Open Journal of Environmental Biology
Nutrition cycles in sessile oak (Quercus petraea Liebl.), Norway spruce (Picea abies L.Karst) and European beech (Fagus sylvatica L.) stands from Central Romania
Raluca-Elena Enescu, Alexandru Liviu Ciuvăț*, Lucian Dincă, Carmen Iacoban, Elena Deleanu and Cristian Anghelus
Cite this as
Enescu RE, Ciuvăț AL, Dincă L, Iacoban C, Deleanu E, et al. (2022) Nutrition cycles in sessile oak (Quercus petraea Liebl.), Norway spruce (Picea abies L.Karst) and European beech (Fagus sylvatica L.) stands from Central Romania. Open J Environ Biol 7(1): 026-032. DOI: 10.17352/ojeb.000031Copyright
© 2022 Enescu RE. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.In Romania, the sessile oak is the widest spread species of all native oak species, forming a lower story of vegetation. The European beech and Norway spruce cover 50% of the Romanian forest area. The purpose of this study was to analyze the variability of the leaf nutrients, as well as of the soil nutrients, for sessile oak, Norway spruce and European beech stands situated under different site conditions. The studied sites are located in the southern part of the Carpathian Mountains at altitudes varying from 600 m to 1300 m. Leaves samples were collected from twenty sessile oak trees, in three moments during the growing season. The same eight elements in total form were determined for soil and leave too: N, P, K, Ca, Mg, Fe, Cu and Zn. Leaves samples for beech and spruce were collected in the II Level Intensive Monitoring Grid of the ICP Forests Programme. The paper focuses on the seasonal concentration variation of eight macro elements in leaves and the correlations between the concentration of the macro elements in soil and litter. In addition, the study highlighted the degree of influence exerted by some soil properties on the foliar concentration of nutrients (through multiple regressions). The obtained results show different mobility of the elements and a greater variability was noticed in the case of certain elements. The same three soil properties were noticed to influence the foliar nitrogen concentration.
Introduction
Romanian forests are situated mainly in the mountainous regions (70%), while hills and especially plains are covered by much smaller areas of forests. European beech (Fagus sylvatica L.) and Norway spruce (Picea abies L. Karst) constitute approximately 50% of Romanian forest habitats (of which 31% is beech), summing up to 3 mils. ha [1], while sessile oak (Quercus petraea Liebl.) covers 10,5% of the afforested area and is the widest spread species of all native oak species [2].
Knowing the nutritional status of these important species is crucial as it is indicative of ecosystem processes that help in understanding and monitoring forest health [3]. With the purpose to evaluate the nutritional status and the potential nutritional deficiencies, the available quantity of nutrients in the soil was determined, as well as the concentration of leaf nutrients (foliar analysis or foliar diagnosis) and foliar ionic reports [4]. The chemical analysis of the soil offers indicators of the potential availability of nutrients [5,6], under certain conditions favorable to absorption, whereas the analyses made either on leaves or on other plant parts, only reflected the current nutritional status. The seasonal variations in the concentration of nutrients, in the leaves of the perennials (forest trees) are relatively small, in comparison with those of the annual species [7].
This paper aims to highlight the evolution of foliar nutrition cycles in the three main Romanian forest tree species in relation to litter and soil properties.
Materials and methods
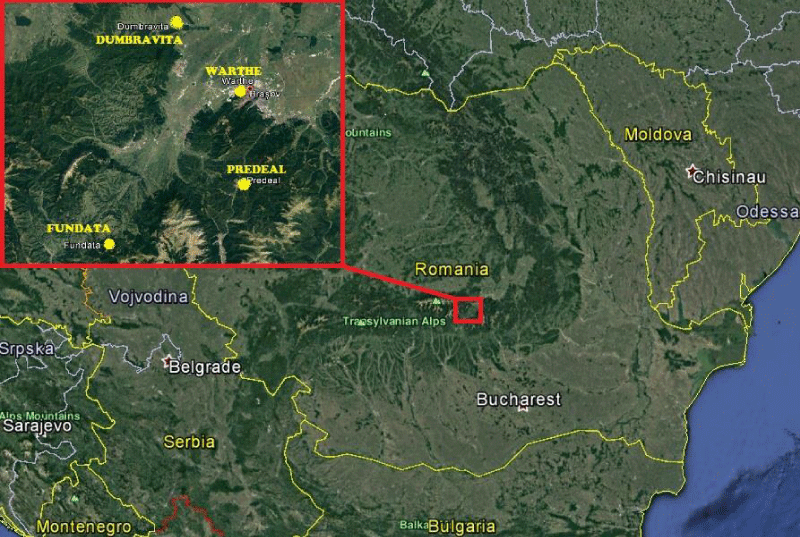
The research was located in four different forest stands: one oak stand was selected in the Cristian Piedmont (Warthe hill near the city of Brasov) and another in the Vlădeni Piedmont (near Dumbravita village), while for European beech and Norway spruce the researches were conducted in core plots Fundata (beech) and Predeal (spruce), both of them belonging to the II Level Intensive Monitoring Grid of the ICP Forests Programme. Fundata core plot is located at 1300 m altitude, has a southern exposition and the stand is 50 years old, while the Predeal Core plot is situated at 1185 m altitude, the exposition is East and the stand age is of 95 years (Figure 1).
In order to examine the nutritional status of sessile oak, the research was conducted at the soil level and also at the leaf level. The soil samples were collected on genetic horizons. The leaf samples were collected from 20 trees (5 around each soil profile). The leaves were collected from apparently healthy, mature trees, which belong to the Ist and IInd Kraft classes and to their upper third, in compliance with the international methodology in force [8].
The number of five macro elements was determined (N, P, K, Ca, Mg) in the laboratory [9] for the soil samples, as well as three microelements (Cu, Fe, Zn), in their total form. The concentrations of macro and microelements (excepting N) were determined by the soil treatment with an oxidant mixture, consisting of HNO3, HClO4, H2SO4, and dosing at the spectrometer in atomic adsorption. The concentration of organic carbon was determined by the method of wet oxidation and titrimetric dosing, also called the Walkley-Black method. In order to determine the concentration of total nitrogen, the Kjeldahl method was used; and with its help, as well as through the concentration of organic carbon, the pedogenetic index, the ratio carbon-nitrogen (C/N) was calculated. The potentiometric method in aqueous soil suspensions was used for the soil pH. The sum of exchangeable bases and total exchangeable hydrogen were determined with the Kappen method; the bases were extracted with hydrochloric acid; while the hydrogen cations, were with the calcium-acetate solution. The total cationic exchange capacity resulted by summing the exchangeable bases and the exchangeable hydrogen.
For the leaf samples, the same 8 nutrients were determined, as in the case of the soil samples. The applied method consists of the wet mineralization of the foliar samples with strong acids (HCl, HNO3) and then, determining the concentration of nutrients, by reading at the spectrometer in atomic absorption. The nitrogen concentration was determined by the Kjeldahl method while the phosphorus concentration, was by colorimetric dosing.
In order to examine the nutritional status of European beech and Norway spruce, the research was conducted at the soil level (mineral and organic horizon), litterfall level and leaf level. For these two species, the methodology is that of ICP Forests Level II intensive monitoring (2010).
Litterfall samples sorted into fractions were dried at maximum temperatures of 70 °C, for 24 hours, before grinding. A subsample of powder, not chemically analyzed, was used for moisture concentration determination, by drying at 105 °C. All results were corrected considering the moisture concentration. Open digestion with HNO3 / H2SO4 was used for litterfall pre-treatment. For analysis of calcium, magnesium and potassium, the AAS flame technique was used, with acetylene/air on Perkin Elmer AAS Analyst 700. Phosphorus was determined by UV/VIS spectrometry, with the molybdenum blue method. Carbon, nitrogen and sulphur analysis were performed using a LECO Truspec CHNS analyzer. In some cases, when the analyzer could not be used, Gerhardt Kjeldahl digestion and titration for nitrogen determination were applied.
Results
The seasonal variation of the 8 nutrients in the sessile oak leaves, is shown in Figures 2-9.
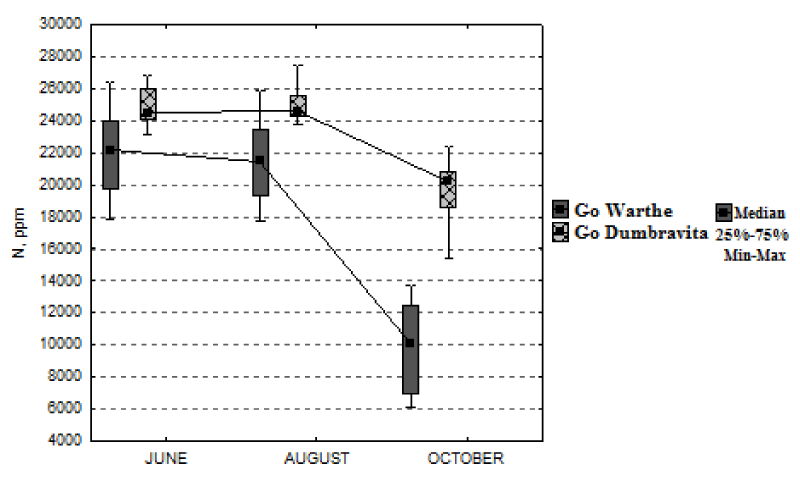
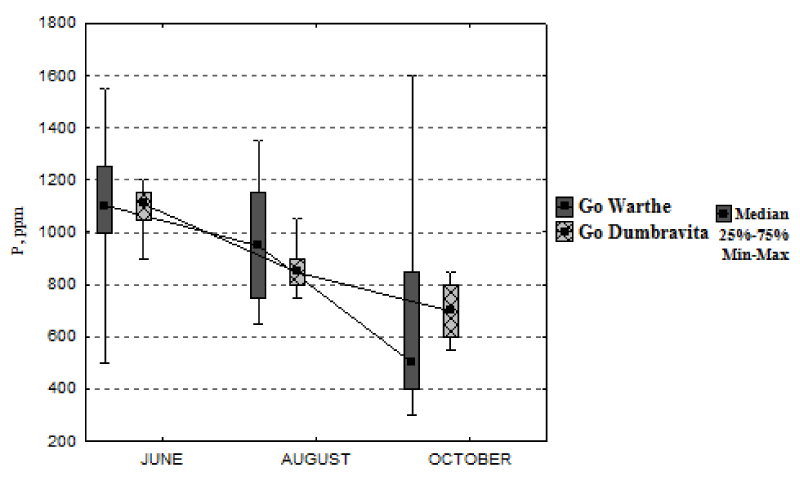
We first notice, in Figure 2, a common tendency of decrease in the foliar-nitrogen concentration, at the end of the growing season, for both locations. From June until August, the nitrogen concentration remains approximately constant. For the sessile oak from the Warthe stand, it decreases by 55%, from June, until October; and for the sessile oak from the Dumbrăvița stand, it drops by 17%, if we relate to the value of the median. In the Warthe stand, the leaves were collected on October 17th; and in the Dumbrăvița stand, on October 9th; hence at a difference of 8 days. Even if the difference is of 8 days, the maximal foliar-nitrogen concentration, determined for the Warthe stand reaches approximately 14000 mg kg-1; whereas, at Dumbrăvița, the minimal concentration approaches the value of 15000 mg kg-1. Just as the nitrogen concentration, the phosphorus concentration follows the same trend, decreasing during the growing season, for both stands (Figure 3). The minimum concentration of phosphorus is much lower for the Warthe stand (approximately 300 mg kg-1) than the Dumbrăvița stand (approximately 550 mg kg-1). The phosphorus concentration drops by 55% for Warthe, from June, until October; and by 37% for Dumbrăvița.
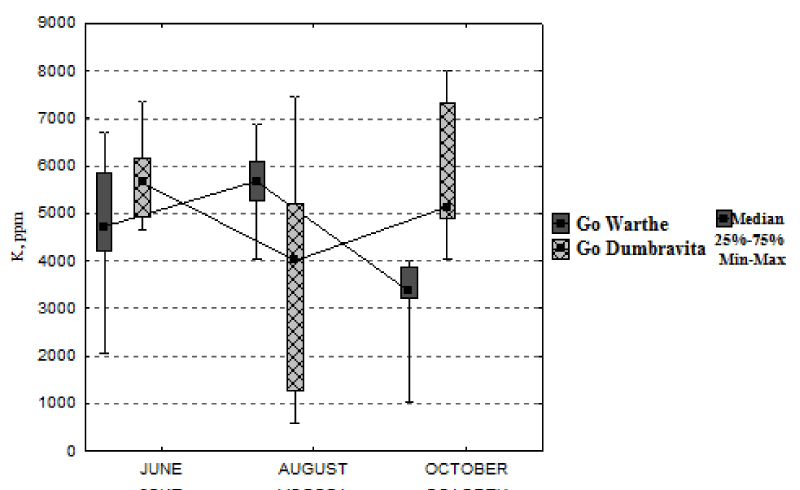
For the sessile oaks from Warthe, the potassium concentration drops, in October, by 29%; and barely decreases (9%) for the one at Dumbrăvița (Figure 4). From June, until August, different (even opposite) variations in the potassium concentration, were recorded. For Warthe, the concentration of potassium increases by 20%, from June, until August, for Dumbrăvița it decreases by 29%, for the same interval, while the concentration of foliar potassium increases by 7% for Warthe and it drops by 21% for Dumbrăvița,
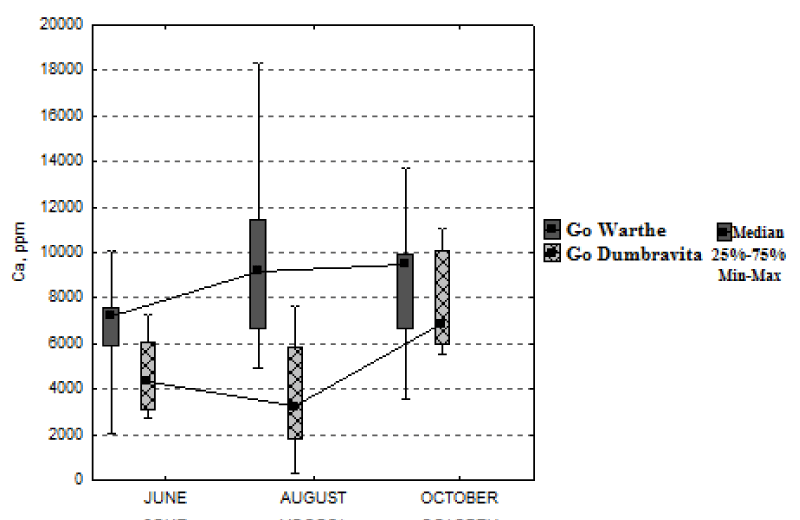
Similar to the case of the potassium variation, for the foliar-calcium concentration, a different situation was recorded for each of the two stands under study (Figure 5). The calcium concentration for the sessile oak from the Warthe stand increases by 31%, from June, until October; and by 58% for the sessile oak from the Dumbrăvița stand. For both locations, the calcium concentration is greater at the end of the season, than at its beginning; quite the opposite of the concentrations of nitrogen, phosphorus and potassium.
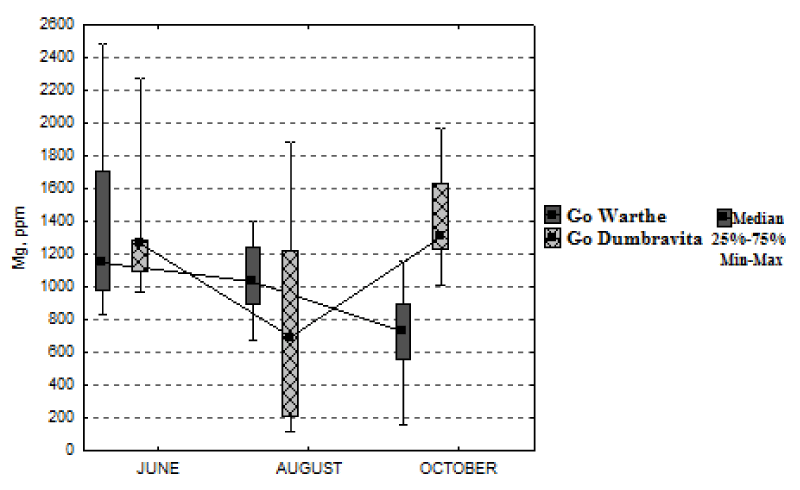
Also, the magnesium concentration has different dynamics in the growing season (Figure 6). Thus, two different situations were recorded: for Warthe, the foliar-magnesium concentration decreases from June, until October, by 36%; while for Dumbrăvița, it increases by only 4%.
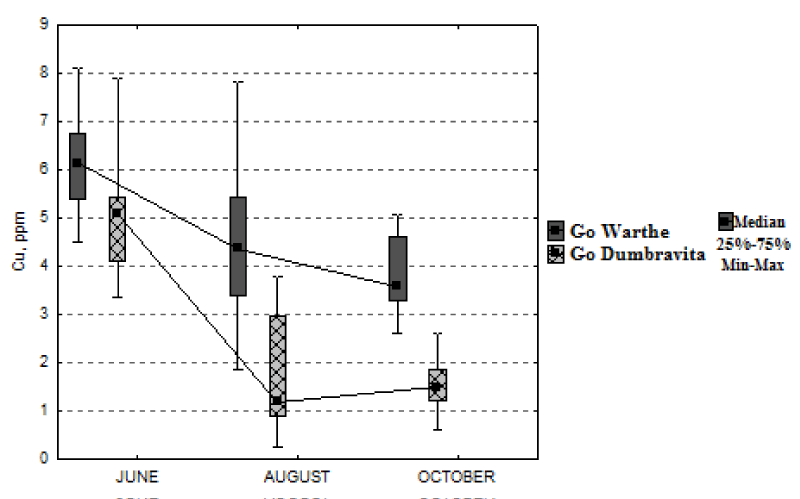
Regarding copper concentration, one can see, from Figure 6, that it has similar dynamics for both locations. For the sessile oak situated on Warthe Hill, the copper concentration drops, from June until October, by 42%; and by 71%, for the sessile oak from Dumbrăvița. The decrease continues throughout the growing season, for both locations, except the interval August-October, for the sessile oak from Dumbrăvița, where there is a slight increase (0.30 mg kg-1).
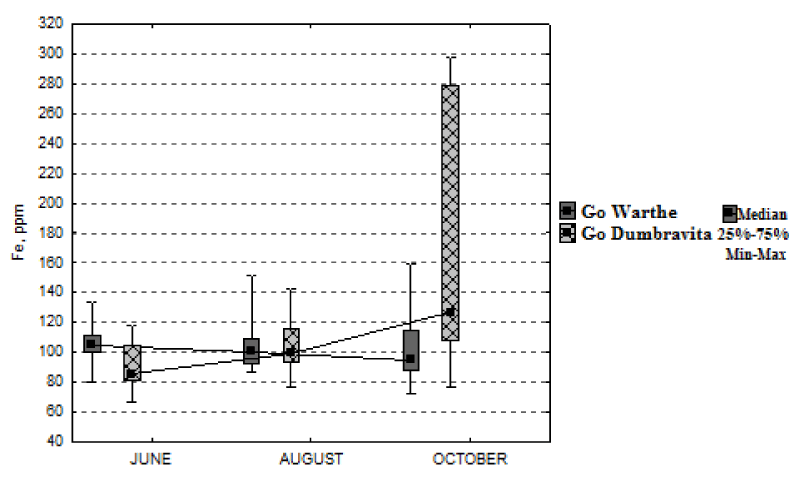
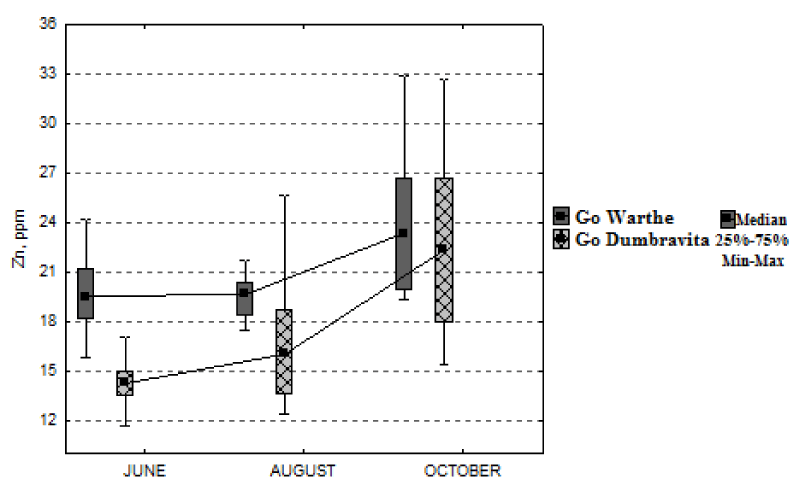
In Figure 8, it can be observed that the variation of iron concentration is low, during the growing season, if we relate it to the value of the median. Thus, for the sessile oak from Warthe, it dropped, during the interval June-October, by 9% (9.74 mg kg-1); and, for the one from Dumbrăvița, it increased, by 49% (41.46 mg kg-1). As regards the foliar-zinc concentration, one can easily see, from Figure 9, that it follows an approximately constant trend, from June, until August; and then, a slight increase, until October, for both locations. For the sessile oak from Warthe stand, the increase, in October, as against the month of June, is 20% (3.87 mg kg-1); and for the one from Dumbrăvița stand, 57% (8.09 mg kg-1).
The physical and chemical properties of the soils from researched stands are shown in Table 1. All four analyzed soils belong to the Luvisols class, luvisol type (according to WRB classification). One profile belongs to the typical subtype; and three of them to the albic subtype (according to Romania’s Soil Taxonomy System, SRTS 2003).
In the top layer (Ao organic-mineral horizon), the soil reaction is strongly acidic. The supply of organic carbon and nitrogen is good and very good. The total cationic exchange capacity is small.
In terms of the degree of base saturation, the studied luvisols are oligo-monobasic. With respect to the assimilated potassium concentration, it varies from middle to high. As regards the mobile phosphorus concentration, one can see that the researched soils show a deficit, this concentration varying from extremely low to low.
Likewise, the correlations between the concentrations of nutrients in the soil and in the leaf were studied. For the soil, the concentration of elements in total form, in horizon A and in horizon B, was considered (Table 2).
Table 2, it can be observed the greater number of correlations between the concentration of elements in the leaf, and the ones in the A horizon. Significant correlations for the calcium element are recorded only in August. The foliar-magnesium concentration, in October, with the one in the soil, achieves significant correlations, also given the fact that, for magnesium, a deficiency at the foliar level is recorded. Copper is another element that marks significant positive correlations, in August and October. The emerging negative correlations show, in fact, an inverse relationship between the concentration of an element in the soil and the one in the leaf, possibly due to some other factors, that for now remain unknown.
Regarding the significant correlations, for the calcium element in the soil, respectively leaf, they are the same, for both A and B horizons. Magnesium achieves, in its turn, significant correlations, yet in June, this time, a negative correlation. The total concentration of magnesium in the B horizon is much greater than the one in the A horizon. The copper in the B horizon achieves a single more significant correlation, with the foliar concentration from October.
Table 3 shows the degree of influence exerted by some soil properties on the foliar concentration of nutrients, during the three growing season’s moments. This degree of influence is rendered by the value of the coefficient of determination R2 and the intensity of the connection between the considered variables, by the multiple-correlation coefficient R.
Only those properties which showed statistical support and which influenced the dependent variable were kept in the regression analysis.
In this way, it can be observed that, for the studied species in June, the variation in two of the eight foliar nutritive elements (nitrogen, zinc) is explained in a proportion of 65%, respectively 71%, by three soil properties, namely the exchange capacity for hydrogen, the organic carbon concentration and the carbon-nitrogen ratio.
The value of the multiple-correlation coefficient (R) indicates a strong-intensity connection, between the aforementioned properties and the foliar concentration of nitrogen and zinc. In August, middle and strong-intensity connections were noticed between the soil properties and four of the eight foliar nutritive elements. The coefficient of determination with the highest value (0.699) shows that approximately 69% of the variation in the foliar-nitrogen concentration is explained by the exchange capacity for hydrogen, the organic carbon concentration and the carbon-nitrogen ratio. In October, the strongest connections between the soil properties and foliar concentrations of nutrients are recorded.
The strongest connection (R = 0.919), was noticed between the foliar concentration of nitrogen and the exchange capacity for hydrogen, the organic carbon concentration, and the carbon-nitrogen ratio. Nevertheless, at the end of the growing season, the foliar-nitrogen concentration decreases, triggering the intensity of this connection. The same soil properties influence, in a proportion of 69%, respectively 74%, the variation of the potassium and copper concentrations. The variation of the magnesium concentration is explained in a proportion of 73% by the exchange capacity for bases, the degree of base saturation, and the organic carbon concentration.
Foliar analysis for Fagus sylvatica in the last 15 years reveals imbalanced nutrition with periodic deficiencies in nitrogen (N), potassium (K) and calcium (Ca) concentrations (Table 4).
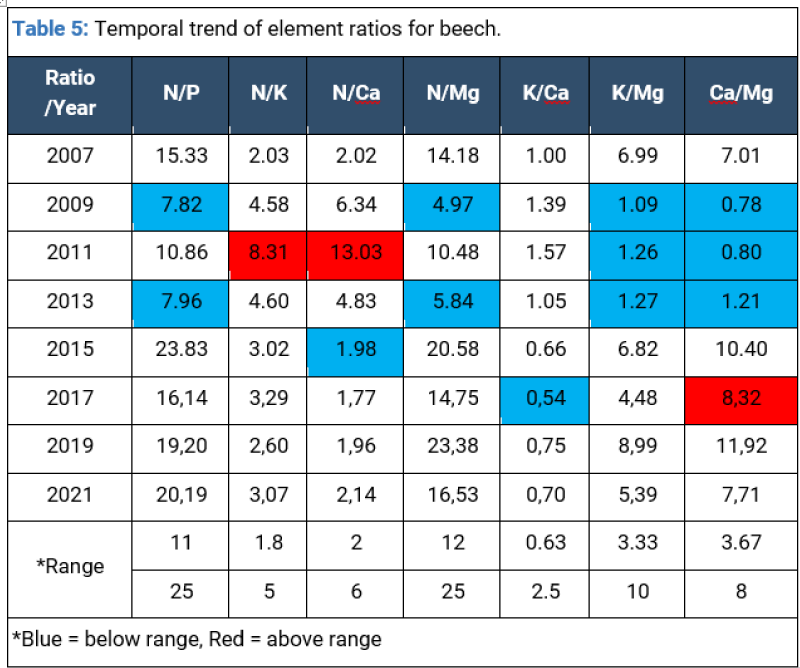
The reduction in N, K and Ca uptake in the first half of the period can be linked to atmospheric CO2-enrichment which leads to a decrease in all foliar element concentrations by a dilution effect as a result of increased biomass [10], but also to the fluctuation of atmospheric deposition process. Sulphur (S), magnesium (Mg) and phosphorus (P) high concentrations are mainly related to soil high base saturation and mycorrhizae activity (Table 5).
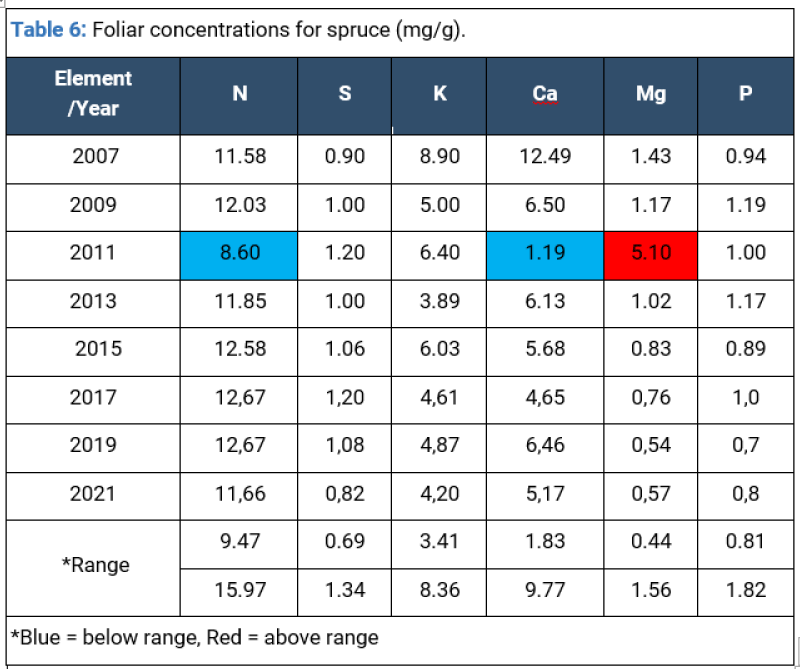
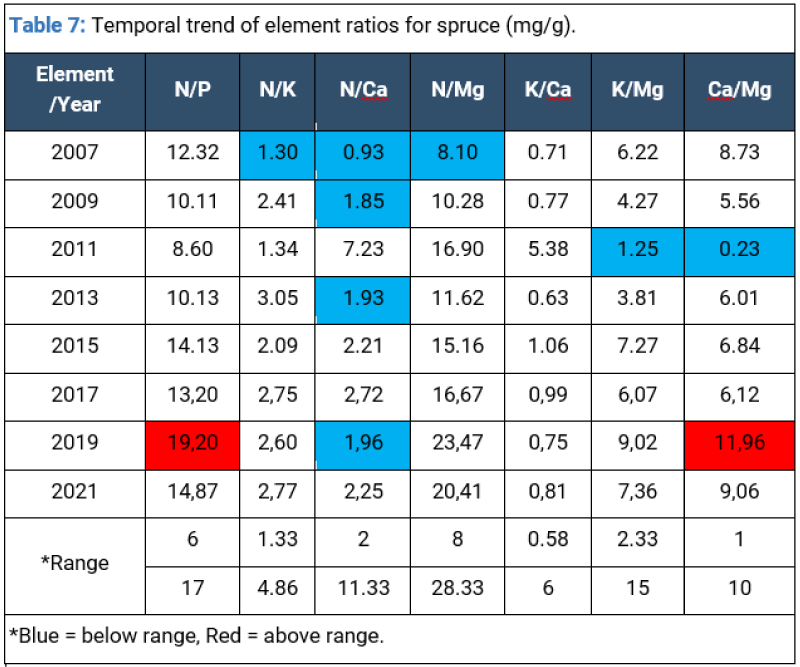
Needle analysis for Picea abies in the past decade shows that K, Ca and Mg exceeded normal range concentrations largely caused by modifications in the level of nitrate leeching induced by the fluctuating level of precipitations (periodic droughts), followed by a gradual stabilization of nutrition in more recent years. Soil humidity and acidity levels determine the availability of K and Ca, which, in turn, influence plant physiological processes (especially transpiration) (Tables 6,7).
Discussion
Referring to the seasonal variation of nitrogen concentration, Parascan & Danciu 2001 [11] noticed that the nitrogen migrates from cells, as they grow old, while within old organs, the nitrogen concentration is lower than in the young ones. A possible explanation would be the higher altitude where the sessile oak on the Warthe Hill (735 m) is situated, as against the one in the Vlădeni Piedmont (611 m); hence a shorter period of the growing season, at higher altitudes, which means earlier senescence, at higher altitudes; the literature quotes that, before the fall of the leaves, high quantities of nitrogen migrate towards the branches and the stalks – approximately 52% [11]. In addition to nitrogen, the literature quotes that, before the fall of the leaves, approximately 27% of the foliar-phosphorus concentration, migrates [11]. In the current study, in both stands, the phosphorus concentration migrated towards other plant parts, which is higher than the one quoted in the literature. Potassium, along with phosphorus and nitrogen, belongs to the category of nutrients with high tissue mobility, which migrate, at the end of the growing season, towards other structures [4,12,13].
The foliar calcium concentration increases from June to October and this is due to the fact that the calcium nutrient has low tissue mobility and blocks in the leaf [4]. Calcium can be found in greater quantities in tissues with lower vital activity [14]. Thus, one can see that the leaves of the sessile oak from the Warthe stand, due to their higher calcium concentration in October, are more inactive than the ones of the sessile oak from the Dumbrăvița stand, where the growing period is longer. In this way, both for calcium and for potassium, great variations can be noticed between individuals, even if they exist in the same forest site conditions. Other researches show that this great variation in the calcium concentration, from one individual to another, might be owed to a certain genetic variability [15]. The studies aimed at examining the nutritional status of the trees, due to the different mobility of the elements and the great variability, among the individuals, should preferably focus on physiology aspects or on the individual genetic characteristics, so that the study might fulfill its purpose as rigorously as possible [16].
The significant correlation between foliar magnesium concentration and the one from soil could be an unusual situation because the literature specifies that, in forest soils, magnesium deficiencies are very rare and that the trees can extract this nutrient even from the minerals [17,19].
Conclusion
A greater variability was noticed in the case of the phosphorus, calcium and magnesium elements, in the two studied stands. During all three growing season moments, the foliar-nitrogen concentration was noticed to be influenced, to various extents, by the same three soil properties: exchange capacity for hydrogen, organic carbon concentration, and carbon-nitrogen ratio. The foliar concentration of nitrogen decreases, with the advancement in the growing season; and the intensity of the connection between this one and the aforementioned properties increases at the end of the growing season. At the A-horizon level, significant correlations are noticed for the calcium element in the soil and in the leaf, in August. In the B horizon, the relations for the calcium macro element are the same as in the A horizon. The copper microelement in the soil correlates with the one in the leaf, both in August and in October. As against the A horizon, in the B horizon, another correlation appears in August, between the phosphorus concentration in the soil and in the leaf.
The potassium deficit in the beech stand could signal a disturbance in the assimilation process with an impact on the tree’s health. The significant trends in foliar N/nutrient ratios were positive for Ca and K and negative for Mg indicating that Ca and K foliar nutrition was deteriorating in comparison to N nutrition while Mg nutrition was decreasing.
For spruce, the negative temporal trend in the foliar N to Mg ratio could be an indication that some forest ecosystems are progressively recovering from past acidification due to the decline in acidifying deposition observed in Europe. However, during the last two years, the spruce stand shows a normal nutrition status.
In the context of Europe’s higher demand for wood biomass used as green energy, it is important to continue monitoring forest health through interdisciplinary approaches that use harmonized and updated methodologies.
This research was funded by the Romanian Ministry of Research, Innovation, and Digitization within the NUCLEU Program – BIOSERV, Project IDs PN19070104, PN19070102, PN 19070101 and CresPerfInst project (contract no. 34PFE/30.12.2021).
- www.roifn.ro. Inventarul forestier Național.
- Sofletea N, Curtu L. Dendrology. Transilvania University Publishing House. Brasov. 2007; 419.
- Badea O, Neagu Ş, Silaghi D, Leca Ş. Development and implementation of the forest monitoring system at the level of the European Union. LIFE 07/ENV/D/000218. Technical report. "Marin Drăcea" National Research and Development Institute. Manuscript. 2011;112.
- Blujdea V, Ionescu M, Surdu A. The variation of essential nutrients throughout the bioaccumulation period and assessments on mineral nutrition of Turkey oak (Quercus cerris L.) and Hungarian oak trees (Quercus frainetto Ten.). Revista Pădurilor. 2001; 116( 1):15-20.
- Aerts R, Chapin FS. The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Advances in Ecological Research. 2000; 30:1-55.
- Ordoñez J, Bodegom PV, Witte JP, Wright I, Reich P, Aerts R. A global study of relationships betweean leaf traits, climate and soil measures of nutrient fertility. Global Ecology and Biogeography. 2009; 18(2):137-149.
- Marschner H. Mineral Nutrition of Higher Plants. Academic Press. London. 1986:674.
- Rautio P, Fürst A, Stefan K, Raitio H, Bartels U. Part XII: Sampling and Analysis of Needles and Leaves. Version 2020-3. In: UNECE ICP Forests Programme Co-ordinating Centre (ed.): Manual on methods and criteria for harmonized sampling, assessment, monitoring and analysis of the effects of air pollution on forests. Thünen Institute of Forest Ecosystems, Eberswalde. Germany. 2020; 16.
- Dincă L, Lucaci D, Iacoban C, Ionescu M. Methods of analysis of soil properties and solution. Forestry Technical Publishing House. Bucharest. 2012; 173.
- Duquesnay A, Dupouey JL, Clement A, Ulrich E, Le Tacon F. Spatial and temporal variability of foliar mineral concentration in beech (Fagus sylvatica) stands in northeastern France. Tree Physiol. 2000 Jan;20(1):13-22. doi: 10.1093/treephys/20.1.13. PMID: 12651522.
- Parascan D, Danciu M. Physiology of Woody Plants. Editura Pentru Viață. Brașov. 2001; 301.
- Larcher W. Physiological Plant Ecology. Springer Verlag Berlin Heidelberg. New York. 1975; 245.
- Kopinga J, v den Burg J. Using soil and foliar analysis to diagnose the nutritional status of urban trees. Journal of Arboriculture. 1995; 21(1):17-24.
- Pop E, Sălăgeanu N, Péterfi Șt, Chirilei H. Plant Physiology. Editura Didactică și Pedagocică. București. 1964; I:353.
- Batos B, Miletic Z, Orlovic S, Miljovic D. Variability of nutritive macroelements in pedunculate oak (Quercus robur L.) leaves in Serbia. Genetika. 2010; 42(2):435-453.
- Enescu RE, Spârchez Gh. Mineral nutrition of Romanian white oak species: a short review. Transilvania University Bulletin. 2014; 7(56):13-20.
- Târziu DR. General and Forest Pedology. Editura Universității Transilvania. Brașov. 2008;272.
- INRA. Biosoil project. File format specification. SOM file. Composite sample analysis. 2007.
- National Institute for Research and Development for Pedology, Agrochemistry and Environmental Protection – ICPA Bucharest, 2003. Romanian Soil Taxonomy System.

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
