Open Journal of Analytical and Bioanalytical Chemistry
The polyphenol composition of Cistus incanus L., Trachystemon orientalis L. and Melissa officinalis L. infusions by HPLC-DAD method
Vanya Dimcheva*, Nikolay Kaloyanov and Maria Karsheva
Cite this as
Dimcheva V, Kaloyanov N, Karsheva M (2019) The polyphenol composition of Cistus incanus L., Trachystemon orientalis L. and Melissa officinalis L. infusions by HPLC-DAD method. Open J Anal Bioanal Chem 3(1): 031-038. DOI: 10.17352/ojabc.000008Because of plants, health benefits consumption of herbal infusions as a dietary additive has increased in the last years. The aim of the present study was to establish the individual phenolic profiles using high-performance liquid chromatography with diode-array detection (HPLC-DAD) method of Bulgarian Cistus incanus leaves harvested in summer and winter seasons, Cistus incanus hard-coated seeds, Greek Cistus incanus leaves harvested in summer, Bulgarian Trachystemon orientalis, and Melissa officinalis leave and stems extracts prepared as infusions. Cistus incanus L. has played a very interesting role in the historical medicine of the Mediterranean region where is well known. Trachytsemon orientalis L. is wild borage typical for Caucasus nature. Both wild plants are widely distributed in Strandja mountain, Bulgaria and the ethnomedicinal description of them as anti-inflammatory and antioxidant herbs are missing. The Melissa officinalis is wildly used as an aromatic, culinary and medicine remedy and its medicinal properties are well known of Bulgarian folk medicine. In the present study, the newly established HPLC-DAD method developed and validated previously for simultaneous separation of 9 natural substances common in plants - tree major catechins, four major flavonoids, gallic and vanillic acid was used. The method was successfully applied to the determination of the specific bioactive substances in the investigated herbs and especially Cistus incanus species. Analyses under the method applied revealed that in the Bulgarian Cistus incanus summer leaves infusion, the values of (-) - epicatechin (3460.54 ± 1.89 μg/mL) and (-) - epigallocatechin (7369.23 ± 2.56 μg/mL) were found to be significantly higher (p > 0.05) than values of the two catechins found in the other infusions. However, the winter Cistus incanus leaves contains 640.03 ± 1.58 μg/mL (-) - epigallocatechin and 489.98 ± 2.89 μg/mL rutin which are significantly higher concentrations (p > 0.05) almost 2 times more than these present in the Greek Cistus incanus leaves infusion 386.69 ± 3.85 μg/mL and 273.07 ± 0.96 μg/mL, respectively. Nevertheless, the Greek Cistus incanus leaves contains the highest concentration of gallic acid (219.04 ± 1.06 μg/mL, p > 0.05) among all analyzed infusions. The highest polyphenol concentration found in Melissa officinalis leaves and stems infusion was (-) - epigallocatechin (188.99 ± 2.08 μg/mL) follow by quercetin, gallic acid, and kaempferol. In Trachystemon orientalis were found only the flavonoids rutin (56.46 ± 0.31 μg/mL) and myricetin (630.23± 1.63 μg/mL). The investigation has led to a better understanding of the polyphenolic profiles of the analyzed plant infusions, especially the non-traditional for the Bulgaria herbs – Cistus incanus and Trachystemon orientalis and has clearly supported the further utilization of the two species in the Bulgarian traditional medicine. Not least the Cistus incanus leaves harvested in the winter season and Cistus incanus hard-coated seeds could be used as an antioxidant infusion with high added value.
Introduction
The research interest towards active compounds, especially polyphenols, of a natural source has greatly increased in recent years [1]. Plants containing natural antioxidants are used as an important source of new drugs formulations and the plant extracts have been used to treat many diseases and their mode of action may well have been based on the phenolic compound content [2,3]. Phenolic compounds are the most widely occurring groups of phytochemicals and are of considerable physiological and morphological importance in plants as well as having strong antioxidant properties [4].
The wild herb Cistus incanus is not traditional in Bulgarian ethnomedicine but is widespread in all Strandja Mountain [5]. The Bulgarian Cistus incanus is a rich source of polyphenols and antioxidants [6]. For the „Pamukliyka“ (Strandja local name) it is reported that the high values of polyphenols, flavonoids, and the high antioxidant capacity occurred not only in the leaves but were also found in the Cistus incanus hard-coated seeds [5]. The history of the Holy rose in the world began an ancient time [6]. The wild herb is wild spread in all Mediterranean. Some of Cistus species are well-known around the world and they are studied in detail. Cistus incanus infusions and products containing extracts are notable examples for such polyphenol-rich food supplements because many research studies have demonstrated that the main biologically active components of the wild herb refer to polyphenolic compounds such as gallic acid, rutin, quercetin, kaempferol, glycoside compounds belonging to the flavonol family, flavan-3-ols as well as catechin, epicatechin [7], gallocatechin and gallocatechin-3-gallate [8]. Traditional Mediterranean folk medicine has used Cistus species for anti-inflammatory, antiulcerogenic, wound healing, antimicrobial, cytotoxic and vasodilator remedies [9]. The antioxidant properties of the Greek Cistus incanus are already studied and few Greece companies offer worldwide „Hairy Rockrose” tea, essential oils, antiseptic sprays, tablets and capsules. The detail polyphenol content of the sub-endemic species Cistus incanus grown in our longitude is not studied yet. There is no literature about polyphenolic identification of the Cistus incanus hard-coated seeds and Cistus incanus leaves collected in the winter harvest season. Such, not-standardized mixtures represent the natural polymorphism of the plant and offer no basis for the manufacturing of a standardized plant extract. Kaempferol, quercetin, and apigenin proved to be the most basic parts of the structures. Research on an extract of the pink rock rose CYSTUS® 052, manufactured from Cistus incanus PANDALIS, shows 26 % of polyphenols, less than 2 % of monomers such as gallic acid, epigallocatechin, catechin, and epicatechin [10].
Trachystemon orientalis L. is commonly known as „Abraham-Isaac-Jacob“ or „Oriental Borage“ in Turkey. The medicinal plant is all edible and all parts of it are consumed as a vegetable in Istanbul and different parts of the black sea [11,12]. Phytochemical constituents are phenolic compounds, tannins, essential oils, mucilages, saponins, nitrates and resins among others. Trachystemon orientalis recorded folkloric usages are as a diuretic, blood purifier, emollient, antipyretic, etc. [13]. In Bulgaria, the „Eastern Lopuh“ (Strandja local name) is not popular and is used as food only from population lives where it is distributed – Eastern Central Balkans and all of Strandja.
Melissa officinalis L., commonly known as lemon balm or balm mint, is a well-known medicinal plant of Lamiaceae family. For more than 2000 years, fragrant leaves of this plant have been widely used in cooking to add flavour to dishes [14]. Melissa officinalis L. is a medicinal plant that has long been used in different ethnomedical systems especially in the European Traditional Medicine and the Iranian Traditional Medicine for the treatment of several diseases [15]. Phytochemical investigations revealed that this plant contains volatile compounds, triterpenoids, phenolic acids and flavonoids [16]. Crude extracts and pure compounds isolated from Melissa officinalis exhibited numerous pharmacological effects, from which only anxiolytic, antiviral and antispasmodic activities of this plant as well as its effects on mood, cognition, and memory have been shown in clinical trials [17]. In traditional medicine, lemon balm is commonly administered as a tea infusion that is used to treat maladies such as gastrointestinal complaints, headache, and fever [15]. The phenolic content of Melissa officinalis varies in different regions and is significantly affected by cultivar [15,18]. Nevertheless, there are no reports about the phenolic profile of garden cultivated in North-eastern Bulgaria Melissa officinalis.
Therefore, identification and quantification of phenols from different sources is becoming increasingly important due to their potential application for treating diseases. A very common method used for the qualitative and quantitative analyses of polyphenols is HPLC coupled with DAD. The HPLC is a powerful technique for rapid analysis of bioactive constituents because it enables systematic profiling of the complex plant samples and specifically focuses on their identification and consistent evaluation of the identified compounds. UV/Vis spectra have long been used for structural analysis of polyphenols and flavonoids [19].
The aim of the present study was identification and quantification of the phenolic compounds of water extracts (infusions) of Cistus incanus leaves, Cistus incanus hard-coated seeds, Trachystemon orientalis, and Melissa officinalis leaves and stems using previously developed and validated HPLC-DAD method.
It is known that the activities of crude extracts obtained from medicinal plants may be subjected to change based on the variations of chemical composition. Also, the activities difference may be due to the geographical origin, locality, climate conditions, and the harvest time of the collected plant material [20,21]. Therefore, it is interesting to discover the polyphenolic profiles of the plants collected in different seasons and plants with different geographical origin.
The purpose of this investigation also was to be compared the polyphenol profiles of Cistus incanus leaves collected in summer and collected in winter harvest seasons and Cistus incanus leaves collected from Bulgaria and Greece. Although, is an interesting aspect to be investigated the infusion’ polyphenol constituents of undiscovered endemic species grown in Bulgaria – Cistus incanus and Trachystemon orientalis. Nevertheless, is interesting to be carried out unstudied before garden cultivated lemon balm (Melissa officinalis) native from North-eastern Bulgaria.
On the whole and because of the health-related potential properties of the herbal infusions, the aim of this work was to provide new knowledge on the content of the antioxidant substances of the selected herbs.
Materials and Methods
Materials
Plant materials: The leaves and hard-coated seeds from the Bulgarian Cistus incanus were collected in Strandja mountain, near the Black Sea region (the area ,,Parnara”, near village Varvara), in the south-eastern of Bulgaria. The samples of two different seasons of harvest were examined: picked up in the summer (August, 2015), and picked up in the winter (March, 2016). The temperatures measured on the days of collection of the plant were 29 °C in August, and 7 °C in March. The leaves of Greek Cistus incanus were collected in Khalkidhiki, Orange beach (Greece). The temperature measured on the day of collection was 28 °C (May). The leaves of cultivated lemon balm (Melissa officinalis) were picked up from a local garden in North-Eastern Bulgaria (Village Sofronievo) in August. The temperature measured on the day of collection was 32 °C. The Trachystemon orientalis was collected in Strandja mountain, near the Black Sea region (the area „Parnara“, near village Varvara), in the south-eastern of Bulgaria. The temperatures measured on the days of collection of the plant were 29 °C in August. The wild plants were identified by experienced biologists from the National Park Strandja. The cultivated lemon balm was identified according to Flora Ibérica [22]. To ensure representative sampling, 1 kg of the plants were collected and 0.30 kg from the hard-coated seeds. The samples were kept in a dry place for further experiments.
Chemicals: Ethanol (96 %) was supplied by Valerus, (Sofia, Bulgaria). Trichloroacetic acid (TCA), (99 %); acetonitrile, HPLC grade; gallic acid anhydride (> 99 %); vanillic acid (> 99 %); rutin hydrate (> 98 %); quercetin hydrate (≥ 95 %) was supplied by Merck, (Sofia, Bulgaria). (-) - Epicatechin gallate (100 %); (-) - catechin (100 %); (-) - epigallocatechin (100 %); myricetin (> 98 %) and kaempferol (> 98 %) were supplied by Alfa Azar, (Sofia, Bulgaria). Deionized water (18.6 MΩ cm resistivity) generated by ELGA water purification system (Sofia, Bulgaria).
HPLC Analysis
The analyses were performed with an Agilent 1100 HPLC system (Agilent 1100 HPLC, Agilent Technologies, California, USA) equipped with DAD detector (G1315B, Agilent Technologies, California, USA), and managed by HP Chemstation. The column used was Purospher star, Hiber RT 125-4; RP18 (Purospher star, Merck, Bulgaria). The column temperature was 25 °C. Separation was carried out using a linear gradient using 0.1 % TCA (A) and 100 % acetonitrile (B). The gradient started with 5 % B, 15 % B at 16.5 min, 33 % B at 22.5 min, 100 % B at 30.5 min, 5 % B at 35 min until 40th for re-equilibration. The flow rate was 1.6 mL/min. The injection volume for the samples and standards was 30 μL. The DAD acquisition was set in the range of 200–400 nm. Identification of the main compounds was performed by comparing the retention times and UV specters of the peaks obtained in the both - sample and standard chromatograms.
Infusions preparation
All samples were kept in a dry place for two years before being ground in the grinder and sieved. The leaves and seeds were used contained water, not more than 10 %. For the experiments, a fraction of 0.5 –1.0 mm particle size was used. The extractions were prepared with the aid of the infusion technique. The solid-to-solvent ratio was fixed at 1:30 (1 g leaves/seeds in 30 mL solvent) for all samples. The infusions were carried out with deionized water for 15 minutes time. The infusions were left to stay at room temperature without additional heading. After each extraction, the exhausted plant material was carefully pressed, and the extract was filtered through cotton, filter paper, and after that was used 0.45 μm Nylon-membrane syringe filter (Acrodisc, Sigma-Aldrich, Bulgaria). The filtered samples were introduced in the 2 mL vials and stored in the fridge before the examination.
Results
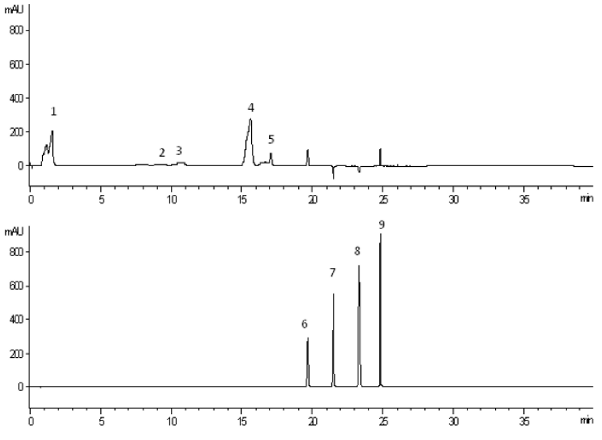
For the analyses in the present study was used developed and validated previously a simple and reliable HPLC-DAD method for simultaneous separation of 9 natural substances common in plants - tree major catechins, four major flavonoids, gallic and vanillic acid. The used references standards were chosen because they are reported to be widely represented in the plant kingdom and have strong antioxidant capacity. The reported method was carried out only for 40 minutes with consequently changing of two wavelengths (278 and 368 nm). The HPLC chromatogram of the used reference substances is given in Figure 1.
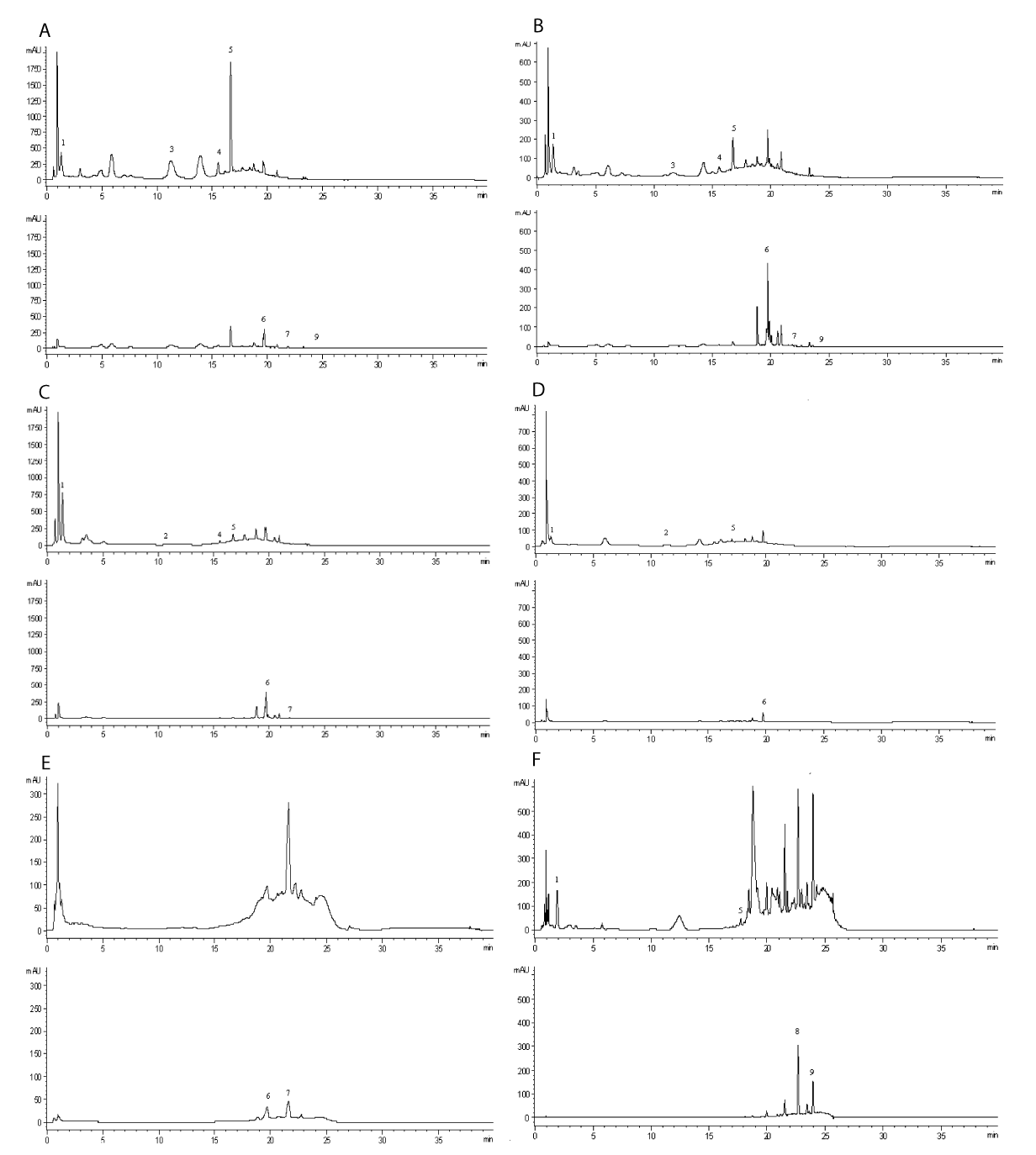
The reported HPLC-DAD method was proved to be appropriate for the identification and quantification of individual polyphenols contain in the selected plants which can be seen from the chromatograms of the analyzed infusions given in Figure 2.
The identification of each peak was possible via retention time as well as spectra of samples and authentic standards. The concentrations in μg/mL of each identified compound from the obtained herbal infusions are presented in Table 1. The values in the table are expressed as means ± SD (n=2). Different letters in the same column indicated significant differences at p < 0.05 according to the analyses of variance (ANOVA), Tukey’s Honest Significant Difference test (Tukey’s HSD test).
The results shown in table 1 revealed that the Cistus incanus summer leaves infusion is richest in a number of found polyphenols. The found constituents were gallic acid, (-) - epicatechin, vanillic acid, (-) - epigallocatechin, rutin, myricetin, and kaempferol as follow concentrations 110.28 ± 1.27, 3460.54 ± 1.89, 47.13 ± 0.92, 7369.23 ± 2.56, 34.22 ± 1.65, 7.79 ± 0.84, and 0.48 ± 0.17 μg/mL. In the Bulgarian Cistus incanus, winter leaves infusion the polyphenol composition was similar but instead the compound (-) - epicatechin has identified the compound (-) - catechin. Also the concentrations of some bioactives were lower than those found in the summer leaves such as concentration of gallic acid (54.38 ± 1.69), vanillic acid (10.07 ± 0.36 μg/mL), (-) - epigallocatechin (640.03 ± 1.58 μg/mL), and myricetin (3.91 ± 0.63 μg/mL). However, other polyphenols like rutin (489.98 ± 2.89 μg/mL) and kaempferol (0.49 ± 0.19 μg/mL) had higher values. Unlike the Bulgarian Cistus incanus leaves in the Greek Cistus incanus summer leaves was found only five polyphenol compounds as gallic acid, vanillic acid, (-) - epigallocatechin, rutin and myricetin in concentrations as fallow 219.04 ± 1.06, 9.88 ± 0.59, 386.69 ± 3.85, 273.07 ± 0.96, 4.32 ± 0.11 μg/mL. In the hard-coated seeds from the Bulgarian Cistus incanus was found only the gallic acid (18.89 ± 0.86 μg/mL), (-) - epicatechin (23.55 ± 0.13 μg/mL), (-) - epigallocatechin (102.75 ± 1.26 μg/mL), and rutin (26.74 ± 0.33 μg/mL). Quercetin, rutin, (-) - epigallocatechin and gallic acid were identified in the Bulgarian Melissa officinalis leaves and stems with quantities of 56.20 ± 0.99, 188.99 ± 2.08, 58.16 ± 2.66, and 0.67 ± 0.19 μg/mL, respectively. At least a number of polyphenolic compounds were detected in the Trachystemon orientalis leaves and stems. In the „Oriental Borage“ was identified only rutin and myricetin with values of 56.46 ± 0.31 μg/mL and 30.23 ± 1.63 μg/mL, respectively.
Discussion
Recent interest in phenolic compounds, flavonoids, in particular, has developed robustly thanks to their antioxidant capacity and their possible beneficial implications in human health. In the search for new anti-inflammatory compounds, special interest has been directed to herbal-based products reputed in traditional medicine to have beneficial effects on inflammatory disorders. Herbal infusions contribute to the major source of phenolic compounds in our diet. They are commonly consumed for their perceived healthcare benefits [23]. Many herbal teas are used medicinally by Indigenous and local peoples for improved nutrition, and the prevention and treatment of health problems ranging from colds and flu to digestive disorders, malaria and cardiovascular disease [24].
It is well recognized that phenolic compounds extraction depends on multiple factors, such as their chemical nature, raw material, storage time and geographical conditions. Furthermore, It depends on the quantification methods chosen, the standards applied, interferences presence [25]. Thus, it is necessary to adjust the identification and quantification method to achieve the optimal estimation of the phenolic compounds. The identification of phenolic acids and flavonoids present in the extracts was of major interest for determining their properties, knowing the fact that the type of phenolics may determine the antioxidant or antibacterial properties of a matrix, in detriment of the quantity [26]. Increasing knowledge of the positive health effects of food polyphenols has prompted the need to develop new separation techniques for their extraction, fractionation, and analysis.
The developed and validated previously method was successfully applied to the determination of the specific bioactive substances in the investigated herbs and especially Cistus incanus species. This becomes clear from the presented results where 7 polyphenols were identified in Bulgarian summer and winter Cistus incanus leaves infusions found to be richest in terms of the number identified phenolic compounds. In the Bulgarian Cistus incanus summer leaves’ infusion, the (-) - epicatechin and (-) – epigallocatechin values (3460.54 ± 1.89 μg/mL and 7369.23 ± 2.56 μg/mL) were significantly higher (p > 0.05) than those among the other herbal infusions where the two catechins also were presented. Catechins, especially epicatechin and epigallocatechin are the major polyphenols in green tea, many dietary products, plants, and fruits [27]. This bioactives have strong antioxidant activity and are the most wildly consumed polyphenols in the world. Likewise, (-) - epicatechin and (-) - epigallocatechin were determined in the Bulgarian Cistus incanus hard-coated seeds but in lower concentrations (23.55 ± 0.13 μg/mL and 102.75 ± 1.26 μg/mL). The presence of these catechins, gallic acid and rutin make possible the usage of these areal parts together with the Cistus incanus leaves as an infusion with high added value as a dietary additive. The infusion of the Bulgarian winter Cistus incanus leaves contains 640.03 ± 1.58 μg/mL (-) - epigallocatechin which is significantly higher concentration (p > 0.05), almost 2 times more than those present in the Greek Cistus incanus leaves infusion (386.69 ± 1.26 μg/mL). Likewise, the (-) - epigallocatechin was quantified in all Cistus incanus infusions and also in the Melissa officinalis leaves and stems infusion in the significant concentration of 188.99 ± 2.08 μg/mL. However, (-) - catechin was identified only in the Bulgarian Cistus incanus winter leaves with the value of 177.65 ± 2.35 μg/mL together with gallic acid, vanillic acid, (-) - epigallocatechin, rutin, myricetin, and kaempferol. The concentration of rutin was the richest in the winter leaves infusion with the value of 489.98 ± 2.89 μg/mL, p > 0.05. The Cistus incanus winter leaves polyphenol profile wasn’t studied before and they are not used as an infusion for human consumption according to the literature. Data are missing, perhaps, because it is well known that plants produce polyphenols in higher concentrations only in summer. The present study on the Cistus incanus leaves harvested in winter season explore their potential as an antioxidant infusion with high-added value.
In the Greek Cistus incanus summer leaves (-) - epicatechin and kaempferol weren’t found, unlike Bulgarian Cistus incanus summer leaves. The differences in the content of the same plants growing at different latitudes could be attributed to environmental and harvesting conditions which can lead to a loss of bioactive compounds in plants.
Plants can also contain a lot of phenolic acids such as gallic and vanillic acids. Gallic acid is a well-known natural antioxidant that is basically a secondary polyphenolic metabolite. Gallic acid is a very important common antioxidant tea formulation [28]. The gallic acid presented in the highest amount in the Greek wild herb - 219.04 ± 1.06 μg/mL, (p > 0.05) among all herb infusions where the compound also consists. Gallic acid wasn’t quantified only in Trachystemon orientalis leaves and stems infusion and was determined with the lowest amounts in the Bulgarian Cistus incanus hard-coated seeds infusion (18.89 ± 0.86 μg/mL). Vanillic acid was quantified only in the Cistus incanus leaves extracts with the highest concentration found in the Bulgarian Cistus incanus summer leaves (47.13 ± 0.92 μg/mL, p > 0.05).
Identified flavonoids in the plant extracts were rutin, myricetin, quercetin, and kaempferol. Rutin was found in all herbal infusions without Melissa officinalis infusion in significant quantities between 26.74 ± 0.33 μg/mL, (p > 0.05) in the Bulgarian Cistus incanus hard-coated seeds and 489.98 ± 2.89 μg/mL, (p > 0.05) in the Bulgarian Cistus incanus winter leaves. Rutin is a common dietary flavonoid glycoside that is consumed in fruits, vegetables, and plant-derived beverages [29].
Myricetin was found in the infusions prepared from the Cistus incanus leaves in the range of 4.32 ± 0.11 to 7.79 ± 0.84 μg/mL and in the Trachystemon orientalis leaves and stems where was found with a higher concentration of 30.23 ± 1.63 μg/mL, p > 0.05. Myricetin is a naturally occurring flavonoid in tea, berries, fruits, vegetables, and medicinal herbs [29].
Quercetin was quantified only in the one extract - mint balm infusion with the amount of 58.16 ± 2.66 μg/mL. Quercetin is one of the important bioflavonoids present in more than twenty plants material and which is known for its anti-inflammatory, antihypertensive, vasodilator effects, antiobesity, antihypercholesterolemic and antiatherosclerotic activities [30].
In the Cistus incanus, summer and winter leave infusions were found similar concentrations of kaempferol 0.48 ± 0.17 and 0.47 ± 0.19 μg/mL, respectively. In the Melissa officinalis leaves and stems’ infusion, the presence of kaempferol was with the highest concentration - 0.67 ± 0.19 μg/mL. Kaempferol is a naturally occurring flavonol that has been isolated from edible plants such as the tea as well as in plants or botanical products commonly used in traditional medicine [29].
Cistus incanus is called a medicine herbal plant in the Mediterranean due to its antimicrobial, anti-inflammatory, cytotoxic and antiulcerogenic properties [5,6,31] Considering these unique properties, quantification of the bioactive compounds in the infusions of the Cistus incanus grown in Bulgaria is very important because of the rising knowledge about consumption and use of this beverage in the country.
The higher number of identified phenolics being found in Mediterranean Cistus incanus infusions but obtained with different extraction methods and used plant material from different geographical locations. Because of this, it would be difficult to compare the results of different studies. Rutin, myricetin, quercetin, kaempferol, catechin, gallocatechin, epicatechin were found by LS-MS in Cistus incanus herbal tea bought from a Polish market [31]. In Cistus, incanus growing in Italy was found gallic acid, catechin, and rutin received from an infusion of powdered plant leaves and analyzed with HPLC-MS [32]. By HPLC-MS analyses of Greek Cistus incanus infusions were identified phenolic acids, flavan-3-of monomers, and dimers as well as flavonol glycosides [9].
Roots and aerial parts of Cistaceae have been used since ancient times in the Mediterranean cultures for its medicinal properties. No previous HPLC reports have been found in the composition of the hard-coated seeds of the Cistus incanus. Also, the polyphenol profile of the Bulgarian Cistus incanus leaves collected in the summer and in the winter harvesting time is unknown till now. The present investigation shows that Bulgarian Cistus incanus leaves infusions could be prepared not only in the summer but also in the winter season. According to the current investigation, the hard-coated seeds of the Strandja „rockrose“ collected at the end of the summer are also a good alternative for the preparation of high polyphenol herbal infusion.
Trachystemon orientalis is borage which commonly used as a vegetable and is preparing as food. The wild herb usually is not used as a beverage and there is no data about its usage as tea infusion for the treatment of deashes. However, our study shows that the „Eastern Lopoh“ native from Strandja mountain, Bulgaria could be used as tea infusion because it is a good source of flavonoids such as rutin and myricetin which are high antioxidants.
The European lemon balm is studied in detail. Phytochemical investigations on Melissa officinalis have revealed the presence of various phytochemicals including phenolic acids, flavonoids, and tannins [15]. Different compositional variations of different Melissa officinalis extracts are reported in the literature. For example, gallic acid, caffeic acid, rosmarinic acid, epicatechin, catechin, and rutin were found in Melissa officinalis ethanolic extracts [33]. These compositional variations in the Melissa officinalis could stem from differences in climatic, seasonal and geographic conditions, harvesting time, and procedural details of the applied extraction technique [34]. The lemon balm infusion native from North-Eastern Bulgaria has never been studied before. The presence of the found polyphenols named gallic acid, epigallocatechin, quercetin, and kaempferol in significant quantities give the full state that the infusion of the garden cultivated herb could be consumed as an infusion with high-added value.
No universal method can be used with all the phenolic compounds: different approaches must be used depending on the different plant extract and polyphenols of interest. The used HPLC-DAD method is more proper for identification and quantification of polyphenols in Cistus incanus infusions than other infusions because was gave a comprehensive polyphenol profile. The reported method is not universal in polyphenol characterization but gives primary information about the composition of the obtained infusions. The method could be evaluated in the future for better representing of the antioxidant profiles or examination of the polyphenols of interests in the examined herbal infusions.
Statistical analyses
Each sample was extracted and analyzed in duplicate and the results are expressed as the mean ± standard deviation (SD). The one-way ANOVA and Tukey’s HSD post hoc tests were performed to identify the differences among the bioactive substance’ concentrations in the different plants. Differences were considered to be significant at p < 0.05. Statistica 9.0 software (Stat-Soft, Tulsa, OK, USA) was used for the statistical calculations.
Conclusion
The role of herbal medicines in improving human health is gaining popularity over the years. Increased consumption of herbal infusions containing phenolic compounds give positive and better results in human health and significantly improves the immune system. Highly selective, susceptible and versatile analytical techniques are necessary for extraction, identification, and quantification of phenolic compounds from plant extracts, which helps to utilize their important biological properties.
The established simple and reliable HPLC-DAD method in this investigation was successfully applied for the simultaneous identification and quantitation of the polyphenol compounds mainly in the Cistus incanus leaves and seeds infusions. The antioxidant potential of the infusions prepared from Strandja’ Cistus incanus summer and winter leaves and hard-coated seeds, Strandja’ Trachystemon orientalis leaves and stems and North-West’ Melissa officinalis leaves and stems were successfully explored.
This work allows the use of the selected infusions with high added value antioxidants to be used such as dietary additives.
The remaining undefined high-added constituents of the investigated herbs infusions must be evaluated in the future for better confirmation of their bioactive properties.
Our research highlights the need the herbal teas to be studied through from an ethnopharmacological perspective. It is required greater consideration of traditional knowledge in the pharmacological research. Product safety and sustainability issues should also be considered, so the traditional applications of herbal teas can be transformed into efficient health-boosting functional products.
This work was partially supported through Grant No 11582 provided by the Research Division of UCTM – Sofia.
- HY Aboul-Enein MA (1984) Loutfy, Pyridoxine hydrochloride. / In book Alanalytical profiles of drug substance. Elsevier Inc 13: 447-478.
- Aliev AM, Onov AO (1972) The complexing properties application of azo derivateves of B6 vitamins for its determination by an electrophoresis on a paper. Laboratornoe delo (in Russian). 8: 470-473.
- Monferrer-Pons L, Alvarez-Rodríguez L, Esteve-Romero J, García-Alvarez-Coque MC (1999) Spectrophotometric determination of B6 vitamins by coupling with diazotized p-sulfanic acid in micellar medium of N-cetylpyridinium chloride. Anal Lett 32: 51-63. Link: https://bit.ly/2RB2LrU
- Monferrer-Pons L, Alvarez-Rodríguez L, Esteve-Romero J, Garcia-Alvarez-Coque MC (2000) Flow-injection analysis of pyridoxine hydrochloride by coupling with the diazonium ion of p-sulfanilic acid. Anal Lett 33: 539–552. Link: https://bit.ly/2ISs31Z
- Rocha F, Filho OF, Reis BF (2003) A multicommuted flow system for sequential spectrophotometric determination of hydrosoluble vitamins in pharmaceutical preparations. Talanta 59: 191-200. Link: https://bit.ly/2IPHYOy
- Portela JG, Costa ACS, Teixeira LSG (2004) Determination of vitamin B6 in pharmaceutical formulation by flow-injection-solid phase spectrophotometry. J Pharm Biomed Anal34: 543-549. Link: https://bit.ly/31Zpkvs
- Kadir ANA (2010) Spectrophotometric determination of vitamin B6 by coupling with diazotized p-nitroaniline. Jour Raf Sci 21: 49-59. Link: https://bit.ly/2xdlctq
- Varygina OV, Chernova RK, Сusina EF (2013) ISE on vitamin B6. Proceedings Saratov University. Chemistry, Biology, Ecology 13: 3-7.
- Hochberg M, Melnick D, Oser BL (1944) On the stability of pyridoxine. J Biol Chem155: 109-119.
- Calatayud JM (2005) Spectrophotometry. Pharmaceutical applications / In book: Encyclopedia of analytical science. 2nd ed. Editors: P. Worsfold, A. Townsend C Poole Elsevier 380-383.
- Hansen SH, Pedersen-Bjergaard S, Rasmussen KE (2011) Introduction to pharmaceutical chemical analysis. Publ Online. Link: https://bit.ly/2X7nLI7
- Watson DG (2012) Pharmaceutical analysis, 3 ed. London. 441. Link: https://bit.ly/31TIe6V
- Ahuja S, Skipinski S (2012) Handbook modern pharmaceutical analysis. V.3. Press 566. Link: https://bit.ly/2FDRJ0e
- Calatayud JM, Zamora LL (2013) Spectrophotometry: pharmaceutical applications. / In book: Reference module in chemistry, molecular sciences and chemical engineering 9-13. Link:
- European Pharmacopoeia. 9th Edition. 2017. Link: https://bit.ly/2KSHXYx.
- Ostrovskaya VM, Zaporozhec OA, Budnikov GK, Chernavskaya NM (2002) Water: indicators systems, (in Russian), Moscow: VINITI 266.
- Zolotov YuA, Ivanov VM, Amelin VG (2002) Chemical test methods of analysis. Series: Comprehensive analytical chemistry, V. 36, Elsevier Science 336. Link: https://bit.ly/2RCwCQV
- Sadomtseva OS, Shakirova VV, Urnova VV, Fadeeva MV (2017) Pyridoxin qualitative and quantitative determination method. RU Patent 2632629 (2017). Bull. 28, 2017.
- Ostrovskaya VM, Fomin NA, Aksenova MS, Kazakova TS, Popova TD, et al. (1989) producing epoxidized chromatographic paper. SU Patent 1651204 (1991). Bull. 19, 1991. A priority 15.05.1989.
- Ostrovskaya VM, Shpigun LK, Shushenachev YaV (2018) Indicator strip of RIB-diazo-test for indicator means to determine authenticity of medicine substance. RU Patent 2680391 (2019). Bull. 5, 2019. A priority 25.04.2018.
- Ostrovskaya VM (2012) The dimensional cutting device to obtain the acid-base reagent and diagnostic indicator strips. RU Patent 117853 U1 (2012). Bull. 19, 2012. A priority 04.04.2012.

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Save to Mendeley
Save to Mendeley
