Journal of Neurology, Neurological Science and Disorders
Therapeutic Hypothermia in improving Neurological outcome after Cardiac Arrest
Darine Ashraf Soudy*
Cite this as
Soudy DA (2018) Therapeutic Hypothermia in improving Neurological outcome after Cardiac Arrest. J Neurol Neurol Sci Disord 4(1): 001-007. DOI: 10.17352/jnnsd.0000023Background: Only a few patients survive cardiac arrest as it with widespread cerebral ischemia frequently leads to severe neurologic impairment.
Aim: Our objective was to evaluate whether induced hypothermia improves neurologic recovery in survivors of primary cardiac arrest or not.
Methods: We included randomized or quasi-randomized, recent controlled trials (from 2002 till now based on the electronic databases of PubMed/Medline, Scopus, and Cochrane were searched for relevant literature and studies of adults who were successfully resuscitated, where therapeutic hypothermia (TH) was applied within 6 hours after arrival at emergency department after sudden cardiac arrest especially when asphyxia is the cause of the arrest and where neurologic outcome was compared with no history of previous cardiac arrest. We excluded studies without a control group and historical researches.
Result: In patients who have been successfully resuscitated after cardiac arrest due to ventricular fibrillation, therapeutic mild hypothermia increased the rate of a favorable neurologic outcome and reduced mortality.
Conclusion: Mild resuscitative hypothermia in patients is feasible and safe. It improves short term neurologic recovery and survival in patients resuscitated from cardiac arrest of presumed cardiac origin. Its long-term effectiveness and feasibility at an organizational level need further research.
Introduction
The heart has electrical system control rhythm of heart beat. And presence of problem or disease cause defect rhythm of heart beat. Arrhythmia has three shapes: -Increase in rhythm of heart beat - Decrease in rhythm of heart beat- stopping of heart beat, Sudden Cardiac Arrest (SCA). SCA is different from cardiac attack which is circulatory problem.
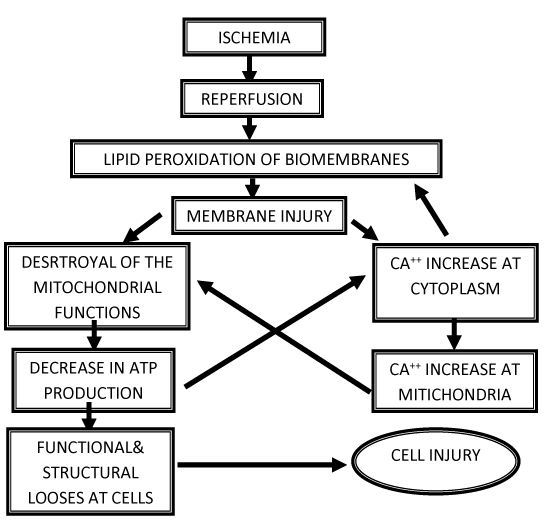
Although return of spontaneous circulation (ROSC) is crucial in the process of recovery from cardiac arrest, it is only the first of many complex stages. Given the complexity of post cardiac arrest syndrome and its impact on the patient. There is cellular changes associated with reperfusion injuries for example the mechanism of free radicals just like hydrogen peroxide or superoxide molecules in the blood causes a lot of damage, inflammation may be part of this damage process as it is a visible sign of the body reaction. And other mechanism that has been proposed for causing reperfusion injury is within the cells themselves through the mitochondria (the power house of the cells) it turns out that they play a vital role in cell life and death.
When you deny the blood flow for the cells or organs so it is ischemia (lack of blood flow), which sets up a number of damaging pathways, these free radicals also called reactive oxygen species (ROS) are created that activate the inflammation cascade to start occurring and enable mitochondria to be dysfunctional or the energy dysfunction.
When these tissues or cells are perfused (return of blood flow) all of the previous complications amplify within minutes after reperfusion and they get more intense and it is a remarkable thing, and if no serious control is done this cascade can become irreversible. Theses mechanisms cause blood vessel instability and blood pressure (BP) comes very unstable, cell death and brain swelling which is well known as cerebral edema that causes severe brain injury that can lasts [1].
Initial neurologic injury occurs when circulatory collapse impairs oxygen flow to the brain. Without oxygen, the brain switches to anaerobic metabolism, which disrupts adenosine triphosphate– dependent cellular pumps, resulting in excessive calcium and glutamate excretion. This excess makes brain cells more excitable, leading to further hypoxemia, in turn causing mitochondrial and cellular death. Cellular death results in cerebral edema, producing further damage. The initial injury also disrupts the blood-brain barrier, which increases fluid in the brain and worsens cerebral edema.
Once circulation is restored, reperfusion injury occurs and may persist for hours. During this time, cell death triggers an inflammatory response in which the immune system releases neutrophils and macrophages to eliminate the dead cells. Unfortunately, this process produces free radicals that cause continued cell damage, worsening the inflammatory response, which exacerbates cerebral edema. This vicious cycle continues to cause neurologic injury.
Discussion
When the Cardiac Arrest (CA) happens, the normal guidelines protocols as (CPR) are done, unfortunately not all the arrested patients survive, and only a few patients return back to their normal circulation. But even patients who return of systemic circulation not all of them are hospital discharged [2] (Figure 1).
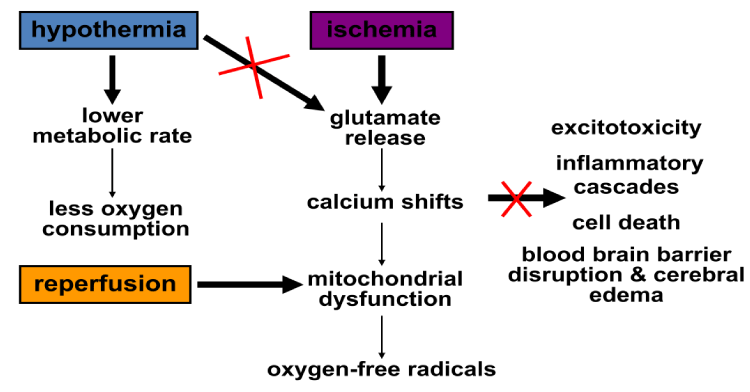
Hypothermia counteracts neuro excitation in brain cells by stabilizing calcium and glutamate release, reducing the degree of cell death. Cerebral metabolism decreases 6% to 10% for every degree Celsius that body temperature drops. As cerebral metabolism declines, the brain needs less oxygen.
In essence, hypothermia counteracts many of the destructive mechanisms of cardiac arrest. Its effects resemble those of cardiac defibrillation, which makes the heart stop and then reset itself to a normal rhythm. Similarly, hypothermia halts destructive brain mechanisms and lets the brain reset itself to normal functioning.
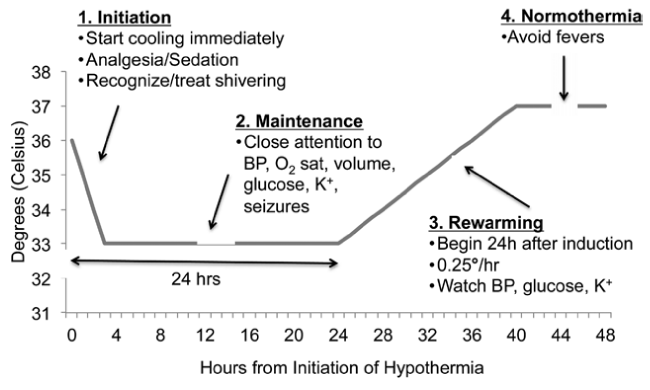
Therapeutic hypothermia occurs in three phases—induction, maintenance, and rewarming. Clinicians must control hypothermia and rewarming to prevent potential adverse effects.
Temperature should be monitored with a method that measures core temperature, such as use of an esophageal, bladder, or pulmonary artery catheter.
Duration of therapeutic hypothermia depends on facility protocol. No research is available on optimal duration. Some facilities start counting duration from the time cooling begins; others start when the patient reaches target temperature.
Nearly all adverse effects can be prevented or managed in an intensive care setting.
Pilot trial and recent studies show the importance of hypothermia for recovery after cardiac arrest .it shows that it was safe and feasible to mildly cool patients after CA.
Surface cooling was initiated within 62 minutes after CA .The target temperature (32-340C) was reached after 287 minutes and was maintained for an additional 24 hour. Thereafter, patients were passively rewarmed and reached temperature>35 after 7 hours (Figure 2).
The outcome was measured with the CPC score which subdivided into good outcome (CPC1 and CPC2), poor outcome (CPC 3 and CPC 4) or death [4].
Dr. Bernard at Dandenong hospital pointed to that cerebral perfusion injury occurs when cerebral blood flow is restored after cardiac arrest and resuscitation .increased intracellular levels of glutamate, an excitatory neurotransmitter released from pre-synaptic terminals., activate ion channel complex that cause calcium to shift from the extracellular to the intracellular fluid leading to the accumulation of oxygen free radicals and the activation of degradative enzymes .In addition, cerebral hemodynamics may remain abnormal after resuscitation from cardiac arrest [5] .
Randomized, controlled studies of pharmacologic interventions to improve patient’s neurologic outcome after CA have tested but have found no benefit .the use of hypothermia has been studied in laboratory animals. The studies demonstrated significantly improved outcome when moderate hypothermia was induced after resuscitation. Although hypothermia reduce pulse rate and increased systemic vascular resistance, there were no clinically significant cardiac arrhythmias in hypothermia group.
In 2007s, Acta Anathesiol Scand discussed that their experience indicates that patients who are haemo dynamically unstable after CA requiring support with IABP and adrenergic drugs can be safely treated with hypothermia. Their strategy was early revascularization with PCI and a liberal use of IABP to support failing circulation. It is not known whether hypothermia favors the heart as well as the brain with respect to the prevention of reperfusion injury. They conclude that TH will probably be a part of CPR in the future.
The rapid successful implementation of therapeutic hypothermia indicates that simple external cooling protocol was well accepted by everyone. Although not 100% successful in achieving the target temperature, the ease of application and absence of costs related to new equipment should make method an attractive way of starting up TH in any system .the present recommendation is to cool the patients as rapidly and as soon as possible [6].
Cooling protocol and feasibility
The rapid successful implementation indicates that our simple external cooling protocol was well accepted by everyone involved; paramedics, critical care nurses and physicians. Bernard et al. described his method as inconvenient for the nursing staff and a barrier to implementation TH in clinical practice [7], Although not 100% successful in achieving the target temperatures, the ease of application and absence of costs related to new equipment should make our method an attractive way of starting up TH in any system. All studies using external cooling methods have reported difficulties with cooling all patients, and similar slow cooling rates have been a universal finding in the studies so far [3,5,7,8], Nonetheless, the present recommendation is to cool the patients as rapidly and as soon as possible [6,7,9,10] .
In 2016 ANZCOR puts guidelines provides advice on (TTM) during (PCA) period which is a therapy associated with improved out comes.
Guideline applies to adult arrested who require (ALS) after CA. it suggests TTM as opposed to no TTM for adults with in hospital cardiac arrest (IHCA) with any initial rhythm who remain unresponsive after ROSC) ,it suggest prevention and treatment of fever in persistently comatose adults after completion of TTM between 32 and 36 [11].
Hypothermia also show great effect in pulmonary complications after SCA, cardiogenic shock that was not responsive to usual therapy ,no risk for bleeding in hypothermic patients compared with normo thermic controls, does not significantly increase metabolic acidosis or lactate levels for cardiac or neurologic surgery [12].
Hypothermia is contraindicated for pregnancy, Hypothermia before cooling efforts begin, bleeding problem ICV filter excluded from long cooling catheters (ICY) must use shorter cooling and existing DNR or DNI order.
Some complications following hypothermia like bradycardia which can be managed by use of agents to stimulate heart rate, but if it became clinically significant, active cooling should be discontinued, patient actively re-warmed. Infection also can be a complication of hypothermia. Treatment should be early initiating for signs of infection.
According to published studies, hypothermia leads to an increased risk of myocardial infarction due to resulted coronary vasoconstriction. Myocardial ischemia in hypothermic patients depends on the previous status of coronary artery in patient, so that in normal individuals, hypothermia has been shown to improve myocardial blood flow; however, in patients with a history of coronary artery disease, it causes vasoconstriction in atherosclerotic arteries.
Patient’s treated with hypothermia are exposed to the risk of hyperglycemia; and increased blood sugar levels are associated with increased morbidity and mortality. Tight control of blood sugar levels and insulin therapy has been associated with decreased morbidity and mortality. Increased blood sugar levels are associated with increase in the rate of infection, neuropathy and renal failure. Therefore, in hypothermic patients, tight control of blood sugar levels is essential [7].
MIH could be associated with shivering which increases oxygen consumption. At the time of induction of hypothermia, body uses several mechanisms to obtain and maintain the heat. At this status, the body prevents lose of temperature through increasing sympathetic tone and vasoconstriction of skin and uses shivering to generate heat. Shivering increases oxygen consumption about 40 to 100% and has adverse effects on patients with neurological injuries and damages caused by hypoxia. This is less of a problem in patients undergoing mechanical ventilation, because shivering does not lead to increased respiratory effort in these people [13].
Other complications
Hypothermia increases serum levels of amylase and liver enzymes. In addition, most patients undergoing hypothermia induction would require intubation, mechanical ventilation, sedation and muscle relaxation. These would limit evaluation and monitoring of neurological function of the patients. Fortunately, although hypothermia has adverse effects, but most of the complication of induction of hypothermia can be prevented or controlled in intensive care conditions.
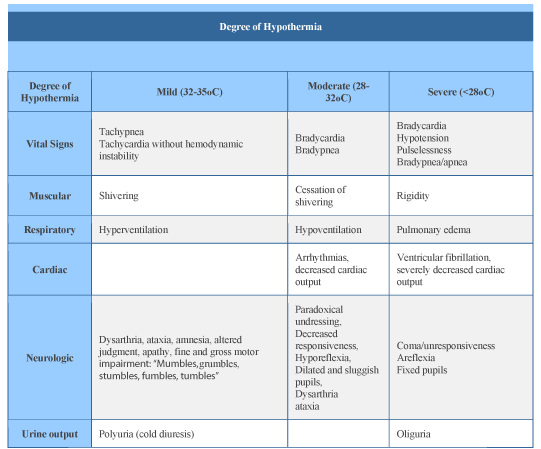
We finally conclude the great effect of hypothermia, but care should be taken during performing it to avoid its complications, continuous monitoring temperature should be done to obtain effective result and safe patient life [12] (Figure 3).
Use of therapeutic hypothermia in children
In 2003 there was insufficient evidence to make a recommendation on the use of TH in children resuscitated from cardiac arrest [14].
Until additional pediatric data become available, clinicians should tailor therapy for individual patients on the basis of their assessment of the risks and benefits of hypothermia. Risk-benefit assessment should take into account relevant data from laboratory models of asp hyxial arrest [6], results from trials of adult cardiac arrest [15], and reports on the use of hypothermia in treatment of neonatal asphyxia. The results of TH are generally favorable in laboratory models of hypoxic ischemic injury to immature brains of various species. Preliminary data from clinical trials of perinatal asphyxia indicate that induced hypothermia is feasible and safe, but data on long-term neurological morbidity are not yet available [15].
It is difficult to extrapolate from these disparate sources of information, and thus there is no consensus yet for use of therapeutic hypothermia among clinicians who care for these critically ill children [14].
Materials and Methods
After the cardiac arrest, level of oxygen decreases and an ischemic cascade takes place. The permanent damage of neuronal cells occurs after 5 min to 10min of complete blood flow cessation .The consequence of this decrease leads to a change in metabolism from aerobic to anaerobic, resulting in intra- and extra-cellular acidosis. The acidosis and high calcium levels also stimulate the cell apoptosis and cause cell death.
Mild Therapeutic Hypothermia (MTH) reduces cerebral metabolic rate for oxygen by 6%–10%for every 1 1C temperature reduction .Hypothermia is likely to lower lactate levels and other wastes from anaerobic metabolism decreasing cellular acidosis.
Hypothermia significantly reduces extracellular levels of excitatory neurotransmitters. The production of free radicals is associated with oxidative damage that is minimized at lower temperatures. (45) Different studies were performed to compare between normothermia and hypothermia and result was they have equal effect in outcome (Figure 4).
Over a 10-year period, the percentage of survivors has increased from 13.7 to 22.4 %, while the rate of clinically significant neurological impairment has declined from 32.9 to 28.1 % .These results are encouraging.
It is thought that therapeutic hypothermia can affect and simultaneously block multiple metabolic pathways, inflammatory reactions and apoptosis processes that occur after an ischemic cascade, so they prefer hypothermia than normothermia [9].
Many single-center studies and metaanalyses demonstrate that therapeutic hypothermia (TH), in which the body temperature is maintained at 32-35°C, exerts significant neuro protection and attenuates secondary intracranial hypertension after traumatic brain injury (TBI). In 2015, two well-designed multi-center, randomized controlled trials were published that did not show favorable outcomes with the use of TH in adult patients with TBI compared to normothermia treatment (NT) [12].
Neurologic outcome was evaluated after return of spontaneous circulation and was expressed in term of cerebral performance category (CPC). CPC 1, conscious and alert with normal function or only slight disability. CPC 2, conscious and alert with moderate disability.
CPC 3, conscious with severe disability. CPC 4, comatose or in a persistent vegetative state. CPC 5, certified brain death or dead by traditional criteria (Figure 5).
Hypothermia performed within three phases, activation, maintenance and rewarming. Clinicians must control hypothermia and rewarming to prevent potential adverse effects, such as arrhythmias and skin breakdown during the cold phases (induction and maintenance) and rapid electrolyte shifts during the rewarming phase. Temperature should be monitored with a method that measures core temperature, such as use of an esophageal, bladder, or pulmonary artery catheter. (See Body temperature during therapeutic hypothermia phases).
Induction phase: Our protocol also calls for sedation and neuromuscular blockade when the cooling process begins, to prevent shivering during induction; this allows rapid cooling to target temperature. Drug selection for neuromuscular blockade varies among formularies.
Maintenance phase: During the maintenance phase, controlling the patient’s temperature within the target range (usually 32° to 34° C) is crucial
This phase can last up to 24 hours from the time the target temperature is reached (depending on facility protocol). Automated invasive and noninvasive methods can be used to keep the patient within range; these methods are much less labor-intensive than non-automated methods.
Rewarming phase: Temperature control remains important during rewarming. Warming the patient too quickly or allowing continued shivering causes dangerous electrolyte shifts, leading to potentially lethal arrhythmias. Controlled rewarming of 0.15° to 0.5° C per hour is recommended. To maintain tight temperature control throughout rewarming, our protocol calls for a neuromuscular blockade.
Careful fluid monitoring during rewarming is crucial because of the vasodilation that accompanies a body temperature rise. Volume replacement may be needed to prevent fluid deficit and hypotension.
Duration of therapeutic hypothermia depends on facility protocol. No research is available on optimal duration. Some facilities start counting duration from the time cooling begins; others start when the patient reaches target temperature (Figures 6,7).
Results
Throughout the studies we have discussed it shows that
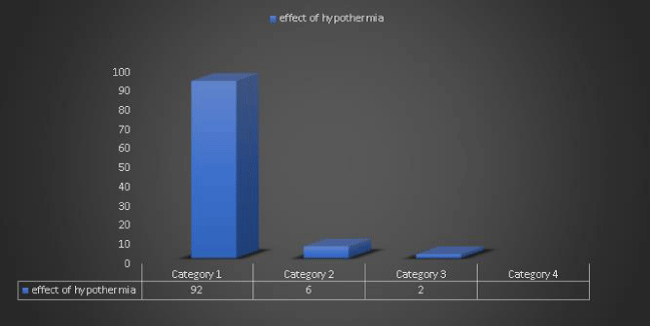
-92% Survival with good neurological outcome as it is feasible, safe used. Neither safety concerns nor costs should be a barrier for implantation of TH.
-4% of survivors after OHCA treated with TH the induced hemodynamic changes reversed after normothermia, while the need for vasopressor support persisted. Patients requiring addition of norepinephrine/epinephrine on top of dopamine had an increased 30-day all-cause mortality, as well as death from neurological injury.
No benefit to prehospital initiation of therapeutic hypothermia in out-ofhospital cardiac arrest.
-6% shows worse outcome as
• Hypothermia and rapid rewarming is associated with worse outcome following traumatic brain injury.
• Increased Thyroxin during Therapeutic Hypothermia predicts death in comatose patients after cardiac arrest.
• -Therapeutic hypothermia after cardiac arrest: another confounding factor in brain-death testing (A 10-month-old infant with reversible findings of brain death).
Groups in knowledge (things for further researches)
- There is no evidence that hypothermia has the same effect on heart as brain.
- Many patients survive from cardiac arrest with high outcome but with different variations which still unclear.
Conclusion
Hypothermia is proving to be an extremely robust and important therapy for cardiac arrest survivors and so far the only therapy consistently shown to reduce mortality and improve neurological outcomes in cardiac arrest survivors.
Mild therapeutic hypothermia improves short term neurologic recovery and survival in patients resuscitated from cardiac arrest of presumed cardiac origin. Its long term effectiveness and feasibility at an organizational level need further.
Therapeutic hypothermia for the treatment of post cardiac arrest coma can be successfully implemented –k---intensive care practice with a major benefit on patient outcome, which appeared to be related to the type and the duration of initial cardiac arrest and seemed maintained in patients with shock.
After studies, they conducted a prospective controlled trial comparing moderate induced hypothermia with normothermia in comatose survivors of (OOHCA). The study proved that hypothermia improves outcome more than normothermia.
The use of induced hypothermia after cardiac arrest requires an understanding of physiolological effect of (IH) in each organ system. -Mild resuscitative hypothermia in patients is feasible and safe. A clinical multicenter trial might prove that mild hypothermia is a useful method of cerebral resuscitation after global ischemic states .and not associated with an increase in length of stay in the ICU. And finally, hypothermia has different consedration for arrested children. Most research contraindicate using hypothermia with them. Mild hypothermia of 33 to 34°C seems to be a sufficient compromise between beneficial effects through mitigation of ischemic membrane damage and reperfusion. Injury and the risk of adverse effects through arrhythmic, inflammatory, hemodynamic, and coagulation disorders. Hyperthermia is not tolerable any longer in the course of intensive care treatment after cardiac arrest; temperature control to normothermia with antipyretic drugs is the least that should be done.
Recommendation
Therapeutic hypothermia is recommended for the treatment of neurological injury after resuscitation from out-of-hospital cardiac arrest. Laboratory studies have suggested that earlier cooling may be associated with improved neurological outcomes.
Much of our recommendations around the therapy are based on limited literature and a lot of extrapolation.
Dr. Muhammad Alsayegh (Surgical Resident at Helwan University – Badr Hospital and intern at Kasr Al-Ainy Medical School)
Dr. Manal R. Abd El-Haleem Assistant Professor at Egyptian Russian University (ERU), Associate Professor at Faculty of Oral and Dental Medicine-ERU and associate Professor at Faculty of Medicine Zagazig University.
- Abella BS (2013)
- Laver S, Farrow C, Turner D, Nalon J (2004) Mode of death after admission to an intensive care unit following cardiac arrest. Intensive Care Medicine 30: 2126-2128. Link: https://tinyurl.com/ya8nwfm5
- The Hypothermia after Cardiac Arrest Study Group (HACA) (2002) Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med 346: 549–556. Link: https://tinyurl.com/yb8dxvos
- Tariq N, Maud A, Shah QA, Suri MF, Qureshi AI (2011) Clinical outcome of patients with acute posterior circulation stroke and bilateral vertebral artery occlusion. J Vasc Interv Neurol 4: 9-14. Link: https://tinyurl.com/ycpn6aw7
- Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, et al. (2002) Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med 346: 557–563. Link: https://tinyurl.com/y8vy5tsd
- Holzer M, Cerchiari EL, Martens PR (2002) Hypothermia after Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med 346: 549-556. Link: https://tinyurl.com/y8uc9ojd
- Bernard SA, Buist M (2003) Induced hypothermia in critical care medicine: a review. Crit Care Med 31: 2041–2051. Link: https://tinyurl.com/yajekrfa
- Polderman KH (2004) Keeping a cool head: How to induce and maintain hypothermia. Crit Care Med 32: 2558–2560. Link: https://tinyurl.com/y9ypktvx
- Badjatia N, Guanci M, Rordorf G (2006) Rapid infusion of cold saline as adjunct treatment of fever in patients. Neurology. Link: https://tinyurl.com/y9mdxgsr
- Sterz F, Holzer M, Roine R, Zeiner A, Losert H, et al.(2003) Hypothermia after cardiac arrest: a treatment that works. Curr Opin Crit Care 9: 205–210. Link: https://tinyurl.com/yaplaj3r
- Management of Cardiac Arrest due to Trauma. Australian resuscitation council. ANZCOR Guideline 11.10.1. Link: https://tinyurl.com/y8bnzq7m
- Scirica BM (2013) Therapeutic Hypothermia after Cardiac Arrest. Circulation 127: 244-250. Link: https://tinyurl.com/yaz9rfbp
- (2011) Maine Medical Center Neuroscience Institute and Department of Critical Care Services 82: 1100-1113. Link: https://tinyurl.com/yc28d5zp
- Nolan JP, Morley PT, Vanden Hoek TL, Hickey RW, Kloeck WG, et al. (2003) Therapeutic Hypothermia After Cardiac Arrest: An Advisory Statement by the Advanced Life Support Task Force of the International Liaison Committee on Resuscitation. Circulation 108: 118-121. Link: https://tinyurl.com/y9pvww4e
- Compagnoni G, Pogliani L, Lista G, Castoldi F, Fontana P, Mosca F (2002) Hypothermia reduces neurological damage in asphyxiated newborn infants. Biol Neonate 82: 222–227. Link: https://tinyurl.com/y7kcm697
- Waters JM (2014) Therapeutic hypothermia after cardiac arrest. American Nurse Today 9. Link: https://tinyurl.com/ycjt4j7p
- González-Ibarra FP, Varon J, López-Meza EG (2011) Therapeutic hypothermia: critical review of the molecular mechanisms of action. Front Neurol 2: 4. Link: https://tinyurl.com/yb2p55cn
- Youfeng Zhu, Haiyan Yin, Rui Zhang, Xiaoling Ye, and Jianrui Wei (2016) Therapeutic hypothermia versus normothermia in adult patients with traumatic brain injury Springerplus 5: 801. Link: https://tinyurl.com/y7gkbxaj
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.




 Save to Mendeley
Save to Mendeley
