Journal of Clinical Research and Ophthalmology
Secukinumab Associated Choroidal Neovascularization
Edward B Miller1* and Oren S Yovel2
2Ophthalmology Department, Kaplan Medical Center, Rehovot, Israel
Cite this as
Miller EB, Yovel OS (2022) Secukinumab Associated Choroidal Neovascularization. J Clin Res Ophthalmol 9(1): 005-007. DOI: 10.17352/2455-1414.000095Copyright License
© 2022 Miller EB, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.A previously healthy 38-year-old woman with non-radiographic spondyloarthritis (nrSpA) developed juxta-foveal Choroidal Neovascularization (CNV) in the right eye three months after starting treatment with the anti-IL-17A inhibitor secukinumab. As there was no sign of ocular inflammation, no known association with CNV and this rheumatic disease, and other etiologic etiologies were excluded, this biologic agent was the suspected cause. Drug discontinuation and treatment with bevacizumab resulted in full suppression of the neovascular activity. IL-17 has a purported etiologic role in ocular neovascular diseases. The onset of this process in a patient receiving anti-IL-17 therapy suggests a possible causative role requiring further clarification.
Introduction
Choroidal Neovascularization (CNV) of the retina involves the growth of fibrovascular membrane from the choroid layer beneath the retina into the sensory retina accompanied by vision loss. It is the main lesion in active age macular degeneration and is uncommon in patients younger than age 50 [1]. The seronegative spondyloarthropathies are a family of rheumatic diseases characterized by sacroiliitis of which ankylosing spondylitis is the best- known. Patients with these disorders frequently suffer from concomitant uveitis, but choroidal neovascularization in these individuals is not typically seen [2]. We describe a previously healthy young woman with non-radiographic spondyloarthropathy (nrSpA) who developed unilateral CNV while receiving biologic therapy with secukinumab (anti-IL-17A). As no know association between secukinumab and CNV has been previously reported a possible association with this medication is discussed.
Case presentation
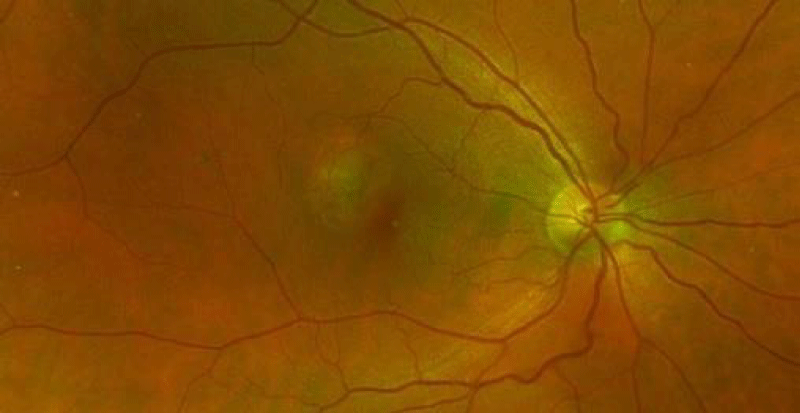
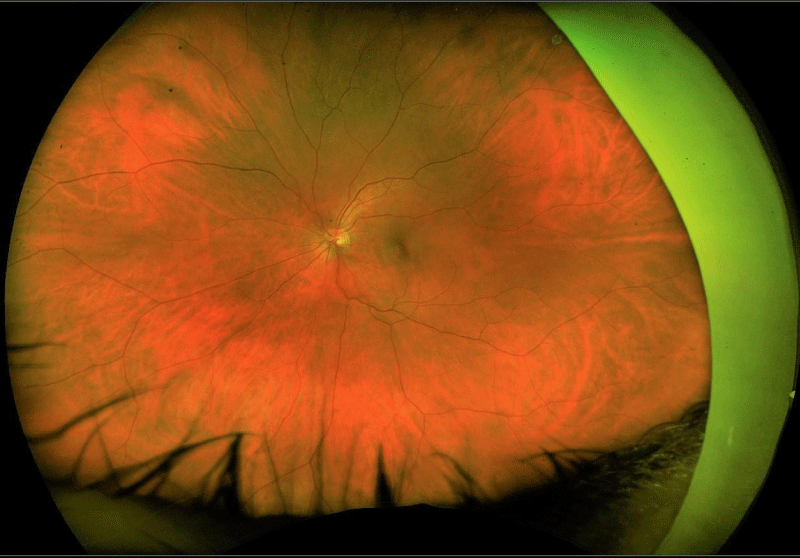
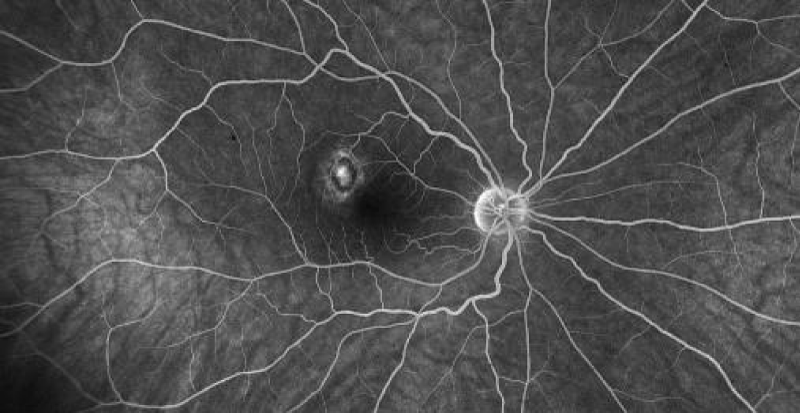
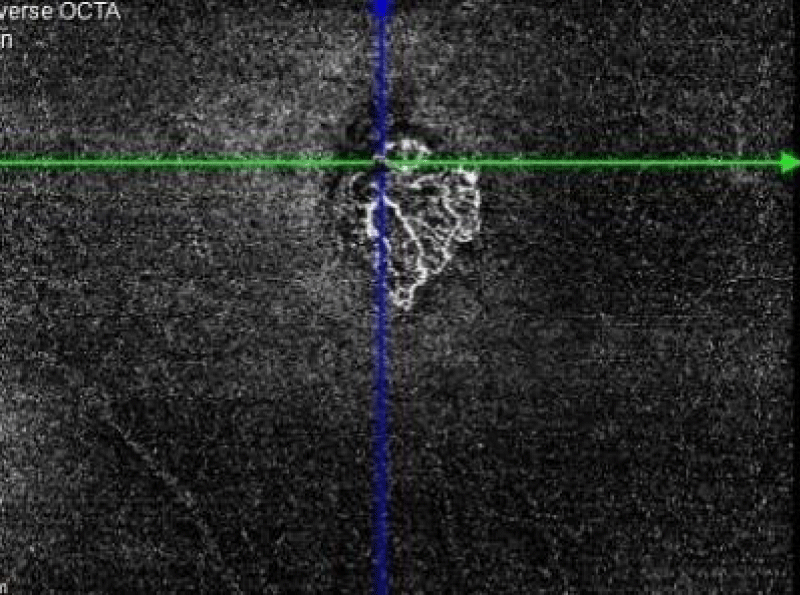
A previously healthy 38-year-old woman was diagnosed with non- radiographic spondyloarthritis (nrSpA) in March 2016 following a two-year history of low back pain, elevated inflammatory markers (ESR and CRP) and an MRI showing bilateral sacroiliitis. HLA B27 allele was not detected and additional serologic studies including rheumatoid factor and antinuclear antibody were negative Therapy with two anti-TNF biologic agents (golimumab, etanercept) were administered until October 2019 without significant benefit. At that time she has switched to secukinumab 150 mg monthly due to prior drug ineffectiveness. In June 2020 she presented to the ophthalmology department in our hospital with complaints of blurred vision. She had no systemic illnesses except the abovementioned secukinumab-treated nrSpA. She had no significant past ocular history, no previous ocular surgery, trauma, or bouts of inflammation and she had never used prescription drops. Her vision was 20/25 in her right eye, and 20/20 in the left with no significant refractive correction. Both anterior chambers were clear and quiet and ocular pressure was normal. Fundoscopy of the right eye revealed a small gray lesion in the temporal fovea (Figure 1A). There were no abnormal findings in the peripheral retina (Figure 1B) and no abnormal findings in the left eye (Figure 1C). Optical coherence tomography (OCT) scan (Figure 2A), OCT angiography (Figure 2B) and fluorescein angiography (Figure 2C) showed a juxta-foveal CNV in the right eye. An extensive review of the literature revealed no prior reported association between CNV and secukinumab, but due to the rarity of this disorder and the lack of any other systemic or ocular risk factors the drug was discontinued. The patient received 3 monthly injections of 1.25 mg bevacizumab with full suppression of the neovascular activity. She received an additional ocular injection at an interval of 2 and 3 months apart, but no reactivation of the CNV was noted and her ocular condition has been stable for more than a year without treatment. No new abnormalities have been found during the 14 months follow-up period.
Discussion
Ocular involvement in inflammatory rheumatic diseases is well known but CNV is very rarely seen. Similarly, anti-rheumatic drugs including newer biologic agents have not been typically reported in this clinical setting [3]. Secukinumab is a human IgG1 monoclonal antibody that selectively binds to the interleukin- 17A (IL-17A) cytokine and inhibits its interaction with the IL-17 receptor. IL-17A is a naturally occurring cytokine that is involved in normal inflammatory and immune responses. Secukinumab is indicated for the treatment of psoriasis, plaque psoriasis, and psoriatic arthritis, but is rarely associated with adverse reactions which most commonly include nasopharyngitis, diarrhea, and upper respiratory tract infections. Secukinumab, to our knowledge, has not been reported with this ocular disease process [4]. It is in this setting that our current case raises a number of interesting questions. First, this patient has no history or ocular signs of other known causative associations for CNV such as high myopia, previous ocular trauma, uveitis, or peripheral retinal findings (indicating subtle active disease). As a diagnosis of nrSpA would automatically raise the suspicion of an intra-ocular inflammation, we, as mentioned before, didn't find any evidence of this process. Although idiopathic cases of CNV have been reported in younger (< 50 years old) patients these are rare [5]. Secondly, IL-17’s proposed key role in this process [6,7]. And secukinumab’s inhibition of this cytokine, suggests a possible etiologic mechanism. Finally, IL-17 inhibition has been proposed as a possible inhibitory treatment for choroidal neovascularization, but in our patient, we propose a possible stimulatory role. While clarification must await further studies, physicians encountering these individuals should be aware of this possible association.
Patient Consent
Written consent to publish the case has been obtained. This report does not contain any personally identifying information.
Acknowledgment
This manuscript does not include any non-author contributors to acknowledge.
Statement of Ethics
This study protocol was reviewed and the need for approval was waived by the Kaplan Medical Center's IRB.
Authorship
Both authors attest that they meet the current ICMJE criteria for authorship contributed equally to the study and have approved the final a version of the manuscript.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author.
- Lee P, Wang CC, Adamis AP (1998) Ocular neovascularization: an epidemiologic review. Surv Opthalmol 43: 245-269.Link: https://bit.ly/3sT5Fws
- Murray PI, Rauz S (2016) The eye and inflammatory rheumatic diseases: the eye and rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis. Best Pract Res Clin Rheumatol 30: 802-825. Link: https://bit.ly/3MznhVU
- Castillejo Becerra M, Ding Y, Kenol B, Hendershot A, Meara AS (2020) Ocular side effects of antirheumatic medications: a qualitative review. BMJ Open Opthalmol 5: e000331. Link: https://bit.ly/3pReEfL
- Deodar A, Mease PJ, McInnes IB, Baraliakos X, Reich K, et al. (2019) Long-term safety of secukinumab in patients with moderate-to-severe plaque psoriasis, psoriatic arthritis and ankylosing spondylitis: integrated pooled clinical trial and post-marketing surveillance data. Arth Res Ther 21: 111. Link: https://bit.ly/3vNs543
- Cohen SY, Laroche A, Leguen Y, Soubrane G, Coscas GJ (1996) Etiology of Choroidal neovascularization in young patients. Opthalmology 103: 1241-1244. Link: https://bit.ly/35LgDeF
- Li Y, Zhou Y (2019) Interleukin-17: the role for pathological angiogenesis in ocular neovascular diseases. Tohoku J Exp Med 247: 87-98. Link: https://bit.ly/3MzQhwT
- Byrne EM, Llorián-Salvador M, Tang M, Margariti A, Chen M, et al. (2021) IL-17A Damages the Blood–Retinal Barrier through Activating the Janus Kinase 1 Pathway. Biomedicines 9: 831. Link: https://bit.ly/3MtqrdQ

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
