarctata traits for salt tolerance are currently under development in the International Rice Research Institute (IRRI; www.irri.org). Normal growth was unaffected at high salt concentration (400 mM of NaCl) but growth of cultivated rice (O. sativa) was hampered [34]. The wild rice O. coarctata has unique leaf morpho-anatomy and physio-molecular mechanism by which they exclude salt (Na+) from the leaf surface using salt hairs and salt glands [17,34-36]. It is signify that O. coarctata keep away from Na+ toxicity in mesophyll cells by compartmentalization of salt in the vacuole and in epidermal hairs maintaining a low Na:K ratio, a mechanism generally found in halophyte grasses [17,34]. This species was placed in the separate genus Porteresia by Tateoka (1965) (Porteresia coarctata) based on morpho-anatomical distinction. Recently it has been returned to the rice genus Oryza (Lu and Ge 2004) (Oryza coarctata) based on morphological similarities and hybridization behaviour with O. sativa. The O. coarctata is endemic to coastal areas in Bangladesh, India, Pakistan, Myanmar, Malaysia and other countries of South Asia is a potential source of salt tolerance for cultivated rice O. sativa. Allotetraploid condition of O. coarctata (KKLL genome) may provide a clue to its unusual morphology and distribution in the salinity coastal regions of the South Asian countries, and may facilitate morpho-anatomical and physiological changes to adapt in such novel high salinity environment. Asian common wild rice species, Oryza rufipogon Griff (AA genome) shows the follows agronomically important traits such as salinity, cold, drought and flood water logging [37,38]. Wild rice accessions O. rufipogon can show salt tolerance and submergence activity [15,39]and may utilize as imperative germplasm for crop improvement program especially abiotic stress tolerance. Grains of wild rice composed of similar kind of starches and other nutritional properties as O. sativa and people are consumed as a delicacy food for nutritional values. Caryopsis of different wild rice was investigated through Scanning Electron Microscopy (SEM) to reveal the ultrastructure of the grain under in situ condition [5,40]. Kasem, et al. [5] studied the comparative grain morphology and histo-anatony among wild rice endosperm without considering wild rice O. coarctata. There is no report about the caryopsis ultrastructure of O. coarctata and comparative analysis with that of caryopsis of other wild rice O. rufipogon. Therefore, a comprehensive comparative caryopsis ultrastructure study was carried out in O. coarctata (first time report) and O. rufipogon using scanning electron microscope to reveal detailed histo-anatomy of grain caryopsis and plant morphology including leaf anatomy. The objective of this study is to reveal the comparative ultrastructure of caryopsis and leaf surface anatomy to help the rice breeder in future to develop salt tolerant climate ready rice varieties through pre-breeding program to provide food for more than >9 Billion World’s population by 2050.
Methods
Plant collection and maintenance
Wild rice Oryza coarctata (Roxb.) was collected from the coastal part of Sundarban Biosphere reserve (Figure 1) of West Bengal and maintained in the plastic bucket to study the plant morphology, paddy grain structure and caryopsis. Similarly, other wild rice Oryza rufipogon was collected from the ditches/marshy land of Raiganj, Uttar Dinajpur, West Bengal, India and maintained in the Department of Botany, University of North Bengal, India to study their morphology and seed caryopsis in detailed. Morphological data recording was carried out based on DUS test protocol (PPV&FR Act 2001, Govt. of India).
Scanning Electron Microscopy (SEM) for the Caryopsis ultrastructure analysis
Mature caryopsis was taken out by removing the husk using fine forceps. Caryopsis cut in to round pieces of 1mm thick length wise by applying slight pressure on the middle of the caryopsis with a razor blade. Fractured surface (solid round ring) facing upwards to get whole inner surface (in situ condition) of the grain was mounted on a specimen stub and coated with thin film of gold by means of a sputter coater (Jeol Model Smart Coater PF 18001006-2) about 2 minutes at high vacuum evaporator condition. Detailed inner structure was viewed through scanning electron microscope (SEM) (Jeol Model JSM-IT100, Japan) at various magnification with an accelerating voltage of 10 kV [41,42] to study the histo-anatomical ultrastructure of the rice caryopsis of O. coarctata and O. rufipogon. Mineral elements are to be qualitatively detected using EDAX system of SEM mainly zinc, and iron. These elemental deficiencies may cause hidden hunger.
Protein profiling through SDS-PAGE Electrophoresis
Protein profiling was carried out using SDS-PAGE electrophoresis based on standard protocol [43]. Leaf sample (1 g) was taken for protein extraction and purification. Phosphate buffer pH 7.5 was used for protein isolation from the tender leaf of wild rice O. coarctata and O. rufipogon. SDS-PAGE analysis was performed using isolated protein from wild rice. The analysis was performed using a SDS-Tris-glycine buffer system with 5% (w/w) stacking gels and 12% (w/w) resolving gels using the standard method [43]. Electrophoresis was initially performed at a constant voltage of 80 V followed by an increase to 100 V. At the end of electrophoresis, the gel was separated and stained with Coomassie brilliant blue G-250 for 10 h and destained with a destainer solution composed of 10% ethanol, 20% acetic acid, and 70% sterile distilled water. The gel images were photographed and subjected to analysis.
Results
Plant morphology and leaf surface anatomy
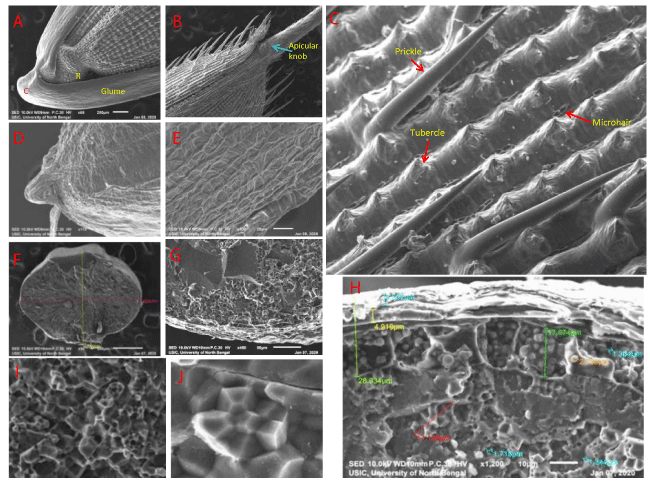
The wild rice Oryza coarctata (Roxb.) is naturally growing as associated vegetation in the mangrove forest of Sudarban Biosphere Reserve area of the district South 24-Parganas, West Bengal, India, which is salt tolerant hydro-halophytic species belongs to the family poaceae under the genus Oryza (Figure 1). This wild rice O. coarctata is morphologically distinct from other species of wild rice Oryza rufipogon (Figure 1). Plant height of O. coarctata varies from 90 cm to 195 cm. It is a perennial herb, solid and round stem with branched stalk (culm). The stems are erect herb, soft green in colour covered with hair and having extensive creeping rhizomes. Primary method of propagation is vegetative. Leaves are narrowly linear, thick leathery in texture with short petiole. Leaves are about 25 mm to 40 cm long, 10 to 15 mm broad, coriaceous, spiny margins including short ligule fringed with hairs. The leaves have many ridges and furrows and each ridge contains one small vascular bundle, margins are tuberculate. The leaves are without midrib, distinct from other rice leaves. Leaves are succulent and waxy in nature. Stomata present on the lower leaf surface are sunken type. Leave blades are coriaceous with two types of salt hairs. One type is peg shaped and other type is finger shaped present on upper surface as observed under SEM (Figures 2,3). The salt hairs are unicellular, without cuticle and highly vacuolated. Peg-shaped (spherical globose) salt hairs are arranged all over the leaf surface in ridges and furrows, finger like salt hairs only distributed in the margins of furrows (Figure 3D,E). Leaves are lanceolate in shape, not leathery; margins are not tuberculate, main vascular bundle is prominent in case of O. rufipogon (Figure 1). Both type of salt exclusion structures (salt glands and salt hairs) are found in O. rufipogon too (Figure 4).
The inflorescence is spikelets type and mature flowers are present on the upper end of the spikelet (non-flattened) in O. coarctata. Panicle is up to 20 cm long. Fertile lemma and palea are smooth in surface with any special types of features. The spikelets are narrowly oblong to narrowly ovate, 12.5 to 14.5mm long (including the awn), and obliquely articulated with the pedicel (Figure 2). A unique callus is expanded as a rigid disk-like appearance which is transversely narrowed. Glumes are linear subequal, one-third as long as the spikelet, lacking apparent epidermal characteristics. The lemma apex bears a rigid small awn of about 4 mm long, with microhairs and stomata. Apicular knobs are not present on the lemma apex.
Globose rachilla is lacking in this O. coarctata. The lemma is 7–9 nerved and the palea fivenerved (O. coarctata) but others species have five-nerved lemma and three-nerved palea. In case of O. rufipogon, spikelets are thin linear ranges from 7.95 mm to 8.52 mm long, somewhat smaller than the O. coarctata. Details comparative description about the plant morphology, leaf morpho-anatomy, caryopsis morpho-ultrastructure between two wild rice O. coarctata and O. rufipogon has been summarized in a tabular outline (Table 1).
Caryopsis ultrastructure under SEM study
The whole kernel, caryopsis of O. coarctata has an unusual large embryo (4.5mm to 5mm) relative to the endosperm (8.8mm to 9.5mm) (half the size of the caryopsis) with a somewhat bent apex, and a short petiole-like attachment at the base (Figure 5). Embryo is narrowly elliptic and 4.5 to 5.0mm long. This large embryo has free epiblast large in size and found a distinct cleft like structure in between the coleorhiza and the scutellum (Table 1; Figure 5) but lacks auricles. In contrast to this embryo of O. coarctata, small size embryo (1.0 to 1.5mm long) is observed in O. rufipogon (one-fifth to one-third size in this wild species) without any cleft or free epiblast (Table 1, Figure 6).
Caryopsis has an outer covering of bran layer which is consisting of pericarp, testa and aleurone layer, collectively known as bran layer. Below the aleurone layer, it is the endosperm layer composed of carbohydrate starch molecules in the form of Compound Starch Granule (CSG) along with protein bodies in the wild species, O. coarctata and O. rufipogon
(Table 2). The thickness of pericarp and testa is 2.264μm, and 2.018μm (combining thickness is 4.282μm) respectively in O. coarcata. In O. rufipogon thickness of pericarp and testa is 4.507μm and 4.919μm respectively. Aleurone Layer Cell (ALC) thickness is vary, which is 8.67 x 7.98μm in O. coarctata and 9.25 x 35.25μm in O. rufipogon. Over all bran thickness is 29.70μm to 37.25μm in O. rufipogon and 19.63μm to 20.59μm in O. coarctata. Aleurone cell layer number varies from 1-2 in O. rufipogon and only single layer in O. coarctata, which is also not so prominent (Table 2). Aleurone Grain size (AG) ranges from 0.70 μm to 4.23μm in O. rufipogon and no such AG is found in O. coarctata.
Compound Starch Granule (CSG) size varies from 5.64μm to 16.50μm in O. rufipogon with polygonal (hexagonal) in shape with moderate angularity and 11.991 μm to 17.140μm in O. coarctata, which are spherical (foot ball like) in appearance. Protein Body (PB) abundantly present in O. rufipogon and very few in numbers in O. coarctata. Endosperm porosity is plentiful in O. coarctata but moderate in O. rufipogon.
The Energy Dispersive X-Ray Analysis (EDAX), is an X-ray technique used to identify the elemental composition of materials and which is attached with the SEM system. Only zinc element has been detected in the wild rice O. coarctata (0.28 % weight), not detected in other wild rice O. rufipogon in this present investigation (Figure 7).
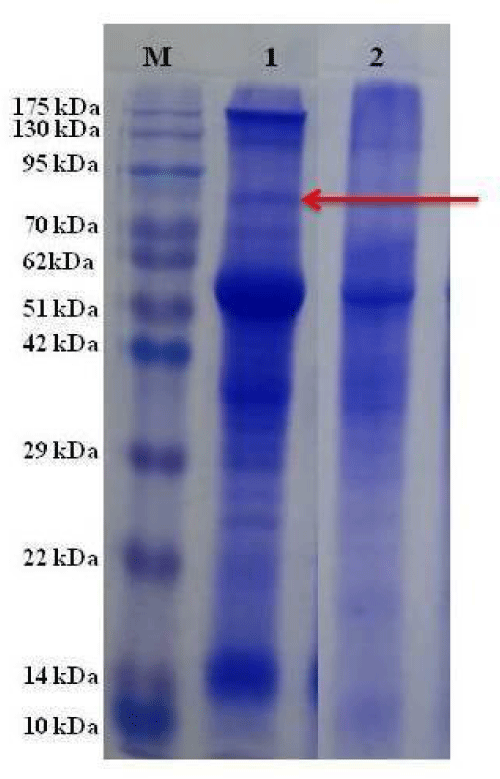
Distinct banding variation was detected in the SDS-PAGE profiling of leaf protein of two wild rice species (Figure 8). Band numbers was more in the wild rice O. rufipogon in compared to O. coarctata in the gel (Figure 8). One extra band was observed at the position in between 75 kDa to 95 kDa in the gel (lane 1), which is not detected in O. coarctata (lane 2).
Discussion
The wild rice species O. coarctata (Roxb.) grow well in the saline soils of river estuaries of India (Bay of Bengal) and other estuaries of Pakistan, Bangladesh, and Myanmar (South Asia). It is a perennial herb that belongs to family poaceae. It is only species in the genus Oryza, which is a halophyte by nature. It is locally called as ‘Uri-dhan’ in India [44] and an important species for ecological succession because it binds with peripheral soils in the mangrove forests. Previously it was placed under the monotypic genus Porteresia coarctata based on morphological dissimilarities between this taxon with other species of rice. Main morphological characteristics of this wild rice are branched stalk (culms), hardy leaves waxy leathery in texture, indistinct articulation below the upper empty glumes, and non-flattened spikelets, embryo morphology, and other characteristics (Table 1, Figure 2). But due to morphological similarities with other wild species such as O. brachyantha, O.granulata and O.schlectheri, and on the molecular biological analysis, it is strongly recommended for retention in the genus Oryza as species O. coarctata [30]. In the present investigation, we have collected wild rice O. coarctata from the mangrove ecosystem of Sundarban Biosphere Reserve, Bay of Bengal (Figure 1), which is highly saline in nature and these salt marsh ecosystem including the intertidal meadow on the river banks is inundated with saline river or sea water (twice a day). Both the wild species are up to 2 meter tall. O. coarctata stem is branched stalk and erect in habitat and perennial, on the other hand O. rufipogon is prostrate (spreading) in habitat sometimes semi-erect (Table 1, Figure 1). Leaves are succulent leathery and waxy in O. coarctata which helps in maintaining relative water of the plant checking transpiration rate, similar to the adaptation seen in the halophytes. Upper leaf surface contains two types of salt exclusion structure such as salt gland (pegshaped) and salt hairs (finger like projection) which are associated to the exclusion of salt ion during high salt stress conditions (Figures 3,4). The salt hairs are unicellular without cuticle and highly vacuolated, can burst in high salt concentration. This finding is consistent with the earlier finding [34]. It was predicted from the experimental support that under high salinity stage number of salt hairs (which is outgrowths of epidermis cells) are increased and secreted out excess salt from the cells. In high salt concentration (300-400 mM NaCl), these salt hairs and salt glands compartmentalize Na+ into vacuals/tonoplast, Na+ toxicity in mesophyll cells are being avoided by this compartmentalization of salt ion in epidermal hairs, a halophytic grass characteristics and maintain low Na:K ratio in the leave cells. This type of ability to maintain a low Na:K ratio in the mesophyll cells, is a consequence of the secretion of ions from the leaf cells (Bal and Dutt 1986) through salt gland and salt hairs. This halophytic wild rice O.coarctata is a saltloving rice species which is able to remove the excess salt through salt glands by the coordination action of various genes [45]. It is facultative halophytes but requires saline environment for initial growth establishment. Therefore, it can be utilized as an important source of genes/alleles and QTLs for salinity and submergence tolerance [46]. In general, O.coarctata is salt tolerant wild rice genotype of saline coastal region of West Bengal and is regarded as a gold mine of abiotic stress tolerant germplasm specifically to study the salt tolerance mechanism and identification of genes which can be used for rice improvement against salinity stress.
Oryza coarctata is an allotetraploid plant (2n=4X=48 chromosomes) with total chromosomal genomic size 665 Mb [47] and consisted of two distinct diploid genomes (KKLL). This halophytic wild rice belongs to secondary gene pool of rice (Oryza sativa). Oryza coarctata has been considered as a precious germplasm because it can withstand salinity up to 300-400 mM (NaCl). There is a scope of salinity tress tolerance trait can be transferred to cultivated rice through conventional breeding [33] or transgenic system. Inflorescence is spikelet type, spikelet is narrowly oblong to narrowly ovate, 12.5 mm to 14.5 mm long, and obliquely articulated with the pedicel. The unique callus is expanded as a rigid, transversely narrowed disk-like structure at the base of spikelet. Globose rachilla is lacking in this O. coarctata but present in O. rufipogon. Lemma and palea is lacking any kind of epidermal features. In O. rufipogon, lemma–palea has so many types of marking features such as prickles, tubercles, and microhairs, these are totally lacking in O. coarctata. The seeds produced by the plant dehisce prematurely and short viability due to recalcitrant type. In case of O. rufipogon, seed viability is long and orthodox nature. Kernel colour is black in O. coarctata, red like kernel is found in O. rufipogon. Husk is yellow colour in O. coarctata, blackish in O. rufipogon (Table 1, Figure 2). The present finding is consisted with the observation of earlier study [48]. Embryo is large in size in O.coarctata (half of the size of caryopsis) with cleft between the scutellum and coleorhiza, no auricle, and the epiblast is very long and separate from the scutellum, in case of O. rufipogon embryo is small in size (one-fifth of the caryopsis) without any cleft (Table 1, Figure 2). Upper surface of the caryopsis is composed of irregular rectangular type grooves in parallel series containing protein body like small spherical structure, which is not found in any other species of Oryza. It is a unique feature observed in this species O. coarctata and first time report of such characteristics in rice species (Figure 5). The pericarp and testa is not fused together in this halophytic species O. coarctata (Table 2; Figure 5), these are partially fused in O. rufipogon and conformed to the typical gramineae arrangement (Table 2; Figure 6). Aleurone layer is not so distinct in O. coarctata, it is distinct of single cell layer in O. rufipogon (Figure 5,6). Seed viability is short lined because due to recalcitrant type in O. coarctata. Aleurone layer has an important role in seed dormancy, viability and germination. In O. coarctata, two important grain traits such as presence of irregular rectangular grooves with protein bodies on the upper surface and indistinct aleurone layer with obscure cellular structure might be the reasons of short seed viability along with large embryo size with cleft between coleoptiles and coleorhizae. We are first time reporting such ultrastructural differentiation between wild rice species O. coarctata and O. rufipogon. There was no earlier report available regarding the ultrastructure of the rice caryopsis in O. coarctata. The endosperm layer composed of two types of starch granules of spherical shape (foot ball like) and different in size. Single Starch Granule (SSG) ranges from 9.14 μm to 11.99μm in diameter, these are loosely arranged (piled up one after another) with void spaces within the parenchyma cells and Compound Starch Granule (CSG) varies from 11.99μm to 17.14μm in diameter (Figure 5) consisting of 5-7 small SSG. Protein bodies (0.576μm to 1.064μm diameter) also observed associated with these starch granules. Parenchyma cell size is large (98.054 x 67.930μm) with thick cell wall (0.7μm to 0.9μm in thickness) and irregular in shape (elliptical/rectangular/ovoid). Pin holes across the starch granule surfaces were obtained in O. rufipogon, O. coarctata, which facilitates enzyme susceptibility and chemical reactivity. It is anticipated that the porosity of the starch granules may assist the digestion of these grains in the human or animal gut [5]. The present investigation of caryopsis ultrastructure analysis will assist in identifying grain traits and starch properties in relation to other histoanatomical features of wild rice species mainly O. coarctata (first time report) for potential genetic enhancement applications. Crop diversity has been reduced drastically and many important traits (biotic and abiotic stress tolerance) has been lost or weakened in course of domestication and cultivation practices. Farmers are selecting only those traits they preferred during agricultural practices and unknowingly (innocently) losing the other important traits (Biotic/abiotic stress tolerance) from the cultivars. It has been demonstrated that cultivated rice O. sativa carries only 10-20% genetic diversity to that of wild rice species [49]. Thus wild rice species are the reservoir of hidden unexploited agriculturally important traits (Biotic/abiotic stress) which are distributed in different agroclimatic conditions and well adapted. It is also imperative to emphasize that isolated populations within species may also hold crucial genes [2,6]. Geenetic diversity within the wild rice species is an essential resource to improve rice production under climate change scenario mainly abiotic stress tolerance [12,50]. Transfer of desired traits from the wild species to cultivated species is not so easy because these wild species are connected with several unwanted weedy traits, like grain shattering trait, poor plant type, poor grain characteristics and very low seed yield. In addition to that, several incompatibility barriers limit the transfer of desired traits related genes from wild species into cultivated species. O. coarctata can be a potential source of genes and other genetic components for improvement of salt tolerance in rice through the application of modern biotechnology [50-57].
Conclusion
The Oryza coarctata (Roxb.) is an extremely halophytic wild rice species highly tolerant to extreme salinity and can withstand salinity levels as high as sea water up to 300-400mM NaCl concentration. It has the ability to thrive and complete their life cycle under high salinity due to their capability to exclude Na+ at the root level, sequester Na+ to the vacuole, and give off excess salt through specialized salt glands in the leaves. It has unusual morphology, habitat (wet saline sites) and distribution to coastal regions of South Asian countries and extremely hydro-halophyte under the genus Oryza. It is assumed that due to its allotetraploid (2n =4x=48, KKLL genome) nature, may provide some clue to its unusual morphology and distribution, and polyploid genomic constitution may assist to adapt in the extremely saline coastal marsh ecosystem. O. coarctata is perennial herb up to 2 meter tall with branched stalk, extremely hydro-halophytic with extensive rhizome system. Leaves are waxy leathery (coriaceous) that are strongly ribbed, with special type of salt glands and salt hairs for the exclusion of excessive salt from the cellular environment to thrive from salinity stress. Embryo size is large with free epiblast including cleft in between the coleorhiza and the scutellum. Embryo is short lived and recalcitrant in nature.
It has an unusual large embryo with a large free epiblast and a distinct cleft between the coleorhiza and the scutellum, short lived recalcitrant in nature. Caryopsis has a bent at tip and petiole like structure at the base. Lemma and palea of O. coarctata is lacking any kind of special features on upper surface mainly tubercles. It is found in all other species of Oryza, a distinctive epidermal out growth and considered as a synapomorphy which support the view of monophyly of the genus Porteresia previously.
Wild species of rice serve as an untapped reservoir of genetic diversity containing agronomically valuable traits that can be used to develop new rice varieties through breeding program. The mangrove associated wild rice O. coarctata can be a good source of salt tolerance gene(s) or QTLs to provide salinity tolerance to improved rice varieties through conventional breeding or through transgenic technology. This salt tolerance species can also furnish C4 like photosynthetic apparatus and mechanism to the C4 rice consortium to develop C4 rice plant to increase the yield potentiality to meet up the food grain demands in 2050 for more than >9 billion people. Breeders and genetic engineers should look in this species very carefully to utilise its C4-like anatomical features and molecular mechanism of photosynthesis in the salinity environment (abiotic stress conditions). Its recalcitrant type seed longevity might be due to irregularities found in the Bran layer components. The bran layer commonly comprises of three distinct layers in other species of rice-pericarp, testa and aleurone layer (altogether called as bran layer). In this species of O. coarctata, aleurone layer is not prominent and does not contain any kind of aleurone grains (AG) like other species. Aleorone layer of the rice bran layer play vital role in seed germination and seed viability. Due to nonperforming aleurone components in the species O. coarctata seed is recalcitrant short lived. Ultrastructure of the caryopsis of O. coarctata (reported first time) has been described in details to provide genetic diversity and variation of the inner histo-anatomical features of such unique traits, which is distinct from that of the other rice species O. rufipogon. Bioprospecting of such genes and proteins coupled with genomic and proteomic approaches remain an exciting area of research in evaluating this plant as a model for salt tolerance for the rice plant.
Authors contribution
SCR was a major contributor in writing the manuscript, proposed the research idea, conceptualized the research design and prepared the final manuscript including the original experimental work, and data recording. AC was involved with material collection, location survey and habitat study, at Sudarban Biosphere Reserve, WB, India.
SCR is thankful to the authority of North Bengal University for providing Laboratory and Experimental Rice Field facilities to carry out rice breeding and conservation of local rice varieties in the Department of Botany, NBU. SCR is also thankful to the USIC Department, University of North Bengal for providing SEM facility.
- Kellogg EA (2009) The Evolutionary History of Ehrhartoideae, Oryzeae, and Oryza. Rice 2: 1–14. Link: https://bit.ly/3wFylZN
- Jacquemin J, Bhatia D, Singh K, Wing RA (2013) The International Oryza Map Alignment Project: development of a genus-wide comparative genomics platform to help solve the 9 billion-people question. Curr Opin Plant Biol 16: 147-156. Link: https://bit.ly/3tgfvqd
- Vaughan DA (1994) The wild relatives of rice: a genetic resources handbook. IRRI, Los Baños, PO Box 933, Manila 1099, Philippines 137. Link: https://bit.ly/2Rc2ZJU
- Sanchez PL, Wing RA, Brar DS (2013) The Wild Relative of Rice: Genomes and Genomics. In: Zhang Q, Wing R (ed) Genetics and Genomics of Rice. Plant Genetics and Genomics: Crops and Models, vol 5. Springer, New York 9-25. Link: https://bit.ly/2RfTQA1
- Kasem S, Waters DLE, Rice NF, Shapter FM, Henry RJ (2011) The Endosperm Morphology of Rice and its Wild Relatives as Observed by Scanning Electron Microscopy. Rice 4: 12–20. Link: https://bit.ly/3dMDmHo
- Atwell BJ, Wang H, Scafaro AP (2014) Could abiotic stress tolerance in wild relatives of rice be used to improve Oryza sativa? Plant Sci 215: 48-58. Link: https://bit.ly/3s2uXoc
- Godfray HC, Beddington JR, Crute IR, Haddad L, Lawrence D, et al. (2010) Food security: the challenge of feeding 9 billion people. Science 327: 812-818. Link: https://bit.ly/3uC9HHK
- Foley J, Ramankutty N, Brauman KA, Cassidy ES, Gerber JS, et al. (2011) Solutions for a cultivated planet. Nature 478: 337-342. Link: https://go.nature.com/3myKAT6
- Palmgren MG, Edenbrandt AK, Vedel SE, Andersen MM, Landes X, et al. (2015) Are we ready for back-to-nature crop breeding? Trends Plant Sci 20: 155-164. Link: https://bit.ly/3s3qlOD
- Singh PK, Venkatesan K, Swarnam TP (2018) Rice Genetic Resources in Tropical Islands. In (Ed): Sivaperuman Chandrakasan, Awnindra Kumar Singh, Ayyam Velmurugan, Iyyappan Jaisankar. Biodiversity and Climate Change Adaptation in Tropical Islands 355-384. Link: https://bit.ly/2PH3DyW
- Mueller N, Gerber J, Johnston M, Ray DK, Ramankutty N, et al (2012) Closing yield gaps through nutrient and water management. Nature 490: 254–257. Link: https://go.nature.com/3t2OJkZ
- Sang T, Ge S (2013) Understanding rice domestication and implications for cultivar improvement. Curr Opin Plant Biol 16: 139-146. Link: https://bit.ly/3dMpk8B
- Hong Y, Zhang H, Huang L, Li D, Song F (2016) Overexpression of a stress-responsive NAC transcription factor gene ONAC022 improves drought and salt tolerance in rice. Front Plant Sci 7: 4-14. Link: https://bit.ly/2Q6qhAs
- Menguer PK, Sperotto RA, Ricachenevsky FK (2017) A walk on the wild side: Oryza species as source for rice abiotic stress tolerance. Genet Mol Biol 40: 238-252. Link: https://bit.ly/3mvx4zT
- Zhou Y, Yang P, Cui F, Zhang F, Luo X, et al. (2016) Transcriptome Analysis of Salt Stress Responsiveness in the Seedlings of Dongxiang Wild Rice (Oryza rufipogon Griff.). PLoS One 11: e0146242. Link: https://bit.ly/3t2ekdC
- Munns R, M Tester (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59: 651-681. Link: https://bit.ly/3d2QpW4
- Sengupta S, Majumder A L (2010) Porteresia coarctata (Roxb.) Tateoka, a wild rice: A potential model for studying salt-stress biology in rice. Plant Cell Environ 33: 526-542.Link: https://bit.ly/2RcJpgE
- Roy SJ, Negrão S, Tester M (2014) Salt resistant crop plants. Curr Opin Biotechnol 26: 115-124. Link: https://bit.ly/2OCRt9I
- Mickelbart M, Hasegawa P, Bailey-Serres J (2015) Genetic mechanisms of abiotic stress tolerance that translate to crop yield stability. Nat Rev Genet 16: 237-251. Link: https://bit.ly/3fTBj75
- Ismail AM, Singh US, Singh S, Dar MH, Mackill DJ (2013) The contribution of Submergence-Tolerant (Sub1) rice varieties to food security in flood-prone rainfed lowland areas in Asia. Field Crops Res 152: 83-93. Link: https://bit.ly/3e2RcWn
- Ren ZH, Gao JP, Li LG, Cai XL, Huang W, et al. (2005) A rice quantitative trait locus for salt tolerance encodes a sodium transporter. Nat Genet 37: 1141-1146. Link: https://bit.ly/3dNQtrJ
- Platten JD, Egdane JA, Ismail AM (2013) Salinity tolerance, Na+ exclusion and allele mining of HKT1;5 in Oryza sativa and O. glaberrima: many sources, many genes, one mechanism?. BMC Plant Biol 13: 32. Link: https://bit.ly/3uG6p6l
- Hauser F, Horie T (2010) A conserved primary salt tolerance mechanism mediated by HKT transporters: a mechanism for sodium exclusion and maintenance of high K(+)/Na(+) ratio in leaves during salinity stress. Plant Cell Environ 33: 552-565. Link: https://bit.ly/3t8zt60
- Scafaro AP, Haynes PA, Atwell BJ (2010) Physiological and molecular changes in Oryza meridionalis Ng., a heat-tolerant species of wild rice. J Exp Bot 61: 191-202. Link: https://bit.ly/3g2VvTQ
- Molla KA, Debnath AB, Ganie SA, Mondal TK (2015) Identification and analysis of novel salt responsive candidate gene based SSRs (cgSSRs) from rice (Oryza sativa L.). BMC Plant Biol 15: 122. Link: https://bit.ly/2QdVXEg
- Thomson MJ, de Ocampo M, Egdane J, RahmanMA, Sajise AG, et al (2010) Characterizing the Saltol Quantitative Trait Locus for Salinity Tolerance in Rice. Rice 3: 148–160. Link: https://bit.ly/3218PzW
- Bal AR, SK Dutta (1986) Mechanisms of salt tolerance in wild rice Oryza coarctata (Roxb.). Plant Soil 92: 399-404. Link: https://bit.ly/3uDAlA6
- Lu F, JS Ammiraju, A Sanyal et al (2009) Comparative sequence analysis of MONOCULM1 -orthologous regions in 14 Oryza genomes. Proc Natl Acad Sci USA 106: 2071-2076.
- Sarkar RH, Samad MA, Seraj ZI, Hoque MI, Islam AS (1993) Pollen tube growth in crosses between Porteresia coarctata and Oryza sativa. Euphytica 69: 129-134. Link: https://bit.ly/3fW7HpG
- Lu BR, Ge S (2003) Oryza coarctata:the name that best reflects the relationships of Porteresia coarctata (Poaceae: Oryzeac). Nordic J Bot 23: 555-558. Link: https://bit.ly/3dTyOPE
- Lu BR, Ge S (2003) Oryza coarctata:the name that best reflects the relationships of Porteresia coarctata (Poaceae: Oryzeac). Nordic J Bot 23: 555-558. Link: https://bit.ly/3dTyOPE
- Garg R, Verma M, Agrawal S, Shankar R, Majee M, et al. (2014) Deep transcriptome sequencing of wild halophyte rice, Porteresia coarctata, provides novel insights into the salinity and submergence tolerance factors. DNA Res 21: 69-84. Link: https://bit.ly/3my5cv5
- Jena KK (1994) Production of intergeneric hybrid between Oryza sativa L. and Porteresia coarctata. Curr Sci 67: 9-10. Link: https://bit.ly/3wQFK8O
- Sengupta S, Majumder AL (2009) Insight into the salt tolerance factors of a wild halophytic rice: Porteresia coarctata: a physiological and proteomic approach. Planta 229: 911-928. Link: https://bit.ly/2OCSjDo
- Kizhakkedath, P, Jegadeeson V, Venkataraman G, Parida A (2015) A vacuolar antiporter is differentially regulated in leaves and roots of the halophytic wild rice Porteresia coarctata (Roxb.)Tateoka. Mol Biol Rep 42: 1091-1105. Link: https://bit.ly/2QagadO
- Joshi R, Mangu VR, Bedre R, Sanchez L, Pilcher W, et al. (2015) Salt adaptation mechanisms of halophytes: improvement of salt tolerance in crop plants. In: GK Pandey (ed.), Elucidation of Abiotic Stress Signaling in Plants. Springer, New York 243-279. Link: https://bit.ly/3uDAW4O
- Tian L, Tan L, Liu F, Cai H, Sun C (2011) Identification of quantitative trait loci associated with salt tolerance at seedling stage from Oryza rufipogon. J Genet Genomics 38: 593-601. Link: https://bit.ly/3t3cgSL
- Xiao N, Huang WN, Li AH, Gao Y, Li HY, et al. (2014) Fine mapping of the qLOP2 and qPSR2-1 loci associated with chilling stress tolerance of wild rice seedlings. Theor Appl Genet 128: 173-185. Link: https://bit.ly/2RlaRsN
- Hattori Y, Nagai K, Furukawa S, Song XJ, Kawano R, et al. (2009) The ethylene response factors SNORKEL1 and SNORKEL2 allow rice to adapt to deep water. Nature 460: 1026–1030. Link: https://bit.ly/2OBapFO
- Shapter FM, Henry RJ, Lee LS (2008) Endosperm and starch granule morphoogy in wild cereal relatives. Plant Genetic Resources 15. Link: https://bit.ly/3mvoMb2
- Wu X, Liu J, Li D, Liu CM (2016) Rice caryopsis development I: Dynamic changes in different cell layers. J Integr Plant Biol 58: 772-785. Link: https://bit.ly/3mwa4kd
- Wu X, Liu J, Li D, Liu CM (2016) Rice caryopsis development II: Dynamic changes in the endosperm. J Integr Plant Biol 58: 786-798. Link: https://bit.ly/3t6aYX3
- Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680-685. Link: https://bit.ly/3dI1laE
- Vaughan D, Kadowaki KI, Kaga A,Tomooka N (2005) Eco-genetic diversification in the genus Oryza: implications for sustainable rice production. IRRI, 44-46.
- Yu J, Chen S, Zhao Q, Wang T, Yang C, et al. (2011) Physiological and proteomic analysis of salinity tolerance in Puccinella tenuiflora. J Proteome Res 10: 38523870. Link: https://bit.ly/3dRl1sA
- Chowrasia S, Rawal HC, Mazumder A, Gaikwad K, Sharma TR, et al. (2018) Oryza coarctata Roxb. In: Mondal T., Henry R. (eds) The Wild Oryza Genomes. Compendium of Plant Genomes. Springer, Cham 87-104.
- Mondal TK, Rawal HC, Gaikwad K, Sharma TR, Singh NK (2017) First de novo draft genome sequence of Oryza coarctata, the only halophytic species in the genus Oryza. F1000Res 6: 1750. Link: https://bit.ly/2PFW2kd
- Dikshit N, SS Malik, RS Rana (1993) Porteresia coarctata (Roxb.) Tateoka, A Wild Relative of Rice. J Eco Taxo Bot I7:1542-1551.
- Zhu Q, Zheng X, Luo J, Gaut BS, Ge S (2007) Multilocus analysis of nucleotide variation of Oryza sativa and its wild relatives: severe bottleneck during domestication of rice. Mol Biol Evol 24: 875-88. Link: https://bit.ly/3wINfOM
- Gross B, Zhao Z (2014) Archaeological and genetic insights into the origins of domesticated rice. Proc Natl Acad Sci USA 111: 6190–6197. Link: https://bit.ly/3myVTL3
- Parida A, K Praseetha (2005) Antiporter gene from Porteresia coarctata for conferring stress tolerance. Patent WO/2007/015268.
- Das-Chatterjee A, Goswami L, Maitra S, Dastidar KG, Ray S, et al. (2006) Introgression of a novel salt-tolerant L-myo-inositol 1-phosphate synthase from Porteresia coarctata (Roxb.) Tateoka (PcINO1) confers salt tolerance to evolutionary diverse organisms. FEBS Lett 580: 3980-3988. Link: https://bit.ly/2PEGf57
- Goswami S, Sengupta S, Mukherjee S, Ray S, Mukherjee R, et al. (2014) Targeted expression of L-myo-inositol 1-phosphate synthase from Porteresia coarctata (Roxb.) Tateoka confers multiple stress tolerance in transgenic crop plants. J Plant Biochem Biotechnol 23: 316-330. Link: https://bit.ly/3wDKDSp
- Mondal TK, Rawal HC, Chowrasia S, Varshney D, Panda AK, et al. (2018) Draft genome sequence of first monocot-halophytic species Oryza coarctata reveals stress-specific genes. Sci Rep 8: 13698. Link: https://go.nature.com/3dMsQQl
- Tuguo T (1964) Notes on some grasses. XVI. Embryo structure of the genus Oryza in relation to the systematic. Amer Jour Bot 51: 539-543. Link: https://bit.ly/3fVir7v
- Trait Locus for Salinity Tolerance in Rice. Rice 3: 148-160.
- Zhang F, Luo X, Zhou Y, Xie J (2016) Genome-wide identification of conserved microRNA and their response to drought stress in Dongxiang wild rice (Oryza rufipogon Griff.). Biotechnol Lett 38: 711-721. Link: https://bit.ly/3s4zgzg
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.







 Save to Mendeley
Save to Mendeley
