Journal of HIV for Clinical and Scientific Research
Progressive multifocal leukoencephalopathy, more than an encephalopathy
David Sáez M1*, Nadia Valdés R1, Cristian Fernández M2 and Rocío Pacheco E1
2Neurologist, Lucio Cordova Hospital for Infectious Diseases, Santiago, Chile
Cite this as
David Sáez M, Nadia Valdés R, Cristian Fernández M, Rocío Pacheco E (2020) Progressive multifocal leukoencephalopathy, more than an encephalopathy, J HIV Clin Sci Res 9(1): 001-004. DOI: 10.17352/2455-3786.000032Copyright
© 2022 David Sáez M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Progressive multifocal leukoencephalopathy is a pathology associated with the presence of the JC virus, although it occurs in other conditions, nowadays most cases occur in patients with AIDS, in severe stages of immunosuppression. This condition, and hence its name, has always been associated with the presence of intracerebral lesions, where it can have a wide distribution. However, it is very rare, if ever, to report lesions of this disease in the spinal cord. We present a case of a 40-year-old patient, an HIV carrier in the AIDS stage, who, in addition to presenting brain lesions, had multiple lesions at the cervical spinal cord level. We believe that these lesions may be more common than believed and should be intentionally sought if possible.
Introduction
Progressive multifocal leukoencephalopathy (PML) is a demyelinating disease of the central nervous system (CNS) with poor prognosis, caused by reactivation of the John Cunningham (JC) polyomavirus in patients with severe immunosuppression.
The clinical presentation is varied, because of lesions that may develop anywhere in the white matter of the CNS. However, in contrast with other demyelinating diseases, it appears to avoid the optic nerves and spinal cord [1,2]. Diagnosis is based on clinical findings, neuroimaging, and the search for JC virus by polymerase chain reaction (PCR) in cerebrospinal fluid (CSF) or by brain tissue biopsy.
In the literature, there are few reports of PML with spinal cord involvement, all of them with histopathologic diagnosis [1,2]. We present a case of PML in a patient with acquired immunodeficiency syndrome (AIDS), who presented lesions in the cervical spinal cord, discovered incidentally during magnetic resonance imaging (MRI) of the brain.
Case report
A 40-year-old woman, with a history of hypothyroidism and a recent diagnosis of HIV, presents symptoms of 2 months of evolution, consisting of dizziness, dysarthria, and gait instability, which is exacerbated during the month before consultation, requiring support to move around. It’s associated with temporospatial disorientation and alteration of working memory, forgetting the purpose of activities, and losing the train of thought. This symptomatology generates difficulties in the performance of instrumental activities of daily living. She denies other symptoms such as paresthesias, weakness, and sphincter incontinence.
Her CD4 T-lymphocyte count was 13, with a viral load of Log 6.3. The patient had not yet started antiretroviral therapy (ART). Her initial neurological examination showed temporal disorientation, inattention, and compromised anterograde memory, with psychomotor slowing, puerile and concrete behavior. MoCA test was applied with a total score of 13 and ACE-III [3] with a total score of 56 (Table 1).
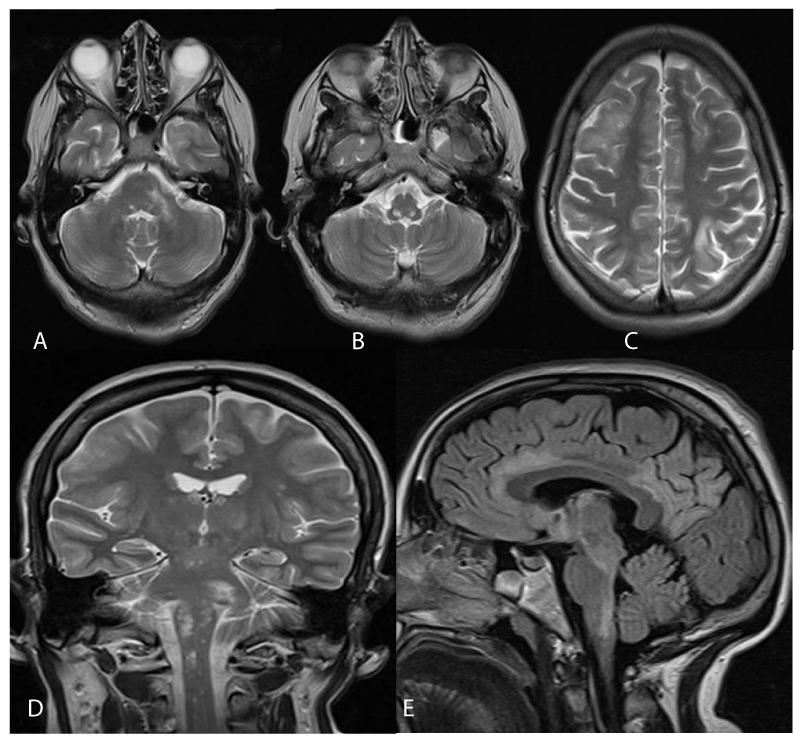
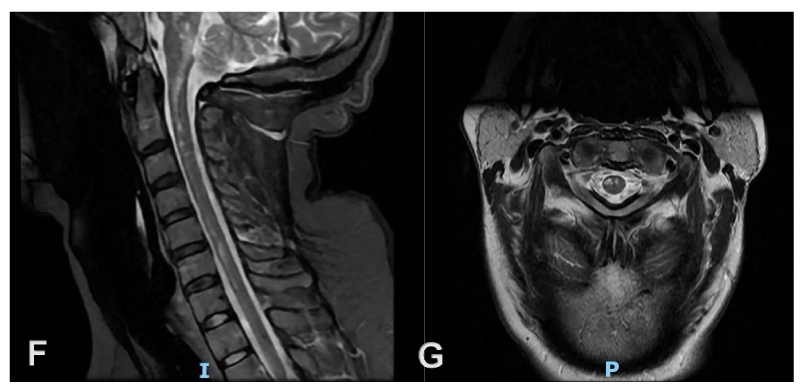
Brain MRI showed supra and infratentorial T2/FLAIR hyperintense lesions in white matter, with and without U-fibers involvement, with involvement of basal ganglia, high spinal cord, medulla oblongata-pontine, and left pontine. Without enhancement post-administration of contrast agents. Some hypointense in T1. No diffusion restriction. (Figure 1 A-E). In Figure 2, cervical spinal cord lesions are shown, in Stir and T2 sequences (F and G respectively) where white matter lesions are seen.
CSF analysis showed: Leukocytes 0, red blood cell 1, proteins 18.4, Glucose 56, lactic acid 17.19 and confirmed the presence of JC virus by PCR. Herpes Simplex Virus 1 and 2 (HSV), cytomegalovirus (CMV), enterovirus, tuberculosis (TB), and Epstein Barr Virus (EBV) were ruled out by the same technique. CSF culture was negative.
Negative results were obtained for hepatitis B and C viruses, VDRL, Chinese ink, capsular antigen for cryptococcus in blood and CSF, blood cultures, IgG Chagas, and toxoplasma. Systemic involvement was discarded by thorax, abdomen, and pelvis CT. Cerebral and cervical vessels CT angiography doesn’t show pathological findings, without signs suggestive of vasculitic process. Electroencephalogram was reported as “Bilateral frontal-temporal permanent theta slowness, without paroxysmal activity”.
ART was started after finding low CMV viral load and no clinical evidence suggestive of retinitis, being well-tolerated, and an absence of adverse reactions.
Discussion
Progressive multifocal leukoencephalopathy (PML) is a demyelinating disease of the CNS, potentially fatal or causing severe neurological sequelae, caused by reactivation of the John Cunningham (JC) polyomavirus. Primoinfection by JC virus usually occurs in childhood, remaining latent in different tissues. 40-80% of the world’s population has been in contact with the JC virus at some time [4-6]. However, subsequent reactivation, with the consequent development of PML, occurs in patients with severe immunodepression, the most frequent cause being secondary to HIV/AIDS, mainly in patients with a low CD4 lymphocyte count.
JC virus is a ubiquitous human pathogen, which generates a lytic infection of oligodendrocytes, with secondary demyelination. The clinical presentation may vary from patient to patient because lesions can develop anywhere in the CNS. However, they seem to avoid the optic nerves and spinal cord [1,2]. The size of the lesions is also variable, ranging from 1 mm to various centimeters, the latter being the result of the confluence of multiple smaller lesions.
The most frequent symptoms are motor weakness, visual disturbances, cognitive involvement, and behavioral changes. The disease can be classified into different variants: Classic LEMP, SIRI-associated LEMP, granulosa cell neuronopathy, encephalopathy, and JC virus meningitis [7].
Lesions are usually located in the subcortical hemispheric white matter. However, it can also affect other areas of the white matter such as the cerebellum, cerebellar peduncles, and brain stem [8-10]. Involvement of deep gray matter structures such as basal ganglia, thalamus, and cerebral cortex has also been reported. There are few reports of medullary and optic nerve involvement [1,2].
Koralnik, et al. [2] performed a neuropathological study on a patient with a history of HIV on ART, showing that the white matter lesions were characterized by extensive loss of oligodendrocytes, numerous macrophages, and reactive gliosis, including bizarre astrocytes. The oligodendroglial nuclei at the edges of demyelination had a characteristic spectrum of enlargement, peripheral marginalized chromatin, and vitreous inclusions. In the samples, thoracic spinal cord involvement was evident, with central myelin loss and characteristic peripheral nuclear changes. The enlarged nuclei of the oligodendrocytes were positively stained by immunohistochemistry with an SV40 antibody, which cross-reacts with the major capsid protein of JCV VP1.
Takeda, et al. [1] described the case of a patient with extensive LEMP to the spinal cord, in whom histopathological analysis showed mild Wallerian degeneration in the lateral cord on both sides and multifocal demyelinating lesions of varying severity. In the cervical segments, small and medium-sized foci of demyelination were found, with macrophage infiltration and gliosis in the anterior and lateral columns. In the thoracic segment, there was less involvement, with smaller lesions. The lumbar and sacral segments revealed more extensive lesions involving the anterior and lateral cords. There was gray matter involvement to varying degrees, with preservation of spinal neurons. In the posterior column, sparse foci of demyelination were visualized in the lumbosacral segments.
Is unknown why PML doesn’t seem to affect the spinal cord in most cases [11]. Among the hypotheses postulate that spinal cord lesions are underdiagnosed, as they most frequently debut with florid cerebral symptoms. Another alternative is that hematogenous dissemination of JC virus favors brain seeding rather than spinal cord seeding, or that immunological elimination of the virus is more effective at the spinal cord level.
The gold standard for diagnosis is brain biopsy, as it allows pathological confirmation of the presence of demyelinating lesions, bizarre astrocytes, and large oligodendroglial nuclei. Currently, the criteria allow making the diagnosis based on laboratory (PCR) and imaging (MRI) studies [12], thus avoiding the risks associated with brain biopsy.
MRI represents a very sensitive tool for diagnosis. In its classic presentation, a single or multiple lesion can be visualized, hypointense in T1 and hyperintense in T2 and FLAIR, typically involving the subcortical white matter, areas of greater blood flow, bilaterally and asymmetrically, tending to confluence, without mass effect and with extension to the subcortical U-fibers. The lesions progress to deep white matter, as in the semioval center and periventricular regions. The parietal and frontal lobes are most frequently affected [10,11]. Less frequent is the involvement of regions such as the internal capsule, external capsule, and corpus callosum [13,14]. Involvement of the white matter of the posterior fossa may also be found, with lesions involving the middle cerebellar peduncle, cerebellum, and pons, with extension into the midbrain. Due to no or little associated inflammation, peripheral enhancement after gadolinium administration is only observed in up to 10% of cases, which could be related to a better long-term prognosis. Spinal cord involvement is extremely rare, with only a few reports of an anatomopathological study confirming the involvement of this area. In the literature, don’t exist images documenting the presence of spinal cord lesions [15].
The disease can present a rapidly progressive course, especially in patients with severe immunosuppression. Doesn’t have a specific treatment, except for the initiation or optimization of antiretroviral therapy. Since its introduction, survival rates have increased from <30% to 38-62%, although associated with significant neurological sequelae [11,16,17]. Immune reconstitution inflammatory syndrome (IRIS) is an important complication, usually associated with ART.
Conclusion
PML corresponds to an acquired demyelinating disease of the CNS, caused by the reactivation of JC virus infection. Spinal cord involvement is extremely rare, and exist few cases reports in the literature that confirm spinal cord involvement by histopathological study, without imaging findings according to the literature search.
Regarding spinal cord involvement, few authors have described cases of PML involving the anterior and lateral cords in different segments, being more extensive at the cervical level. We present a reported case of a recently diagnosed patient with HIV, who presents PML lesions of unusual distribution, with extension to the cerebellar peduncle, brainstem, and cervical spinal cord, as evidenced in the MRI study.
- Takeda S, Yamazaki K, Miyakawa T, Takahashi H, Ikuta F, Arai H. Progressive multifocal leukoencephalopathy showing extensive spinal cord involvement in a patient with lymphocytopenia. Neuropathology. 2009 Aug;29(4):485-93. doi: 10.1111/j.1440-1789.2008.00981.x. Epub 2008 Nov 19. PMID: 19019177.
- Bernal-Cano F, Joseph JT, Koralnik IJ. Spinal cord lesions of progressive multifocal leukoencephalopathy in an acquired immunodeficiency syndrome patient. J Neurovirol. 2007 Oct;13(5):474-6. doi: 10.1080/13550280701469178. PMID: 17994433.
- Bruno D, Slachevsky A, Fiorentino N, Rueda DS, Bruno G, Tagle AR, Olavarria L, Flores P, Lillo P, Roca M, Torralva T. Argentinian/Chilean validation of the Spanish-language version of Addenbrooke's Cognitive Examination III for diagnosing dementia. Neurologia (Engl Ed). 2020 Mar;35(2):82-88. English, Spanish. doi: 10.1016/j.nrl.2017.06.004. Epub 2017 Sep 1. PMID: 28865943.
- Bozic C, Subramanyam M, Richman S, Plavina T, Zhang A, Ticho B. Anti-JC virus (JCV) antibody prevalence in the JCV Epidemiology in MS (JEMS) trial. Eur J Neurol. 2014 Feb;21(2):299-304. doi: 10.1111/ene.12304. Epub 2013 Nov 30. PMID: 24894998.
- Wattjes MP, Richert ND, Killestein J, de Vos M, Sanchez E, Snaebjornsson P, Cadavid D, Barkhof F. The chameleon of neuroinflammation: magnetic resonance imaging characteristics of natalizumab-associated progressive multifocal leukoencephalopathy. Mult Scler. 2013 Dec;19(14):1826-40. doi: 10.1177/1352458513510224. Epub 2013 Nov 5. PMID: 24192217.
- Wattjes MP, Vennegoor A, Steenwijk MD, de Vos M, Killestein J, van Oosten BW, Mostert J, Siepman DA, Moll W, van Golde AE, Frequin ST, Richert ND, Barkhof F. MRI pattern in asymptomatic natalizumab-associated PML. J Neurol Neurosurg Psychiatry. 2015 Jul;86(7):793-8. doi: 10.1136/jnnp-2014-308630. Epub 2014 Sep 9. PMID: 25205744.
- Camporro J, Bruno V, Cammarota A, Del Castillo M, Alessandro L. Espectro clinico de la leucoencefalopatia multifocal progresiva: diferencias y similitudes en pacientes con y sin virus de la inmunodeficiencia humana [The clinical spectrum of progressive multifocal leukoencephalopathy: differences and similarities in patients with and without human immunodeficiency virus]. Rev Neurol. 2019 Aug 16;69(4):152-158. Spanish. doi: 10.33588/rn.6904.2019040. PMID: 31334558.
- Barkhof F, Koeller KK. Demyelinating Diseases of the CNS (Brain and Spine). In: Hodler J., Kubik-Huch R., von Schulthess G. (eds) Diseases of the Brain, Head and Neck, Spine 2020–2023. IDKD Springer Series. Springer, Cham. 2020.
- Sakuraba M, Watanabe S, Nishiyama Y, Takahashi K, Nakamichi K, Suzuki M, Nawata T, Komai K, Gono T, Takeno M, Suzuki T, Kimura K, Kuwana M. Infratentorial onset of progressive multifocal leukoencephalopathy in a patient with systematic lupus erythematosus complicated with lymphoma: a case report. Mod Rheumatol Case Rep. 2021 Jul;5(2):272-277. doi: 10.1080/24725625.2021.1899763. Epub 2021 Apr 6. PMID: 33719867.
- Augusto L, Neves N, Reis C, Abreu C, Sarmento A. Clinical and Radiological Characterization of Progressive Multifocal Leukoencephalopathy in HIV-Infected Patients: A Retrospective Analysis and Review of the Literature. Acta Med Port. 2015 May-Jun;28(3):286-96. doi: 10.20344/amp.5950. Epub 2015 Jun 30. PMID: 26421780.
- Cortese I, Reich DS, Nath A. Progressive multifocal leukoencephalopathy and the spectrum of JC virus-related disease. Nat Rev Neurol. 2021 Jan;17(1):37-51. doi: 10.1038/s41582-020-00427-y. Epub 2020 Nov 20. PMID: 33219338; PMCID: PMC7678594.
- Bartsch T, Rempe T, Leypoldt F, Riedel C, Jansen O, Berg D, Deuschl G. The spectrum of progressive multifocal leukoencephalopathy: a practical approach. Eur J Neurol. 2019 Apr;26(4):566-e41. doi: 10.1111/ene.13906. Epub 2019 Feb 8. PMID: 30629326.
- Sahraian MA, Radue EW, Eshaghi A, Besliu S, Minagar A. Progressive multifocal leukoencephalopathy: a review of the neuroimaging features and differential diagnosis. Eur J Neurol. 2012 Aug;19(8):1060-9. doi: 10.1111/j.1468-1331.2011.03597.x. Epub 2011 Dec 5. PMID: 22136455.
- Wattjes MP, Richert ND, Killestein J, de Vos M, Sanchez E, Snaebjornsson P, Cadavid D, Barkhof F. The chameleon of neuroinflammation: magnetic resonance imaging characteristics of natalizumab-associated progressive multifocal leukoencephalopathy. Mult Scler. 2013 Dec;19(14):1826-40. doi: 10.1177/1352458513510224. Epub 2013 Nov 5. PMID: 24192217.
- Bag AK, Curé JK, Chapman PR, Roberson GH, Shah R. JC virus infection of the brain. AJNR Am J Neuroradiol. 2010 Oct;31(9):1564-76. doi: 10.3174/ajnr.A2035. Epub 2010 Mar 18. PMID: 20299430; PMCID: PMC7965014.
- Moulignier A, Lecler A. Complications neurologiques de l’infection par le virus JC : revue générale [Neurological complications of JC virus infection: A review]. Rev Med Interne. 2021 Mar;42(3):177-185. French. doi: 10.1016/j.revmed.2020.08.021. Epub 2020 Nov 6. PMID: 33168355.
- Moulignier A, Lecler A. Complications neurologiques de l’infection par le virus JC : revue générale [Neurological complications of JC virus infection: A review]. Rev Med Interne. 2021 Mar;42(3):177-185. French. doi: 10.1016/j.revmed.2020.08.021. Epub 2020 Nov 6. PMID: 33168355.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
