Journal of Cardiovascular Medicine and Cardiology
In Silico modeling of immune-cardiovascular-endocrine interactions
Kristen A Windoloski1, Johnny T Ottesen2 and Mette S Olufsen1*
2Centre for Mathematical Modeling, Human Health and Disease and Department of Science and Environment, Roskilde University, Roskilde, Denmark
Cite this as
Windoloski KA, Ottesen JT, Olufsen MS. (2022) In Silico modeling of immune-cardiovascular-endocrine interactions. J Cardiovasc Med Cardiol 9(4): 037-041. DOI: 10.17352/2455-2976.000186Copyright License
© 2022 Windoloski KA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.The immune system provides an intricate, balanced response to combat the effects of inflammatory stimuli. It incorporates both positive and negative feedback from multiple physiological systems such as the cardiovascular and endocrine systems including mechanisms functioning on a variety of time scales. They have been studied individually via scientific experiments and using mathematical modeling. However, more analysis is needed to study the interactions between these three systems during an inflammatory event. We present the first dynamical systems model studying immune, cardiovascular and endocrine responses to a 2 ng/kg bolus dose of endotoxin. The model is calibrated to experimental data from two endotoxin challenge studies and we use this model to investigate the effects of endotoxin dosage, administration timing and administration method. Our model shows that most repercussions of endotoxin administration clear the system within 24 hours, but effects can linger for up to 72 hours.
Introduction
It is well-known that the immune system, the cardiovascular system and the endocrine system interact during an immune event that incites an inflammatory response, such as an infection or injury [1-9] These three complex systems function on varying time scales, ranging from seconds to days, which affects the progression and resolution of inflammation and resulting symptoms accordingly. While numerous experimental in vitro and in vivo studies have been conducted to study the inflammatory response and its bi-directional communication with the cardiovascular and endocrine systems [2,7,10-12] in silico experiments have the capability to further investigate dynamics between systems, challenge the included concepts and mechanisms and extrapolate the model to study clinically relevant scenarios not captured in the traditional experimental setting. Because of the abundance of data from in vitro and in vivo studies, in silico experiments can make more accurate predictions by using the data to calibrate and validate the model. The use of mathematical modeling to improve our knowledge surrounding the inflammatory response is critical to expedite diagnosis, develop and test treatment regimes and decrease mortality associated with inflammatory diseases such as sepsis.
Methods
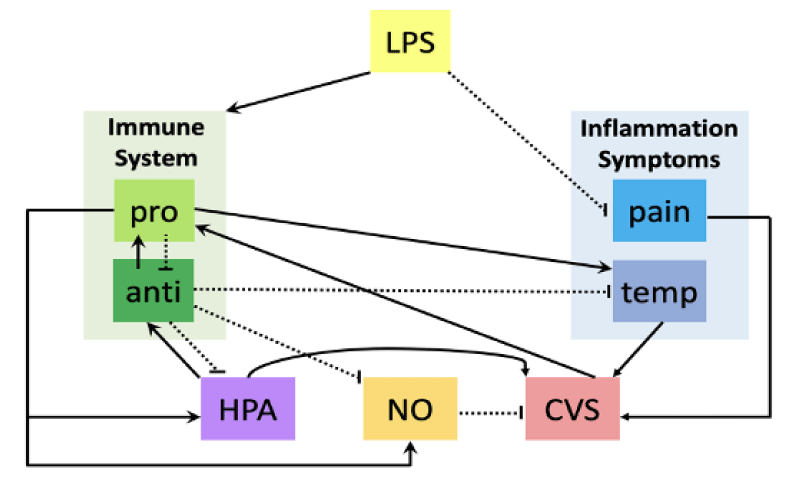
Here, we discuss our recent results of a mathematical model coupling immune, cardiovascular, and endocrine dynamics across various time scales during an endotoxin (lipopolysaccharide, LPS) challenge [13]. To study the effect of time scales on dynamics, we utilize our model to simulate variations in endotoxin dosage, administration timing, and administration method. We also gain insight into relevant but understudied experimental setups investigating inflammatory diseases, which are of interest in the clinical environment. Our dynamical systems model relating immune, cardiovascular, and endocrine interactions consists of 18 state variables, and over 100 parameters, and is composed of three submodels. The model includes 17 ordinary differential equations and one delay differential equation. Individual model subsystems were derived in previous work [14-16], but the combined immune-cardiovascular-endocrine model is the first model examining the dynamics between these three subsystems integrating various time scales. The model schematic is shown in Figure 1.
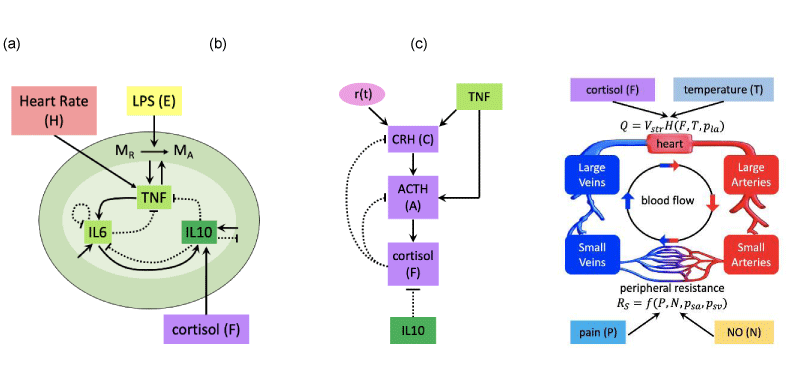
The first submodel (Figure 2a) details the immune system’s response to endotoxin. The model predicts time-varying concentrations of the endotoxin (lipopolysaccharide, LPS), resting and activated monocytes and pro and anti-inflammatory cytokines (TNF-, IL-6 and IL-10). The immune subsystem contains major elements integral to the activation of the innate immune system and thus, the development of inflammation. Primary symptoms of inflammation at the site of injury or infection include heat and pain [7]. The whole-body inflammatory response to endotoxin incites fever and activation of pain receptors [17]. Therefore, body temperature and pain components are also included in the model. The second subsystem (Figure 2b) describes cardiovascular dynamics. We use a four-compartment model of systemic circulation to describe changes in volume, pressure and flow. We also model cardiovascular control mechanisms such as vascular resistance, heart rate, blood pressure and nitric oxide. Finally, the third submodel (Figure 2c) consists of tracking concentrations of hormones released in the hypothalamic-pituitary-adrenal (HPA) axis. These include corticotropin-releasing hormone (CRH), adrenocorticotropic hormone (ACTH) and cortisol. Stimulation of the hypothalamus both from the body’s natural circadian rhythm as well as the immune system prompts the start of this hormone cascade [6,14]. There is extensive evidence of communication between these three submodels during an inflammatory event [1-9]. These couplings in the model are highlighted in Figures 1,2.
The mathematical model described above was calibrated to mean experimental data from two separate studies, both administering 2 ng/kg of LPS to male volunteers. We used cytokine, temperature, and hormonal data from Clodi, et al. and cardiovascular and pain data from Janum et al. to calibrate the model [18,19]. Parameters used in [14-16] for the original individual submodels were scaled and recalibrated to fit the experimental data. New parameters corresponding to couplings between the three systems were added sequentially into the model and were manually adjusted.
Results and discussion
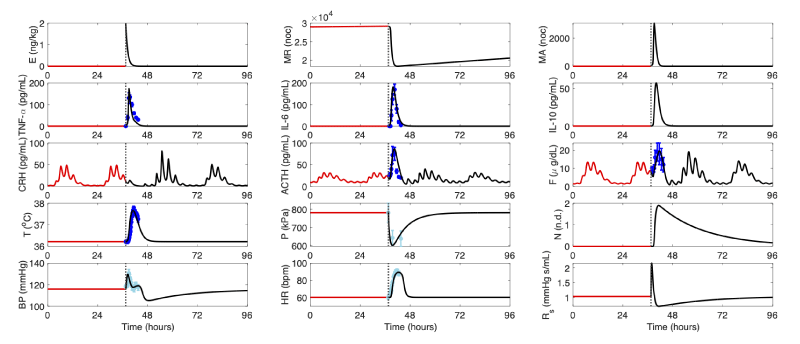
The initial model calibration (Figure 3) shows the model output for the bolus administration of 2 ng/kg of LPS into the system at t =37.5 hours (corresponding to 1:30 pm). The model follows the time series data from [18,19] by exhibiting an initial reduction in pain threshold and an increase in cytokine, cardiovascular, hormonal and thermal states followed by a decrease in blood pressure and resistance below their initial values. While immune components return to baseline within six hours, other model states had longer recovery times. Hormonal status, under the circadian and ultradian influence, took at least 24 hours to recover and thermal, cardiovascular and pain states took between 10 and 72 hours to fully recover from an endotoxin insult.
Following model calibration, we performed several endotoxin challenge experiments in silico such as varying LPS administration time, the total bolus dose administered and the method of administration. The first scenario showed minor effects on immune, cardiovascular, temporal and pain states relative to Figure 3. Administration timing did, however, impact hormone dynamics. When endotoxin was administered in the early morning or late evening, CRH exhibited larger concentration spikes and all three HPA states showed increased ultradian oscillations following endotoxin exposure. Hormone states returned to baseline after approximately 24 hours while recovery of other model states followed that in Figure 3.
The latter two in silico experiments we performed are clinically pertinent. Since endotoxin doses must be safe for participants, the LPS threshold is low [20] which prevents more realistic scenarios of inflammation seen in disease from being experimentally studied in vivo. Here, we increased the bolus dose administered from the original 2 ng/kg to 4, 8 and 16 ng/kg. The immune response intensified as the dose increased by activating at a faster rate and producing much larger concentrations of IL-6 and IL-10. A lower pain threshold, a slightly higher fever and a drop in blood pressure approaching hypotension also appeared as the LPS dose was elevated. In the HPA axis, initial hormone release was suppressed and oscillations were dampened in subsequent 24-hour cycles as the LPS dose increased.
The method of endotoxin administration is also a pertinent topic within the scientific community. Repeated endotoxin dose studies simulate the recurrence of infection following an initial insult and have been done experimentally [21-23] and in silico [14,24]. Our in silico repeated dose study showed that immune, temporal, pain and heart rate states are exacerbated when the second dose is administered at or near peak levels from the first dose (within 6 hours) but suppressed when the second dose is administered after recovery. Like these dynamics, blood pressure drops close to hypotension when the second dose is given at or just after peak levels but narrowly deviates from baseline if given after recovery. Effects on hormone release are seen up to 48 hours after the first dose, including increased or damped oscillations and peaks.
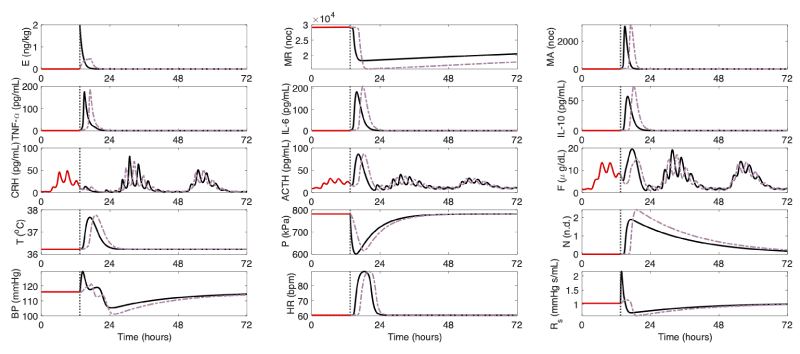
Another variation of endotoxin dose administration is a continuous infusion. While a bolus dose of LPS is the commonly accepted endotoxin challenge form, it is recognized that a continuous infusion of endotoxin over several hours better portrays immunostimulant exposure in inflammatory diseases [13,17,25,26]. Therefore, we use our model to simulate a 2 ng/kg continuous infusion of endotoxin over a 4-hour period (Figure 4). As seen in data conducted from in vivo experiments [27], our model shows a delay in the activation of the immune system. This delay then cascades to delay cardiovascular, hormonal, temporal and pain reactions. Once activated, the system exhibits larger peaks in immune, temporal and nitric oxide states. A more extreme cardiovascular response is seen, too, where blood pressure drops to about 100mmHg nearing hypotension. Hormone release was moderately suppressed and oscillation frequency was somewhat increased in the following 24-hour cycle. For a more in-depth look at the results regarding in silico experiments conducted using our model, we refer to our publication [13].
Here, we presented a complex, dynamic systems model studying coupled immune, cardiovascular and endocrine dynamics during an endotoxin challenge. We incorporated various time scales into the model including both circadian and ultradian rhythms and studied endotoxin variations in administration timing, dose and method. The novelty of our model is that it is the first model to investigate dynamics between the immune, cardiovascular and endocrine systems in response to endotoxin up to several 24-hour cycles following endotoxin administration. The strength of the couplings between the systems is of great interest in gaining an understanding of how the submodels interact. Our work builds the foundation for further investigation into these couplings, including mathematically rigorous sensitivity analysis and parameter estimation schemes to determine which parameters have the greatest influence on system dynamics. Because of the lack of knowledge associated with the coupling parameter values, it would also be appropriate to formally quantify the uncertainty of these parameters.
- Bartekova M, Radosinska J, Jelemensky M, Dhalla NS. Role of cytokines and inflammation in heart function during health and disease. Heart Fail Rev. 2018 Sep;23(5):733-758. doi: 10.1007/s10741-018-9716-x. PMID: 29862462.
- Burford NG, Webster NA, Cruz-Topete D. Hypothalamic-Pituitary-Adrenal Axis Modulation of Glucocorticoids in the Cardiovascular System. Int J Mol Sci. 2017 Oct 16;18(10):2150. doi: 10.3390/ijms18102150. PMID: 29035323; PMCID: PMC5666832.
- John CD, Buckingham JC. Cytokines: regulation of the hypothalamo-pituitary-adrenocortical axis. Curr Opin Pharmacol. 2003 Feb;3(1):78-84. doi: 10.1016/s1471-4892(02)00009-7. PMID: 12550746.
- Mulla A, Buckingham JC. Regulation of the hypothalamo-pituitary-adrenal axis by cytokines. Baillieres Best Pract Res Clin Endocrinol Metab. 1999 Dec;13(4):503-21. doi: 10.1053/beem.1999.0041. PMID: 10903811.
- Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N Engl J Med. 2005 Oct 20;353(16):1711-23. doi: 10.1056/NEJMra050541. PMID: 16236742.
- Silverman MN, Pearce BD, Biron CA, Miller AH. Immune modulation of the hypothalamic-pituitary-adrenal (HPA) axis during viral infection. Viral Immunol. 2005;18(1):41-78. doi: 10.1089/vim.2005.18.41. PMID: 15802953; PMCID: PMC1224723.
- Tracey KJ. The inflammatory reflex - Introduction. Nature. 2002 420: 853–859.https://doi.org/10.1111/j.1365-2796.2004.01440.x
- Turrin NP, Plata-Salamán CR. Cytokine-cytokine interactions and the brain. Brain Res Bull. 2000 Jan 1;51(1):3-9. doi: 10.1016/s0361-9230(99)00203-8. PMID: 10654575.
- Verma V, Sheikh Z, Ahmed AS. Nociception and role of immune system in pain. Acta Neurol Belg. 2015 Sep;115(3):213-20. doi: 10.1007/s13760-014-0411-y. Epub 2014 Dec 30. PMID: 25547878.
- Agelaki S, Tsatsanis C, Gravanis A, Margioris AN. Corticotropin-releasing hormone augments proinflammatory cytokine production from macrophages in vitro and in lipopolysaccharide-induced endotoxin shock in mice. Infect Immun. 2002 Nov;70(11):6068-74. doi: 10.1128/IAI.70.11.6068-6074.2002. PMID: 12379683; PMCID: PMC130344.
- Morimoto K, Morimoto A, Nakamori T, Tan N, Minagawa T, Murakami N. Cardiovascular responses induced in free-moving rats by immune cytokines. J Physiol. 1992 Mar;448:307-20. doi: 10.1113/jphysiol.1992.sp019043. PMID: 1593469; PMCID: PMC1176201.
- van der Poll T, Barber AE, Coyle SM, Lowry SF. Hypercortisolemia increases plasma interleukin-10 concentrations during human endotoxemia--a clinical research center study. J Clin Endocrinol Metab. 1996 Oct;81(10):3604-6. doi: 10.1210/jcem.81.10.8855809. PMID: 8855809.
- Windoloski KA, Bangsgaard EO, Dobreva A, Ottesen JT, Olufsen MS. A unified model for the human response to endotoxin challenges. Preprint on arXiv [q-bio.TO], http://arxiv.org/abs/2210.05712.
- Bangsgaard EO, Hjorth PG, Olufsen MS, Mehlsen J, Ottesen JT. Integrated Inflammatory Stress (ITIS) Model. Bull Math Biol. 2017 Jul;79(7):1487-1509. doi: 10.1007/s11538-017-0293-2. Epub 2017 Jun 22. PMID: 28643132.
- Brady R, Frank-Ito DO, Tran HT, Janum S, Møller K, Brix S, Ottesen JT, Mehlsen J, Olufsen MS. Personalized mathematical model of endotoxin-induced inflammatory responses in young men and associated changes in heart rate variability. Math Model Nat Phenom. 2018; 13(5): 1–21. https://doi.org/10.1051/mmnp/2018031
- Dobreva A, Brady-Nicholls R, Larripa K, Puelz C, Mehlsen J, Olufsen MS. A physiological model of the inflammatory-thermal-pain-cardiovascular interactions during an endotoxin challenge. J Physiol. 2021 Mar;599(5):1459-1485. doi: 10.1113/JP280883. Epub 2021 Feb 11. PMID: 33450068.
- Bahador M, Cross AS. From therapy to experimental model: a hundred years of endotoxin administration to human subjects. J Endotoxin Res. 2007;13(5):251-79. doi: 10.1177/0968051907085986. PMID: 17986486.
- Clodi M, Vila G, Geyeregger R, Riedl M, Stulnig TM, Struck J, Luger TA, Luger A. Oxytocin alleviates the neuroendocrine and cytokine response to bacterial endotoxin in healthy men. Am J Physiol Endocrinol Metab. 2008 Sep;295(3):E686-91. doi: 10.1152/ajpendo.90263.2008. Epub 2008 Jul 1. PMID: 18593851.
- Janum S, Nielsen ST, Werner MU, Mehlsen J, Kehlet H, Møller K. Pain perception in healthy volunteers: effect of repeated exposure to experimental systemic inflammation. Innate Immun. 2016 Oct;22(7):546-56. doi: 10.1177/1753425916663638. Epub 2016 Aug 22. PMID: 27554053.
- Hack CE, Aarden LA, Thijs LG. Role of cytokines in sepsis. Adv Immunol. 1997;66:101-95. doi: 10.1016/s0065-2776(08)60597-0. PMID: 9328641.
- Beeson PB. Tolerance to bacterial pyrogens. J Exp Med. 1947 86: 29–38.https://doi.org/10.1084/jem.86.1.29
- Cross AS. Endotoxin tolerance-current concepts in historical perspective. J Endotoxin Res. 2002;8(2):83-98. doi: 10.1179/096805102125000227. PMID: 12028748.
- Rittig N, Thomsen HH, Bach E, Jørgensen JO, Møller N. Hormone and Cytokine Responses to Repeated Endotoxin Exposures-No Evidence of Endotoxin Tolerance After 5 Weeks in Humans. Shock. 2015 Jul;44(1):32-5. doi: 10.1097/SHK.0000000000000384. PMID: 25895146.
- Day J, Rubin J, Vodovotz Y, Chow CC, Reynolds A, Clermont G. A reduced mathematical model of the acute inflammatory response II. Capturing scenarios of repeated endotoxin administration. J Theor Biol. 2006 Sep 7;242(1):237-56. doi: 10.1016/j.jtbi.2006.02.015. Epub 2006 Apr 17. PMID: 16616206.
- Kiers D, Koch RM, Hamers L, Gerretsen J, Thijs EJ, van Ede L, Riksen NP, Kox M, Pickkers P. Characterization of a model of systemic inflammation in humans in vivo elicited by continuous infusion of endotoxin. Sci Rep. 2017 Jan 5;7:40149. doi: 10.1038/srep40149. PMID: 28054645; PMCID: PMC5215288.
- Parker SJ, Watkins PE. Experimental models of gram-negative sepsis. Br J Surg. 2001 Jan;88(1):22-30. doi: 10.1046/j.1365-2168.2001.01632.x. PMID: 11136305.
- Taudorf S, Krabbe KS, Berg RM, Pedersen BK, Møller K. Human models of low-grade inflammation: bolus versus continuous infusion of endotoxin. Clin Vaccine Immunol. 2007 Mar;14(3):250-5. doi: 10.1128/CVI.00380-06. Epub 2007 Jan 31. PMID: 17267590; PMCID: PMC1828854.

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
