Journal of Dental Problems and Solutions
Effect of the additive dentine conditioning in all-in-one adhesives on the bonding strength at simulated intrapulpal pressure
Gerhardt T, Hasselmann S, Humpich T, Fielmann N, Giraki M, Rüttermann S and Gerhardt-Szép S*
Cite this as
Gerhardt T, Hasselmann S, Humpich T, Fielmann N, Gerhardt-Szép S, et al. (2019) Effect of the additive dentine conditioning in all-in-one adhesives on the bonding strength at simulated intrapulpal pressure. J Dent Probl Solut 6(1): 021-027. DOI: 10.17352/2394-8418.000068The aim of the study was to investigate the influence of additive etching of dentine with phosphoric acid on the shear bond strength of two different, self-conditioning, all-in-one adhesives [Adect (ADE), and One-Up-Bond F (OUP)]. Dentine discs from 90 human molars (n = 15 per group) were exposed to penetration by Ringer's solution (60 cm height, 22 °C) by means of a perfusion device. A two-step, self-conditioning adhesive system [Clearfil SE (CSE)] was used as the control group. The dentine was either treated (+) after preliminary additive etching wssith 36% phosphoric acid for 20 seconds or left untreated (-). Subsequently, the samples were provided with a composite cylinder (Herculite XRV). After thermocycling (5/55 °C, 5000 cycles, 20 s holding time, 4 s transportation time), the shear bond strength (MPa) was determined with a Zwick universal testing machine (feed rate 5 mm/min). Furthermore, parametric Weibull regression models were applied to evaluate whether there was a significant association between shear bond strength and the method of surface conditioning. Weibull analysis was performed using R software (version 2.11.1, R Foundation for Statistical Computing, Vienna, Austria). Differences between groups were tested using Wald tests, and p-values below 0.05 were considered significant.
No significant difference could be detected between the individual systems and the variation in the application of additive phosphoric acid etching of dentine. Under the limitations of the present study, it can be stated that the additive etching of dentine with phosphoric acid does not lead to a statistically significant decrease or increase in the dentine adhesion strength values for all-in-one adhesives.
Introduction
Dentine adhesives or bondings have become indispensable in modern restorative dentistry. Their application constitutes the standard practice in the processing of adhesive filling materials. Current trends are moving away from the classic so-called multi-step adhesives to systems with reduced work steps that are easier and faster to use and more user-friendly [1]. An example of reduced working steps is the development of self-conditioning or self-etching adhesives that can prepare enamel and dentine, for example, with the help of acidic monomers. Using these dentine bonding agents eliminates the separate etching with subsequent rinsing off of the acid. The acidic monomers contained in the primer or the all-in-one preparations dissolve hydroxyapatite crystals of the hard structures on teeth in order to ensure the subsequent infiltration of the monomers [1]. Some studies suggest that all-in-one adhesives might perform as well as etch-and-rinse and two-step self-etch adhesives. Others, however, report that all-in-one adhesives have a lower dentine bonding strength than etch-and-rinse and two-step self-etch adhesives [2]. The elimination of the separate etching process saves the user time on the one hand, and, on the other hand, technology-sensitive problems can be prevented, such as those that may occur in the application of total-etch adhesives. However, it is known from the literature that they show lower bonding strength to the enamel than do etch-and-rinse systems.
To overcome this disadvantage, some manufacturers recommend preliminary selective enamel etching, especially if non-instrumented margins are present [3-5]. Preliminary etching could represent a clinical handling problem in small cavities because small excesses of etching gel could extend into the dentine and unintentionally demineralise the substrate [3]. While preliminary etching of the enamel improves the bond strength and margin stability for all self-etch (or etch-and-dry) adhesives, preliminary etching of dentine has shown contradictory results [3,6,7]. Moreover, recent studies have highlighted the importance of enzymatic degradation of suboptimally impregnated collagen fibrils that remain within the hybrid layer, and that can be degraded following the activation of collagen-bound matrix metalloproteinases [3,8-12].
In general, attempts should be made to create real conditions in in vitro tests in order to ensure careful transferability to in vivo human situations. These include, among other things, the simulation of intrapulpal pressure, which has already been frequently requested by several authors [13-15], as well as the preparation of a cavity as a dentine bonding surface [16]. Distilled water or saline has often been used to simulate dentinal liquor. Studies have shown varied results as the effect of hydrostatic intrapulpal pressure on shear bond strength (SBS) depends on the type of dentine bonding system employed [17,18].
To our knowledge, no previous study has investigated the SBS of the adhesive interface created by pre-etched dentine and one-step self-etch adhesives under a simulated intrapulpal pressure protocol. In this study, we evaluated the bond strength of two one-step self-etch adhesive systems (with one versus two components) after pre-etching of the dentine surface compared with one two-step self-etch adhesive as a control group. The research hypothesis tested was that dentine pre-etching with phosphoric acid would affect the SBS.
Materials and Method
Apparatus for measuring the shear bond strength
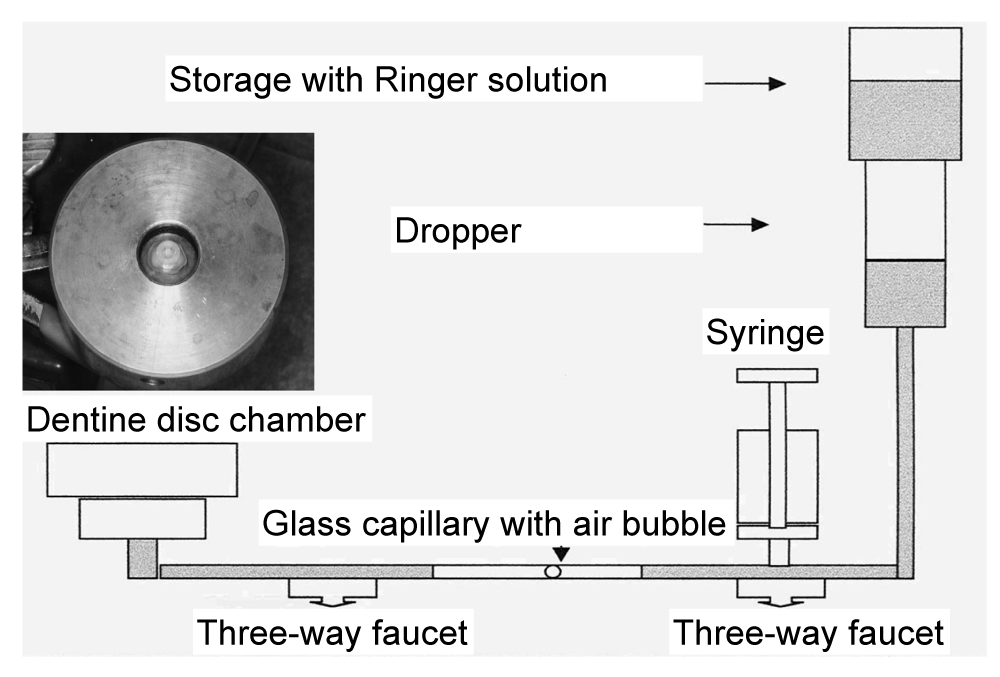
As a precondition for the further procedure, the test room was set up to have a constant temperature of 22 °C and a humidity of 50%. The dentine discs produced were inserted into a disk chamber that had been modified by Szép in 2003 and 2012 (Figure 1); the disk chamber allows the simulation of the intrapulpal fluid flow [19,20]. At the end of the apparatus was a 60 cm high liquid column of Ringer's solution (22 °C), the level of which was kept constant by a gravity infusion device (Intrafix Air, Braun, Melsungen, Germany) (Figure 2). The dentine discs were placed in the disc chamber so that the composite samples (Herculite XRV, colour B1, Kerr-HaweNeos, Karlsruhe, Germany) could be applied to the polished side (away from the pulp). After screwing the brass chamber closed, it was first checked whether the disc closed tightly or whether Ringer's solution seeped onto the disc sideways. If the latter was the case, the disk had to be placed again on the silicone seal rings, centrically.
Measurement of SBS
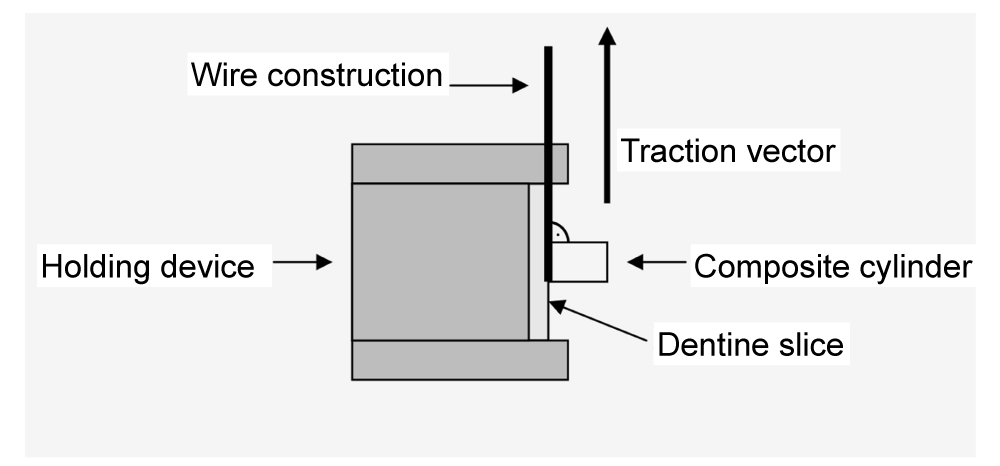
A universal testing machine (Zwicki 1120, Zwick, Ulm, Germany) was used for the SBS test (Figure 3). SBS was determined after storing the samples in water at 37 °C for 24 h. The test samples were locked in a clamp device with screws that consisted of a measuring cylinder with a one-sided round milling (0.21 cm). A ligature wire (3M Unitek, strength 0.1 mm, 3M, Monrovia, CA) served as the connection between the sample and the force generator. This wire was bent into a loop and fitted to the base of the composite cylinder, without applying tension. The two ends were fixed tightly to the pull arm of the shearing machine. The dentine discs were always aligned parallel to the direction of pull. The crosshead speed was 5 mm/min. SBS was calculated from the force at specimen failure, divided by the bonded area.
Tooth substrate and time after extraction
A total of 90 intact human molars stored in 0.1% thymol solution was used for the study. All teeth were extracted for not longer than six months, were caries-free, unrestored and not root filled.
Tooth surface preparation
All teeth were cleaned of tissue remnants using scalers and curettes. After randomised splitting, the teeth were roughened in the enamel area with a torpedo-shaped, diamond-coated instrument (ISO 012, Brasseler, Lemgo, Germany) under water cooling. Subsequently, the enamel surface was conditioned by means of 36% phosphoric acid (De Trey Conditioner 36, Dentsply, Constance, Germany) and sprayed off with water for 20 s. After the excess moisture was removed with the air blower, the samples were embedded in cold polymers (Technovit 4004, Kulzer, Hanau, Germany). Perpendicular to their crown axis under water cooling at a polishing unit (Jean Wirz, Düsseldorf, Germany) with grinding wheels of different grain sizes (80-180-320-600 grit) at 6000 rpm, the samples were reduced. The complete removal of enamel from the dentine was visually determined. Subsequently, the dentine surface was polished under water cooling with a felt disk (Leco Corporation, MI) at
3000 rpm and an aqueous solution of polishing earth, the remains of which were removed using distilled water. The production of 800-μm-thick discs took place using a saw microtome (SP 1600, Leica, Bensheim, Germany) under constant water cooling. The basal surface of the prepared discs was examined for the presence of any broached pulp horns, which were removed if found. The 90 discs obtained for the study were cleaned with distilled water and stored at 22 °C in Ringer's solution (DAB 7, Braun, Melsungen, Germany).
Application of the dentine adhesives and the composite
The liquor flow was started during the application of the dentine adhesives and the composite. The minimal displacement of the air bubble within the glass capillary allowed the permeability of the dental discs to be controlled. Even the dark areas, which could be observed after a short time on the windows, were an indicator of their permeability.
This was followed by pretreatment of the dentine (Table 1). Subsequently, a prepared glass cylinder (Schott, Mainz, Germany; interior diameter: 3 mm; outer diameter: 6 mm; height: 4 mm) was placed centrically on the dentine disc so that it did not extend beyond the dentine disc. The hybrid composite was applied in layers using a metal instrument (condenser) and hardened and a polymerisation lamp (Elipar Highlight No. 3803395, Espe, Seefeld, Germany) for 40 s/layer. The cylinder was held in place with a dentist's tweezers. The maximum thickness of the layer was 2 mm. The lamp was checked for constant luminance with the aid of a luxameter before use and positioned so that the opening for the light exit was aligned at a small distance and parallel to the dentine discs. Upon completing the hardening process, the simulated intrapulpal fluid flow was discontinued by closing the anterior three-way valve (between the dentinal disc chamber and the glass capillary). The disc with the attached composite cylinder was removed from the chamber and then the glass cylinder, still in place, was removed gently and without force. During the experiment, the temperature was kept constant at 22 °C using an air conditioner. All samples were then exposed to 5000 cycles of thermal cycling (Willytech Thermocycler V.2.8, Haake, Munich, Germany) between 5 °C and 55 °C; each bathing time lasted 30 s and there was a 10 s transportation time between baths.
Treatment of results and statistical analysis
To detect a difference between the groups, a sample of 15 dentine discs per test and control group was necessary to have an 80% power at alpha = 0.05 [19]. The utilised minimum clinically significant difference was stated with 3 MPa. For the statistical analysis of the results, the Kruskal-Wallis multiple-comparison Z-value test with a significance level of p ≤ 0.05 was applied. The test was performed using the statistical analysis package NCSS (Version 6.0.2.1. Kaysville, Utah). Furthermore, parametric Weibull regression models were applied to evaluate whether there was a significant association between SBS and the method of surface conditioning. Weibull analysis was performed using R software (version 2.11.1, R Foundation for Statistical Computing, Vienna, Austria). Differences between groups were tested using Wald tests, and p-values below 0.05 were considered significant.
Results
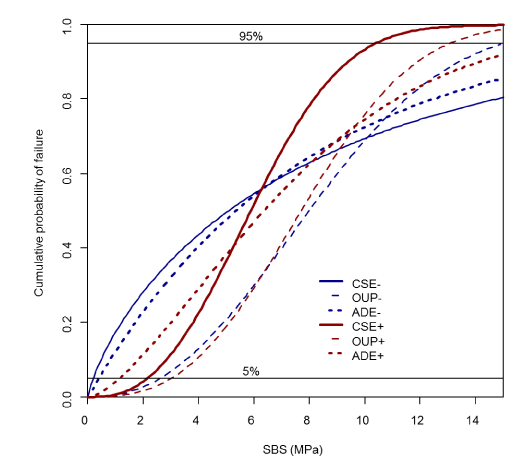
The individual results are shown in tables 2,3 and Figure 4. No significant difference could be detected between the individual systems and the variation in the application of additive phosphoric acid etching of dentine.
Discussion
There is consensus in the scientific literature that clinically prospective tests are most suitable for realistically assessing the quality of dentine adhesives in the oral cavity environment [21,22]. Since the materials are developed and presented in the market in a short time, it is necessary to find experimental methods with which one can quickly and easily make statements about products’ effectiveness before subjecting them to timeconsuming and expensive in vivo studies [23]. In general, attempts should be made to create real conditions in in vitro tests in order to ensure a careful transferability to in vivo human conditions. These include, among other things, the simulation of intrapulpal pressure, which has already been frequently requested by several authors, as well as the preparation of a cavity as a bonding surface [13,16]. However, the evaluation of durability is essential for clinical success. To achieve this and a possibility to include the ageing process in vitro, the simulation of the thermal cycling in the intra-oral environment is important. The ISO standard for these so-called thermocycling tests in connection with bond tests calls for 500 cycles in water between 5 °C and 55 °C for the artificial ageing of the samples [23]. In this context, the current work with its near-in vivo simulation is intended to contribute to bridging the experimental gap between laboratory tests and clinical studies. For this purpose, the intrapulpal pressure was simulated (height of the liquid column: 60 cm; medium: Ringer's solution) during the application of the composite samples to the dentine slices, and the samples were artificially aged before debonding (thermocycling: 5000 cycles/5–55 °C).
The dentine adhesives investigated differ in their composition, which may also explain the determined shear bonding strengths. They contain, among other ingredients, various solvents. Clearfil SE Bond and One-up Bond F are water-based adhesives, while Adect has acetone and water among its components. Solvents, as so-called carriers, have the task of transporting the ingredients of the adhesion promoters dissolved in them into the demineralised dentine structures and then releasing them by evaporation. Publications report lower dentine bonding values of hydrous adhesives compared to acetone- or ethanol-containing adhesives. They speculate that residual water, which has not evaporated, prevents complete polymerisation of the monomers within the hybrid layer [24]. In the current study, the type of solvent did not affect the results.
One of the three adhesion-promoting systems investigated, Clearfil SE Bond, contains silanised colloidal silicon as the filler particles in the bonding liquid. The filling content is about 10%. Clearfil SE Bond is one of the socalled filled adhesives. Adhesives transferred with filling materials tend to produce better bonding values and higher edge impermeability than those without filling [25]. In the current work, no significant differences could be determined in the bonding values between the unfilled (Adect and One-Up-Bond F) and the filled dentine adhesives (Clearfil SE Bond). Montes et al. (2001) determined bonding values for a filled and an unfilled adhesive using the microtensile method [26]. They were also unable to achieve better results for the filled product. It can be assumed that the advantage of filled bonding promoters in the current experimental setup is not noticeable because the composite was applied to a flat dentine surface. The polymerisation shrinkage can proceed without exerting a considerable tensile load on the adhesive composite since the C-factor is imperceptible. The influence of fillers on the efficiency of adhesives will not be realised until the bonding of samples recovered from restored cavities is determined.
Comparing adhesion values obtained in this work with those of other investigators, considerable difference is evident in some cases with our test results being the lowest. There is ample evidence that intrapulpal pressure in the form of intrinsic moisture can affect the efficiency of dentine adhesives [22,27]. Besnault et al. (2001) investigated the shear bonding strength of Clearfil SE Bond under in vivo conditions by which the bonding process took place at 35 °C and 95% humidity [28]. In an oral cavity, the average temperature is 30 °C and the average humidity is between 80 and 95%. These values depend on the location inside the mouth and are measured without a rubber dam. The bonding values determined under these conditions are in the ballpark of their determined values.
In dentine, acidic agents remove the smear layer, demineralise the dentine surface, open the dentine tubules, and increase the microporosity of the intertubular dentine. The interaction of the etching agents with dentine is limited by the buffering effect of the hydroxyapatite, smear layer, and protein components [29]. Despite these results, when the self-etching bonding systems tested in this study were used associated with phosphoric acid pretreatment in dentine, no significant differences in shear bond values were found, as already described [29]. Excessive etching of the dentine surface might have decreased—however, not statistically significantly—the bond strength owing to an incomplete infiltration of the adhesive to the base of the over-etched demineralised collagen network [29]. However, it should be noted that the SBS in one of the adhesives tested (Adect) led to an increase in the values but without any determinable statistical significance. The adhesive capabilities of self-etch adhesives show important differences among formulations, mainly as a result of differences in pH [29]. Typically, 'strong' self-etch systems have a pH <1.0; this high acidity results in deep demineralisation, similar to that of phosphoric acid [29]. The application of such adhesives to dentine exposes the collagen fibrils and dissolves nearly all the hydroxyapatite [29]. Conversely, ‘mild’ self-etch adhesives have a reduced etching potential as a result of their higher pH (pH 1.5–2.5) [29]. Because the pH values of the adhesive systems tested in the present study were 1.2–2.0 (Table 1), the smear layer was sometimes removed fully or partially and the dentine-bonding systems interacted with smear-layer-covered dentine up to a few hundred nanometres. As not all hydroxyapatite was dissolved from the interaction zone, much calcium was available for additional chemical interaction with specific monomers [29]. Clearfil SE Bond, a well-documented, mild, two-step self-etch adhesive system is considered the gold standard for self-etch adhesives and its bonding ability has been validated by several in vitro studies [29]. Its improved ability to bond to dentine in vitro has been related to the presence of 10-MDP, which can react with residual hydroxyapatite within the hybrid layer [29]. Clinically, Peumans et al. reported that the use of Clearfil SE Bond achieved a 98% retention rate at 8 years in non-carious cervical lesions, with or without separate enamel etching [30,31]. Similarly, another in vivo study showed that Clearfil SE Bond achieved better annual retention rates up to 8 years in comparison with a two-step etch-and-rinse system [31, 32]. In the present study, Clearfil SE Bond did not yield the highest SBS values; One-up-Bond F performed better, but no statistically significant difference was found between these adhesives after ageing. Interestingly, our results did not show superior performance of two-step versus one-step self-etch adhesives as reported in some previous studies [31]. Among currently available systems, one-step self-etch adhesives contain the highest percentages of hydrophilic monomers [31]. Simplified (one-step) self-etch adhesives are characterised by increased water sorption (vs two-step adhesives), which promotes polymer swelling and other water-mediated degradation phenomena [31]. Recent studies have also indicated that adhesive permeability is correlated with the presence of unreacted monomers, and one-step self-etch adhesives have shown greater permeability associated with the lowest degrees of conversion in comparison with unsimplified (two-step) systems [31].
From a clinical point of view, mean SBS values are less relevant than the likelihood that strong adhesion will be achieved with a given system or that its bond strength will be low and the risk of adhesive failure high [19]. The Weibull distribution takes this probabilistic approach of adhesive evaluation into account. The Weibull analysis in this study showed no differences between the groups. The Weibull parameter m was highest in the study group B (One-Up-Bond F (OUP)+) with 2.76. Lowest values were achieved in the control group (Clearfil SE (CSE)-) with 0.79, indicating low reliability. Thus, we conclude that adhesives showing low Weibull moduli are generally technique-sensitive and should not be favoured [19]. In this study, different adhesives were used in two different protocols. The protocol in group A (Adect (ADE)-) produced lower m-values in comparison with ADE+ and with OUP-, OUP+. Nevertheless, SBS was poor in both test groups.
Our results suggest that shear bond strength is either related to the number of steps characterising the dentine bonding systems or to their chemical formulations [31]. A systematic review and meta-analysis were performed to compare the longevity of self-etch dentine bonding adhesives to etch-and-rinse dentine bonding adhesives. In this study, the longevity of dentine adhesives was related to the measured mTBs. Although etch-and-rinse bondings showed higher values at all times, the meta-analysis found no difference in longevity between the two types of adhesives at the examined ageing times [33], which corresponds with our findings. The preliminary etching with phosphoric acid did not lead to a significantly detectable change in the shear bond values determined when applying the adhesives used. This is partly consistent with previously published data by the von Erhardt research group [29]. However, another publication reports a decrease in the bonding values of selfetching adhesives with preliminary etching by 35% phosphoric acid [34]. This shows that there is still insufficient evidence present. The current study may clarify the problem one step further. And yet, the use of allin-one systems in composite-to-dentine bonding restorative techniques needs still further improvement.
Conclusion
Under the limitations of the current study, it can be stated that the additive etching of dentine with phosphoric acid does not lead to a statistically significant decrease or increase in the dentine bonding strength values for allin-one adhesives tested within a simulated intrapulpal pressure protocol.
- Sofan E, Sofan A, Palaia G, Tenore G, Romeo U, et al. (2017) Classification review of dental adhesive systems: From generation IV to the universal type. Ann Stomatol (Roma) 8: 1-17. Link: http://bit.ly/2XPjDRl
- Walter R, Swift EJ, Nagaoka H, Chung Y, Bartholomew W, et al. (2012) Twoyear bond strengths of "all-in-one" adhesives to dentine. J Dent 40: 549-555. Link: http://bit.ly/2XulVG8
- Taschner M, Nato F, Mazzoni A, Frankenberger R, Falconi M, et al. (2012). Influence of preliminary etching on the stability of bonds created by one-step self-etch bonding systems. Eur J Oral Sci120: 239-48. Link: http://bit.ly/30eiWhT
- Tay FR, Pashley DH, King NM, Carvalho RM, Tsai J, et al. (2004). Aggressiveness of self-adhesive adhesives on unground enamel. Oper Dent 29: 309-316. Link: http://bit.ly/32bX74D
- Khosravi K, Ataei E, Mousavi M, Khodaeian N (2009) Effect of phosphoric acid etching of enamel margins on the microleakage of a simplified all-in-one and a self-etch adhesive system. Oper Dent 34: 531. Link: http://bit.ly/30ema50
- Taschner M, Nato F, Mazzoni A, Frankenberger R, Krämer N, et al. (2010). Role of preliminary etching for one-step self-etch adhesives. Eur J Oral Sci 118: 517-524. Link: http://bit.ly/2L0zxCl
- Erickson RL, Barkmeier WW, Kimmes NS (2009) Fatigue of enamel bonds with self-etch adhesives. Dent Mater 25: 716-720. Link: http://bit.ly/30bVNgb
- Pashley DH, Tay FR, Yiu C, Hashimoto M, Breschi L, et al. (2004) Collagen degradation by host-derived enzymes during aging. J Dent Res 83: 216-221. Link: http://bit.ly/2FX8xzI
- Mazzoni A, Mannello F, Tay FR, Tonti GA, Papa S, Mazzotti G, et al. (2007) Zymographic analysis and characterization of MMP-2 and -9 forms in human sound dentine. J Dent Res 86: 436-440. http://bit.ly/2Nz0FL3
- Nishitani Y, Yoshiyama M, Wadgaonkar B, Breschi L, Mannello F, et al. (2006) Activation of gelatinolytic/collagenolytic activity in dentine by self-etching adhesives. Eur J Oral Sci 114: 160-166. Link: http://bit.ly/327dkrE
- De Munck J, Van den Steen PE, Mine A, Van Landuyt KL, Poitevin A, et al. (2009) Inhibition of enzymatic degradation of adhesive-dentine interfaces. J Dent Res 88: 1101-1106. Link: http://bit.ly/2xyfu5r
- Osorio R, Yamauti M, Osorio E, Ruiz-Requena ME, Pashley D, et al. (2011) Effect of dentine etching and chlorhexidine application on metalloproteinase-mediated collagen degradation. Eur J Oral Sci 119: 79-85. Link: http://bit.ly/2JbLF1c
- Pashley DH (1992) Mechanistic analysis of fluid distribution across the pulpodentine complex. J Endod 18: 72-75. Link: http://bit.ly/2xuev6m
- Mitchem JC, Gronas DG (1991) Adhesion to dentine with and without smear layer under varying degrees of wetness. J Prosthetic Dent 66: 619-622. Link: http://bit.ly/2RUBi4G
- Hannig M, Friedrichs C (2001) Comparative in vivo and in vitro investigation of interfacial bond variability. Oper Dent 26: 3-11. Link: http://bit.ly/2Xqn1mh
- Pashley DH (1991) In vitro simulations of in vivo bonding conditions. Am J Dent 4: 237-240. Link: http://bit.ly/2G6HxxR
- Sirisha K, Rambabu T, Ravishankar Y, Ravikumar P (2014) Validity of bond strength tests: A critical review-Part II. J Conserv Dent 17: 420-426. Link: http://bit.ly/2JCQccj
- Sirisha K, Rambabu T, Ravishankar Y, Ravikumar P (2014) Validity of bond strength tests: A critical review-Part I. Journal of Conservatory Dentistry 17: 305-311. Link: http://bit.ly/2LxlaVI
- Gerhardt-Szep S, Werelius K, de Weerth F, Heidemann D, Weigl P (2012) Influence of femtosecond laser treatment on shear bond strength of composite resin bonding to human dentine under simulated pulpal pressure. J Biomed Mater Res 100: 177-184. Link: http://bit.ly/32aUQGM
- Szep S, Langner N, Bayer S, Börnichen D, Schulz C, et al. (2003) Comparison of microleakage on one composite etched with phosphoric acid or a combination of phosphoric and hydrofluoric acids and bonded with several different systems. J Prosthetic Dent 89: 161-169. Link: http://bit.ly/30hkMim
- Retief DH (1991) Standardizing laboratory adhesion tests. Am J Dent 4: 231-236. Link: http://bit.ly/2JvpHWa
- Perdigão J (2010) dentine bonding-variables related to the clinical situation and the substrate treatment. Dent Mater 26: e24-37. Link: http://bit.ly/30kgdUp
- Technical Specification ISO/TS 11405: 2015(E), Dentistry - Testing of adhesion to tooth structure. Geneva, Switzerland (2015)
- Jacobsen T, Söderholm KJ (1995) Some effects of water on dentine bonding. Dent Mater 11: 132-136. Link: http://bit.ly/2Jf1693
- Azad E, Atai M, Zandi M, Shokrollahi P, Solhi L (2018) Structure-properties relationships in dental adhesives: Effect of initiator, matrix monomer structure, and nano-filler incorporation. Dent Mater 34: 1263-1270. Link: http://bit.ly/2xvoMPN
- Montes MA, de Goes MF, da Cunha MR, Soares AB (2001) A morphological and tensile bond strength evaluation of an unfilled adhesive with low-viscosity composites and a filled adhesive in one and two coats. J Dent 29: 435-441. http://bit.ly/2xz9jOw
- Sengun A, Ozturk B, Ozer F (2003) The effect of simulated intrapulpal pressure on bond strength to enamel and dentine. J Oral Rehabil 30: 550-555. Link: http://bit.ly/329iJhM
- Besnault C, Attal JP (2001) Influence of a simulated oral environment on dentine bond strength of two adhesive systems. Am J Dent 14: 367-372. Link: http://bit.ly/30gdlYx
- Erhardt MC, Cavalcante LM, Pimenta LA (2004) Influence of phosphoric acid pretreatment on selfetching bond strengths. J Esthet Restor Dent 16: 33-40. Link: http://bit.ly/32bETjp
- Peumans M, De Munck J, Van Landuyt KL, Poitevin A, Lambrechts P, et al. (2010) Eightyear clinical evaluation of a 2-step self-etch adhesive with and without selective enamel etching. Dent Mater 26: 1176-1184. Link: http://bit.ly/2LHZ1nP
- Marchesi G, Frassetto A, Visintini E, Diolosà M, Turco G, et al. (2013) Influence of ageing on self-etch adhesives: one-step vs. two-step systems. Eur J Oral Sci 121: 43-49. Link: http://bit.ly/2xyh3Al
- van Dijken JW (2010) A prospective 8-year evaluation of a mild two-step self-etching adhesive and a heavily filled two-step Etch-and-rinse system in non-carious cervical lesions. Dent Mater 26: 940-946. Link: http://bit.ly/2Yy5WmR
- Masarwa N, Mohamed A, Abou-Rabii I, Abu Zaghlan R, Steier L (2016) Longevity of Self-etch dentine Bonding Adhesives Compared to Etch-and-rinse dentine Bonding Adhesives: A Systematic Review. J Evid Based Dent Pract 16: 96-106. Link: http://bit.ly/2LCjpX9
- Torii Y, Itou K, Nishitani Y, Ishikawa K, Suzuki K (2002) Effect of phosphoric acid etching prior to self-etching primer application on adhesion of resin composite to enamel and dentine. Am J Dent 15: 305-308. Link: http://bit.ly/2YtZFZv
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
