Journal of Vaccines and Immunology
Intranasal vaccination with a Chimeric Chlamydial Antigen BD584 confers protection against Chlamydia trachomatisgenital tract infection
Steven Liang1 and James B Mahony1,2*
2Department of Pathology and Molecular Medicine, Faculty of Health Sciences, McMaster University, Hamilton, Ontario, Canada
Cite this as
Liang S, Mahony JB (2020) Intranasal vaccination with a Chimeric Chlamydial Antigen BD584 confers protection against Chlamydia trachomatisgenital tract infection. J Vaccines Immunol 6(1): 010-017. DOI: 10.17352/jvi.000030Chlamydia is the most common sexually transmitted infection of bacterial origin, and a broadly protective vaccine is urgently needed given the largely asymptomatic nature of the infection and the severe reproductive sequelae in women with untreated infections. The primary aim of this study was to characterize the immune response to vaccination with BD584, a novel recombinant antigen consisting of three type III secretion (T3S) proteins, and to evaluate its efficacy against a Chlamydia trachomatischallenge in three strains of mice. C57BL/6, BALB/c, and C3H/HeN mice were vaccinated three times intranasally with BD584 and CpG adjuvant, then challenged intravaginally with C. trachomatis. BD584/CpG vaccination induced strong cell-mediated and humoral immune responses in all three strains of mice, suggesting that this vaccine should be immunogenic in a genetically heterogenous population. BD584/CpG vaccination reduced vaginal shedding of C. trachomatis in C57BL/6 and C3H/HeN mice. Together, these results strengthen the rationale for further investigating the use of T3S proteins in a C. trachomatis vaccine.
Introduction
Chlamydia trachomatisinfections are the most prevalent sexually transmitted bacterial infections in the world. The World Health Organization estimates that 131 million new cases of C. trachomatis infection occur each year [1]. Up to 90% of women and 50% of men with these infections are asymptomatic [2] and consequently do not seek testing and treatment. Untreated infections in women can lead to many complications including Pelvic Inflammatory Disease (PID), tubal factor infertility, and ectopic pregnancy [3]. Public health programs, including screening, partner identification, and treatment, have contributed to improve infection control, but to curb the infection rates, there is a need for the development of an effective C. trachomatis vaccine [4] as a necessary next step to control this public health problem.
Almost immediately following the isolation of the causative agent of trachoma, human trials were initiated using vaccines based on whole Chlamydiae[5]. While these studies demonstrated that some protection can be achieved using whole-cell vaccines, protective efficacy appeared to be serovar-specific, which, together with concerns about potentially detrimental antigens in intact bacteria, highlighted the need for a broadly protective subunit vaccine. Although five decades of vaccine research efforts have led to the discovery of a large number of potential vaccine antigens [6,7], only one such vaccine has reached phase I clinical trials [8]. Therefore, sustained efforts are needed to identify and evaluate promising antigens for testing in humans.
The type III secretion (T3S) system is an essential virulence factor used by many Gram-negative bacterial pathogens to manipulate host cell biology to ensure successful invasion. Surface exposed T3S proteins are therefore attractive vaccine candidates as they are essential for infection and amenable to neutralization by antibodies. While they have been successfully used in the development of vaccines against other pathogenic bacteria [9-11], only within the last few years have they been tested as vaccine antigens against Chlamydia. Recently, Koroleva et al. reported that immunization with the T3S needle protein (TC_0037), an ortholog of C. trachomatis CdsF, protected mice against C. muridarum infection and the associated pathology [12]. A multistage vaccine containing proteins associated with the T3S system (CdsF and CT584), inclusion membrane (IncA and CT229), and outer membrane (MOMP and PmpG) conferred greater protection against infection and pathology than vaccines targeting outer membrane proteins alone [13]. Previously, our group has shown that C57BL/6 mice immunized with BD584, a fusion protein consisting of highly conserved C. trachomatis T3S components CopB, CopD, and CT584, were protected against infection and upper genital tract (UGT) pathology upon challenge with the mouse pathogen C. muridarum [14]. However, given that significant differences exist between C. muridarum and C. trachomatis infections [15,16], vaccine-mediated protection against C. muridarum does not imply protection against C. trachomatis, and it is important to determine the effectiveness of BD584 vaccination against the human pathogen. Furthermore, a more detailed analysis of the cell-mediated and humoral immune response to BD584 vaccination will enhance our understanding of BD584-mediated protective immunity.
Most Chlamydia vaccine studies utilizing the murine model have been conducted using a single inbred mouse strain, which is not ideal given the genetic heterogeneity of humans, suggesting the need for vaccine studies using animals with different genetic backgrounds. Moreover, use of animals with genetically determined differences in immune predispositions can also provide insight into host factors that contribute to effective anti-Chlamydial immunity. In this study, we used C57BL/6 mice (Th1-polarized; Chlamydia-resistant), C3H/HeN mice (Th1-polarized; Chlamydia-susceptible), and BALB/c mice (Th2-polarized; Chlamydia-susceptible) to model human genetic heterogeneity and to identify potential correlates of BD584-mediated protection [17,18]. We show here that intranasal immunization with BD584 elicits robust cell-mediated and humoral immune responses in addition to strong mucosal immunity in all three strains of mice and that BD584 immunization confers protection against C. trachomatis infection in a strain dependent way.
Materials and methods
Ethics statement
This study was carried out in accordance with the Canadian Council on Animal Care guidelines and approved by the Animal Research Ethics Board at McMaster University. Every effort was made to minimize animal suffering. Isoflurane and ketamine/xylazine were used during vaccination and challenge procedures, respectively. The method of euthanasia was cervical dislocation.
BLASTp
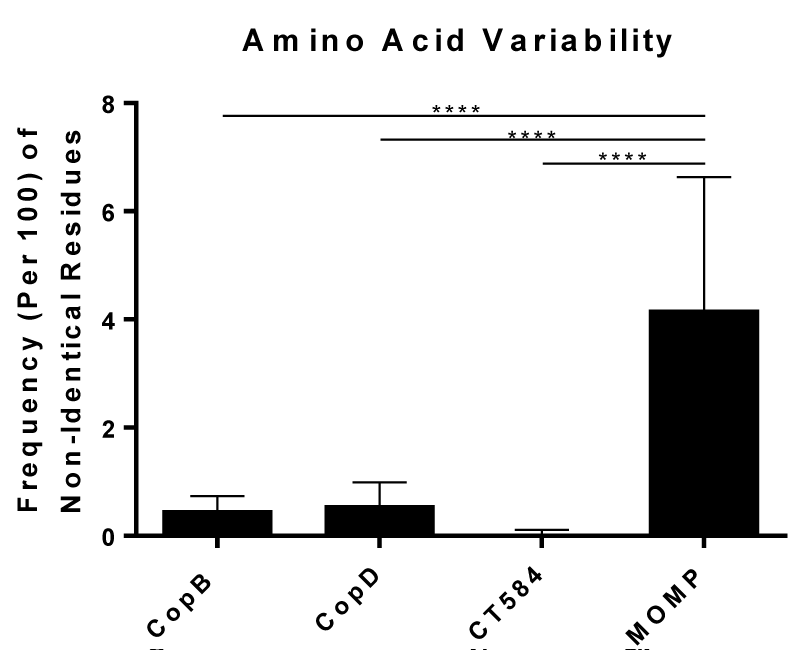
Non-redundant protein sequences were searched with BLASTp using CopB, CopD, CT584, or MOMP from C. trachomatis serovar D (UW-3/Cx) as query sequences. Using the queries as references, the number of non-identical residues in each identified C. trachomatis sequence was calculated, tabulated, and represented as a frequency of non-identical amino acids.
Chlamydia trachomatis
A clinical isolate of C. trachomatis serovar D was propagated in McCoy cells (ATCC) in RPMI 1640 containing 25 mM HEPES, 10% FBS, 2 mM L-glutamine, 50 µg/ml gentamicin, and 1 µg/ml cycloheximide (Infection Media). After 48 hours of infection, C. trachomatis was harvested and stored in multiple aliquots in 250 mM sucrose, 10 mM sodium phosphate, 0.5 mM L-glutamic acid (SPG, pH 7.4) at -70°C.
Cloning
C. trachomatis (UW-3/Cx) copB1-300 and copD1-300 were cloned into pDEST17 using the Gateway cloning system according to manufacturer’s instructions (Thermo Fisher Scientific, ON, Canada). Briefly, attB-containing gBLOCK DNA fragments (Integrated DNA Technologies, ON, Canada) coding for first 100 amino acids of CopB or CopD were cloned, via BP reaction, into pDONR201 donor vectors, which were then used in LR reactions to generate expression vectors pDEST17::copB1-300 and pDEST17::copD1-300. Cloning of CT584 in pET-21b and has been described previously [19].
Cloning of BD584, which consists of the first 100 amino acids of CT578 (CopB) and CT579 (CopD) and full length CT584, has been described previously [14]. Briefly, PCR products containing the first 300 nucleotides of copB, copD, and full length ct584 were generated from genomic DNA from C. trachomatis (UW-3/Cx) using primers with appropriate restriction sites and cloned into the multiple cloning site 1 of pETDuet-1 to generate pETDuet-1::BD584. All constructs included a His-tag for protein purification via affinity chromatography.
Protein expression and purification
To generate the antigens required in this study, pDEST17::copB1-300, pDEST17::copD1-300, pET-21b::CT584, and pETDuet-1::BD584 were transformed separately into Escherichia coli BL21 (DE3) cells for protein expression and purification as previously described [14]. Briefly, bacteria were grown to an OD600nm of 0.6 at 37°C in LB broth and induced for 2-3 hours with 0.2 mM Isopropyl β-D-1-thiogalactopyranoside (IPTG). Cells were harvested by centrifugation and lysed via sonication. The lysate was further centrifuged and the insoluble fraction was discarded. Fast Protein Liquid Chromatography (FPLC) using Ni-NTA HisTrap HP column (GE Healthcare, ON, Canada) was used to purify recombinant protein (at > 95% purity), which was buffer exchanged into PBS using a desalting column, filter sterilized, aliquoted, and stored at -80°C.
Vaccination and challenge
C57BL/6, BALB/c, and C3H/HeN female mice were purchased from Charles River Laboratory. Six to eight week old mice were immunized three times at intervals of three weeks. Animals were anaesthetized with 5% isoflurane and vaccinated intranasally with either 20 µg of BD584 plus 20 µg of CpG oligodeoxynucleotide 1826 (with a phosphorothioate backbone) (ACGT Corp, ON, Canada), in 20 µl of PBS (BD584/CpG) or with 20 µl of vehicle alone (PBS). Mice were pretreated with 2.5 mg of Depo-Provera (Pfizer, ON, Canada) in 100 µl of saline administered subcutaneously 10 and 3 days prior to challenge. Three weeks after the final immunization, mice were challenged intravaginally with 105 inclusion forming units (IFUs) of C. trachomatis serovar D suspended in 10 µl of SPG.
Monitoring of infection
To assess bacterial shedding following challenge, vaginal samples were collected on days 3, 7, 10, 14, 21, and 28 post challenge using FLOQSwabs (REF No. 516CS01, Copan, Italy). Swabs were placed into 0.5 ml of SPG, vortexed with 1 mm Mini-BeadBeater Glass Mill Beads (BioSpec, OK, USA) for 30 seconds, and stored at -70°C until ready for analysis. Serial dilutions of swab samples were plated onto McCoy cells in 96 well tissue culture plates and centrifuged at 1,500 x g for 1 hour at 25°C. The inoculum was then replaced with fresh Infection Media and incubated for 24 hours at 37°C / 5% CO2. Cells were fixed with Fixation Buffer (BD Biosciences, ON, Canada) for 20 min at room temperature, blocked with Perm/Wash Buffer (BD Biosciences, ON, Canada) overnight at 4°C, and stained with rabbit anti-Chlamydia antibody (Genetexa, CA, USA) followed with Alexa Fluor 647-conjugated mouse anti-rabbit antibody (Jackson ImmunoResearch Laboratories, PA, USA) at 37°C for 1 hour each. C. trachomatis inclusions were identified manually under an EVOS FL Cell Imaging System microscope (Thermo Fisher Scientific, ON, Canada). The total number of IFUs per swab was calculated based on the number of IFUs per well, accounting for dilution factors, inoculation volumes, and sample volumes.
Collection of vaginal washes and blood
Seven days after the final immunization, vaginal washes were collected for antibody analysis for five consecutive days by pipetting twice with 30 µl of PBS in and out of the vagina to give a total of 60 µl per mouse per day and stored at -70°C. Blood (submandibular) was collected 14 days after the final immunization, allowed to clot at room temperature for 30 minutes, centrifuged at 1,000 x g for 10 minutes at 4°C, and serum was removed and stored at -70°C.
Antibody ELISA
Antigen-specific antibodies were quantified by Enzyme-linked immunosorbent assay (ELISA). NUNC MaxiSorp 96-well plates (Thermo Fisher Scientific, ON, Canada) were incubated with 10 µg/ml of CopB, CopD, CT584, or BD584 in PBS for one hour. Plates were then blocked with 50 mg/ml of skim-milk in PBS + 0.1% Tween 20 (PBST) for one hour. Serially diluted serum or vaginal wash samples were added to the wells and incubated for two hours, followed by the addition of horseradish peroxidase-conjugated goat antibodies specific to mouse IgA, IgG1, IgG2a, IgG2b, IgG2c, IgG3 (Abcam, ON, Canada), or whole IgG (Sigma-Aldrich, ON, Canada) for one hour. The substrate, 3,3’,5,5’-tetramethylbenzidine (BD Biosciences, ON, Canada) was added to each well and incubated for 10 – 20 minutes before stopping the reaction with 1 M H2SO4. Absorbance at 450 nm (A450nm) was measured on a ELx800 microplate spectrophotometer (BioTek, VT, USA). All steps were performed at room temperature and plates were thoroughly washed with PBST between incubations. The endpoint titre of a sample was defined as the dilution at which the A450nm of the sample was equal to the mean A450nm of the negative samples plus three times the Standard Deviation (SD).
Splenocyte recall assay
To determine cell-mediated immune responses, another cohort of mice was vaccinated with BD584/CpG or PBS and sacrificed seven days later. Mouse spleens were mechanically disrupted through 40 µm cell strainers (Corning), re-suspended in RPMI 1640 containing 25 mM HEPES, 10% FBS, 2 mM L-glutamine, 50 µM β-mercaptoethanol, 100 IU/ml penicillin, and 100 µg/ml streptomycin (Splenocyte Media), and seeded into U-bottom 96-well plates at 5 x 105 live cells per well. Splenocytes were then supplemented with either Splenocyte Media alone or Splenocyte Media containing 10 µg of BD584 and incubated for 96 hours at 37°C / 5% CO2. Cell culture supernatants were assayed for IFN-γ, IL-17, IL-4, TNF-α, or IL-10 using DuoSet ELISA kits (R&D Systems, MN, USA) according to manufacturer’s instructions.
Statistical analysis
GraphPad Prism 6 was used for statistical analysis. Differences were assessed using Student’s t test for parametric data and Mann-Whitney test for nonparametric data, one-way ANOVA when comparing more than two groups, or two-way ANOVA when comparing more than one independent variable. Statistical significance is indicated in figures as *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
Results
CopB, CopD, and CT584 are highly conserved among C. trachomatis serovars
A C. trachomatis vaccine should be effective against all C. trachomatis serovars, and one way to achieve this would be to include antigens that are highly conserved. Therefore, we began our investigation by comparing the degree of amino acid variability in CopB, CopD, and CT584, using MOMP as a reference antigen. As shown in Figure 1, alignment of known sequences of each protein showed that CopB, CopD, and CT584 have, on average, 0.46, 0.55, and 0.02 non-identical amino acids per 100 residues, respectively, which are significantly fewer compared to MOMP, which averages 4.16 non-identical amino acids per 100 residues. These results show that CopB, CopD, and CT584 are significantly more conserved than MOMP in C. trachomatis, which is an advantage for subunit vaccine for a pathogen with multiple serotypes.
BD584/CpG vaccination induces robust cell-mediated immune responses
Cell-mediated immunity has been reported to be essential for protection against Chlamydia [15,20]. Therefore, we examined whether vaccination with BD584 and CpG induces antigen-specific cell-mediated immune responses. C57BL/6, BALB/c, and C3H/HeN mice were intranasally vaccinated with BD584/CpG or PBS, and BD584-induced production of cytokines from splenoctyes was analyzed 7 days later. Antigen-specific splenocyte production of IFN-γ and IL-17 in all three strains of mice indicates that a mixed Th1 and Th17 response was induced by intranasal vaccination with BD584/CpG, independent of the genetic background of the animal (Figure 2). No antigen-specific cytokines were produced by splenocytes from mice vaccinated with PBS. As expected with a Th1-polarizing adjuvant, BD584/CpG vaccination did not elicit IL-4 production. Interestingly, the level of BD584-induced production of IFN-γ was strain dependent, with C3H/HeN mice developing significantly higher IFN-γ responses compared to C57BL/6 and BALB/c mice. However, IFN-γ production induced by the three separate subcomponents of BD584 (CopB, CopD, and CT584) relative to that induced by BD584 were not significantly different between strains (Supplementary Figure 1A). BD584/CpG vaccinated C3H/HeN mice exhibited a moderately but significantly higher level of antigen-specific TNF-α and IL-10 production compared to C57BL/6 and BALB/c mice (Figure 2). These results suggest that intranasal BD584/CpG vaccination induces a mixed Th1/Th17 antigen-specific cell-mediated immune response with variable immunogenicity dependent on the animal’s genetic background.
BD584/CpG vaccination elicits robust systemic and mucosal humoral immune responses
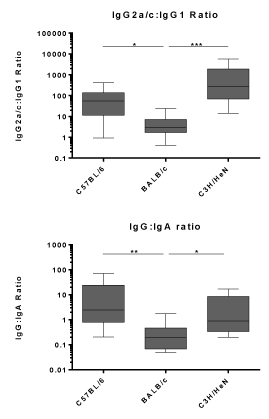
While cell-mediated immunity is essential for protection against C. trachomatis, accumulating evidence suggests that a C. trachomatis vaccine should also generate strong humoral immunity. Therefore, C57BL/6, BALB/c, and C3H/HeN mice were intranasally vaccinated three times, three weeks apart, and antibody responses were assessed two weeks after the final vaccination. Intranasal vaccination with BD584/CpG elicited robust BD584-specific IgG and IgA responses in both serum and vaginal secretions in all three strains of mice (Figure 3A). PBS-vaccinated mice did not generate antigen-specific responses. Interestingly, BD584-specific antibody titres were consistently and significantly lower in C57BL/6 mice compared to BALB/c and C3H/HeN mice. However, CT584-specific serum IgG, when normalized against the level of BD584-specific serum IgG in each mouse, was significantly higher in C57BL/6 mice (Supplementary Figure 1B). This suggests that the immunogenicity of the CT584 subcomponent relative to that of BD584 is greater in this strain. BD584-specific IgG1, IgG2a/c, IgG2b, and IgG3 titres were lower in C57BL/6 mice compared to BALB/c and C3H/HeN mice (Figure 3B). Compared to BALB/c mice, C57BL/6 and C3H/HeN mice exhibited higher IgG2a/c to IgG1 ratios (Figure 4), suggesting a more polarized Th1 response in these strains. Furthermore, C57BL/6 and C3H/HeN mice also had higher BD584-specific vaginal IgG to IgA ratios (Figure 4). Taken together, these results show that BD584/CpG is highly immunogenic in all three strains of mice, and that the subtype of antibodies associated with antigen-specific Th1 and Th2 responses is at least partially dependent on the animal’s genetic background.
BD584/CpG immunization reduces bacterial shedding upon intravaginal C. trachomatis challenge
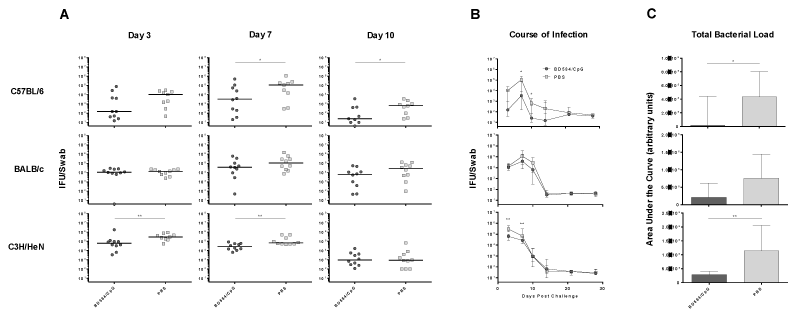
Next, we evaluated the ability of BD584/CpG immunization to protect against C. trachomatis infection as measured by reduction in bacterial shedding following intravaginal challenge. As shown in Figures 5A and 5B, C57BL/6 mice immunized with BD584/CpG showed a nearly 100-fold reduction of C. trachomatis shedding on days 7 and 10 post challenge compared to the PBS group. Shedding in the unvaccinated group had ceased on day 10 and no difference was seen in bacterial shedding between the two groups after day 14. Reduction in shedding was seen as early as day 3 post challenge, although the difference at this time point was not statistically significant. Compared to C57BL/6 mice, C3H/HeN mice immunized with BD584/CpG exhibited less reduction in shedding. BD584/CpG immunized C3H/HeN mice shed 4.7- and 1.9-fold less C. trachomatis on day 3 and day 7, respectively, compared to the PBS group. An area under the curve (AUC) analysis, which allows for a simultaneous comparison of both the magnitude and duration of infection, showed that BD584/CpG immunization reduced the overall bacterial load by 96% in C57BL/6 mice and 75% in C3H/HeN mice (Figure 5C). Interestingly, no statistically significant reduction in shedding was observed at any point for BALB/c mice. Overall, these results demonstrate that BD584/CpG vaccination reduces C. trachomatis shedding upon challenge in C57BL/6 and C3H/HeN mice.
Discussion
In this study, we show that vaccination with the conserved T3S fusion antigen, BD584, elicits strong cell-mediated and humoral immune responses in animals with different genetic backgrounds and, upon an intravaginal C. trachomatis challenge, reduces bacterial shedding in two out of three strains of mice.
Previously, we have shown that intranasal immunization with C. trachomatis BD584/CpG reduces shedding and incidence of hydrosalpinx upon intravaginal challenge with C. muridarum [14]. Given that BD584 was cloned from the C. trachomatis genome, its ability to afford cross-species protection speaks to its conserved nature and its potential as a broadly protective immunogen for all 18 C. trachomatis serovars. The amino acid identity of BD584 between CT and Cmu is 57% while the identify between the ocular, genital and LGV strains of C. trachomatis is 92%. We have shown recently that antibodies raised against BD584 neutralizes ocular (serovars A and C), genital (serovars D and E) and LGV (LGV1) strains of C. trachomatis (unpublished data). The T3S components that make up the fusion antigen, CopB, CopD, and CT584, have significantly less antigenic diversity among C. trachomatis strains compared to MOMP (Figure 1) , which reflects the conservation of these proteins that make up an essential virulence factor required for cell invasion. Therefore, a vaccine consisting of BD584 should provide protection against all serovars of CT and may be less susceptible to vaccine escape.
To evaluate the ability of BD584 vaccination to confer protection against infection, we immunized three strains of mice with BD584 plus CpG before challenging them intravaginally with C. trachomatis. Compared to control mice, BD584/CpG vaccinated C57BL/6 and C3H/HeN mice had reduced shedding at the peak of infection but similar clearance kinetics. Thus, while BD584/CpG vaccination appears to have no effect on the duration of infection, it can reduce the magnitude of infection, which has the potential to reduce the incidence of disease by limiting transmission of infections. Ideally, however, a vaccine would confer sterilizing immunity, but this is difficult to evaluate in the C. trachomatis mouse model since reliable infection requires the use of a high inoculum that could overwhelm an otherwise sterilizing immune response [16]. In a previous study, we showed that BD584 vaccination reduced shedding of C. muridarum assessed by quantitative PCR, which does not necessarily imply a reduction in the shedding of live organisms [14]. In the current study, we show that BD584 vaccination reduced shedding of infectious C. trachomatis, which was quantified by culture, thus providing stronger evidence for vaccine efficacy.
BD584 vaccination induced robust cell-mediated and humoral immune responses in all three strains of mice, suggesting that the antigen should be immunogenic in a genetically heterogenous population. Splenocytes from BD584/CpG immunized mice exhibited increased IFN-γ and IL-17 production with no detectable IL-4 production following in vitro BD584 stimulation, suggesting that a mixed Th1 and Th17 response was generated by vaccination. This was expected given that BD584 was delivered intranasally, a route that preferentially generates Th17 responses [21], with a Th1 polarizing adjuvant, CpG [22]. Vaccinated C57BL/6 mice had significantly lower levels of BD584-specific IgG and IgA compared to vaccinated BALB/c and C3H/HeN mice, but exhibited the highest level of protection against shedding, suggesting that an increase in the magnitude of BD584-specific humoral responses, at least above a certain level, may not necessarily improve protection against infection. Instead, genetically determined differences in innate and adaptive immunity likely accounted for the difference in vaccine efficacy between the three strains. Increased susceptibility to Chlamydia infection in C3H/HeN mice, compared to C57BL/6 mice, may have masked the protection afforded by BD584/CpG vaccination [18]. However, it is unclear why BALB/c mice were not significantly protected by vaccination. Compared to vaccinated BALB/c mice, vaccinated C57BL/6 and C3H/HeN mice had a significantly higher BD584-specific IgG2a/c to IgG1 ratio, suggesting a more Th1 polarized response. However, whether higher IgG2a/c directly enhances protection via Fc-dependent mechanisms [23,24], or is merely a surrogate marker for a protective Th1 response [25], or is associated with an unknown mechanism of protection, remains unknown. A high IgG2a/Ig1 ratio may also be functionally similar to a high IgG/IgA ratio; in both cases, the higher ratio may reflect a greater engagement with Fc-dependent effector functions. We did not evaluate pathology in this study. In a previous study using C. muridarum we showed that UGT pathology viz. hydrosalpinx was reduced by 85% after vaccination with BD584. Importantly, an immune-correlates analysis of an HIV vaccine clinical trial found that higher antigen-specific IgG and lower antigen-specific IgA correlated with protection [26]. The authors suggested that IgA interfered with protective IgG effector functions. Interestingly, in the current study, we also found that the two strains of mice protected by BD584 vaccination exhibited higher BD584-specific IgG/IgA ratios than the unprotected strain. However, conclusive evidence for the role of specific antibody isotypes and subtypes will need to come from mechanistic studies, which are beyond the scope of the current study. Experiments are underway to better understand the mechanisms of BD584-mediated protection. Given that different strains of Chlamydia infect different animals (mice and humans) and different immune mechanisms may be required to clear Chlamydial infections in different animals, the protection provided by BD584 should be evaluated additional animal models. Although cell-mediated responses obtained in this study appeared to be good, and this is important in clearing an intracellular pathogen infection, the duration of immune protection could not be evaluated in this study, because C. trachomatis infection doesn’t persist in non-vaccinated mice. The use of progesterone treated animals may help elucidate the question of duration.
Conclusion
we show that immunization with the BD584 fusion antigen consisting of three highly conserved T3S proteins is immunogenic in mice with different genetic backgrounds, elicits robust cell-mediated and humoral immune responses, and confers protection against C. trachomatis infection in two out of three strains of mice tested, strengthening the rationale for exploring the use of T3S proteins in a human C. trachomatis vaccine. Evaluation of the protection provided by BD84 in additional animal models may help elucidate the full potential of this vaccine for humans.
Supplementary materials
Supplementary Figure 1 shows CopB-, Cop-D, and CT584-specific IFN-γ and serum IgG responses following intranasal vaccination of C57BL/6, BALB/c, and C3H/HeN mice with BD584.
Conceptualization, James Mahony; Methodology, Steven Liang; Project administration, James Mahony; Writing – original draft, Steven Liang; Writing -review and editing, James Mahony. Steven Liang is the recipient of a Vanier Scholarship. We are grateful to Dr. Michael Barta for providing us with plasmids for protein expression, and Dr. Tiffany Leighton for critical reading of the manuscript.
- World Health Organization (2015) Progress report of the implementation of the global strategy for prevention and control of sexually transmitted infections: 2006-2015. Link: https://bit.ly/2C7L2VH
- Peipert JF (2003) Clinical practice. Genital chlamydial infections. New England journal of medicine 349: 2424-2430. Link: https://bit.ly/32cSU2Q
- Brunham RC, Gottlieb SL, Paavonen J (2015) Pelvic inflammatory disease. The New England journal of medicine 372: 2039-2048. Link: https://mayocl.in/3fp5W16
- Brunham RC, Rappuoli R (2013) Chlamydia trachomatis control requires a vaccine. Vaccine 31: 1892-1897. Link: https://bit.ly/38RnAbt
- Bell SD, Nichols RL, Haddad NA (1963) The Immunology of the Trachoma Agent with a Preliminary Report on Field Trials on Vaccine. Investigative ophthalmology 2: 471-481. Link: https://bit.ly/3j0aixJ
- Finco O, Frigimelica E, Buricchi F, Petracca R, Galli G, et al. (2011) Approach to discover T- and B-cell antigens of intracellular pathogens applied to the design of Chlamydia trachomatis vaccines. Proc Natl Acad Sci U S A 108: 9969-9974. Link: https://bit.ly/2WeJa4r
- Karunakaran KP, Rey-Ladino J, Stoynov N, Berg K, Shen C, et al. (2008) Immunoproteomic discovery of novel T cell antigens from the obligate intracellular pathogen Chlamydia. J Immunol 180: 2459-2465. Link: https://bit.ly/32aeNQi
- Olsen AW, Follmann F, Erneholm K, Rosenkrands I, Andersen P (2015) Protection Against Chlamydia trachomatis Infection and Upper Genital Tract Pathological Changes by Vaccine-Promoted Neutralizing Antibodies Directed to the VD4 of the Major Outer Membrane Protein. J Infect Dis 212: 978-989. Link: https://bit.ly/2AX2N9Q
- Zauberman A, Cohen S, Levy Y, Halperin G, Lazar S, et al. (2008) Neutralization of Yersinia pestis-mediated macrophage cytotoxicity by anti-LcrV antibodies and its correlation with protective immunity in a mouse model of bubonic plague. Vaccine 26: 1616-1625. Link: https://bit.ly/3iZ1jx1
- Holder IA, Neely AN, Frank DW (2001) PcrV immunization enhances survival of burned Pseudomonas aeruginosa-infected mice. Infect Immun 69: 5908-5910. Link: https://bit.ly/2OlKUVi
- Martinez-Becerra FJ, Kissmann JM, Diaz-McNair J, Choudhari SP, Quick AM, et al. (2012) Broadly protective Shigella vaccine based on type III secretion apparatus proteins. Infect Immun 80: 1222-1231. Link: https://bit.ly/2Oj8vWv
- Koroleva EA, Kobets NV, Shcherbinin DN, Zigangirova NA, Shmarov MM, et al. (2017) Chlamydial Type III Secretion System Needle Protein Induces Protective Immunity against Chlamydia muridarum Intravaginal Infection. BioMed research international 2017: 3865802. Link: https://bit.ly/2OgKOOA
- ">O'Meara CP, Armitage CW, Andrew DW, Kollipara A, Lycke NY, et al. (2017) Multistage vaccines containing outer membrane, type III secretion system and inclusion membrane proteins protects against a Chlamydia genital tract infection and pathology. Vaccine 35: 3883-3888. Link: https://bit.ly/38PaQC5
- Bulir DC, Liang S, Lee A, Chong S, Simms E, et al. (2016) Immunization with chlamydial type III secretion antigens reduces vaginal shedding and prevents fallopian tube pathology following live C. muridarum challenge. Vaccine 34: 3979-3985. Link: https://bit.ly/2OwJ7wN
- Brunham RC, Rey-Ladino J (2005) Immunology of Chlamydia infection: implications for a Chlamydia trachomatis vaccine. Nature reviews Immunology 5: 149-161. Link: https://go.nature.com/3fA6q4k
- De Clercq E, Kalmar I, Vanrompay D (2013) Animal models for studying female genital tract infection with Chlamydia trachomatis. Infect Immun 81: 3060-3067. Link: https://bit.ly/3ehJFRu
- Lyons JM, Morre SA, Airo-Brown LP, Pena AS, Ito JI (2005) Comparison of multiple genital tract infections with Chlamydia trachomatis in different strains of female mice. J Microbiol Immunol Infect 38: 383-393. Link: https://bit.ly/3gW5KH8
- Darville T, Andrews CW, Laffoon KK, Shymasani W, et al. (1997) Mouse strain-dependent variation in the course and outcome of chlamydial genital tract infection is associated with differences in host response. Infect Immun 65: 3065-3073. Link: https://bit.ly/3en3ZRv
- Barta ML, Hickey J, Kemege KE, Lovell S, Battaile KP, et al. (2013) Structure of CT584 from Chlamydia trachomatis refined to 3.05 A resolution. Acta crystallographica. Section F, Structural biology and crystallization communications 69: 1196-1201. Link: https://bit.ly/3fqCuYt
- Farris CM, Morrison RP (2011) Vaccination against Chlamydia genital infection utilizing the murine C. muridarum model. Infect Immun 79: 986-996. Link: https://bit.ly/328vzPX
- Zygmunt BM, Rharbaoui F, Groebe L, Guzman CA (2009) Intranasal immunization promotes th17 immune responses. Journal of immunology 183: 6933-6938. Link: https://bit.ly/3iWkuYe
- Chu RS, Targoni OS, Krieg AM, Lehmann PV, Harding CV (1997) CpG oligodeoxynucleotides act as adjuvants that switch on T helper 1 (Th1) immunity. J Exp Med 186: 1623-1631. Link: https://bit.ly/2C3904o
- Moore T, Ananaba GA, Bolier J, Bowers S, Belay T, et al. (2002) Fc receptor regulation of protective immunity against Chlamydia trachomatis. Immunology 105: 213-221. Link: https://bit.ly/3fjqTuh
- Moore T, Ekworomadu CO, Eko FO, MacMillan L, Ramey K, et al. (2003) Fc receptor-mediated antibody regulation of T cell immunity against intracellular pathogens. J Infect Dis 188: 617-624. Link: https://bit.ly/2WdeIYq
- Snapper CM, Paul WE (1987) Interferon-gamma and B cell stimulatory factor-1 reciprocally regulate Ig isotype production. Science 236: 944-947. Link: https://bit.ly/3gTCCQF
- Haynes BF, Gilbert PB, McElrath MJ, Zolla-Pazner S, Tomaras GD, et al. (2012) Immune-correlates analysis of an HIV-1 vaccine efficacy trial. The New England journal of medicine 366: 1275-1286. Link: https://bit.ly/30aAu0i
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
