Journal of Cardiovascular Medicine and Cardiology
FMTVDM Study Proposal: WID2H - Breast Trial. (Weight, Inflammation, Diet v Drug, Heart disease) – Breast Trial
Richard M Fleming1*, Matthew R Fleming1 and William C Dooley2
2Oklahoma University Health Science Center, Oklahoma City, Oklahoma, USA
Cite this as
Fleming RM, Fleming MR, Dooley WC (2020) FMTVDM Study Proposal: WID2H - Breast Trial . J Cardiovasc Med Cardiol 7(2): 072-078. DOI: 10.17352/2455-2976.000116Introduction
As we enter this new decade, medicine is presented with the daunting task of resolving incongruities in our existing data and the ever-increasing incidence of obesity, Coronary Artery Disease (CAD) and cancer – including but not limited to breast cancer. The consequence of our failure to quantitatively measure the impact of diets and drugs at the tissue level where the actual CAD and cancer are occurring has limited our ability to better define the dietary, lifestyle and medication changes required to limit and potentially reverse these inflammatory diseases.
This paper proposes a study which will answer these fundamental questions-a study which readers of this journal are all to familiar with the need to complete. A study, which will provide the evidence we and our patients have been looking for; defining which dietary practices are detrimental and which are clearly beneficial and it will do so quantitatively-removing any doubt about the true impact of these diets and any potential subjective interpretation of outcomes.
Inflammatory coronary artery disease [1].
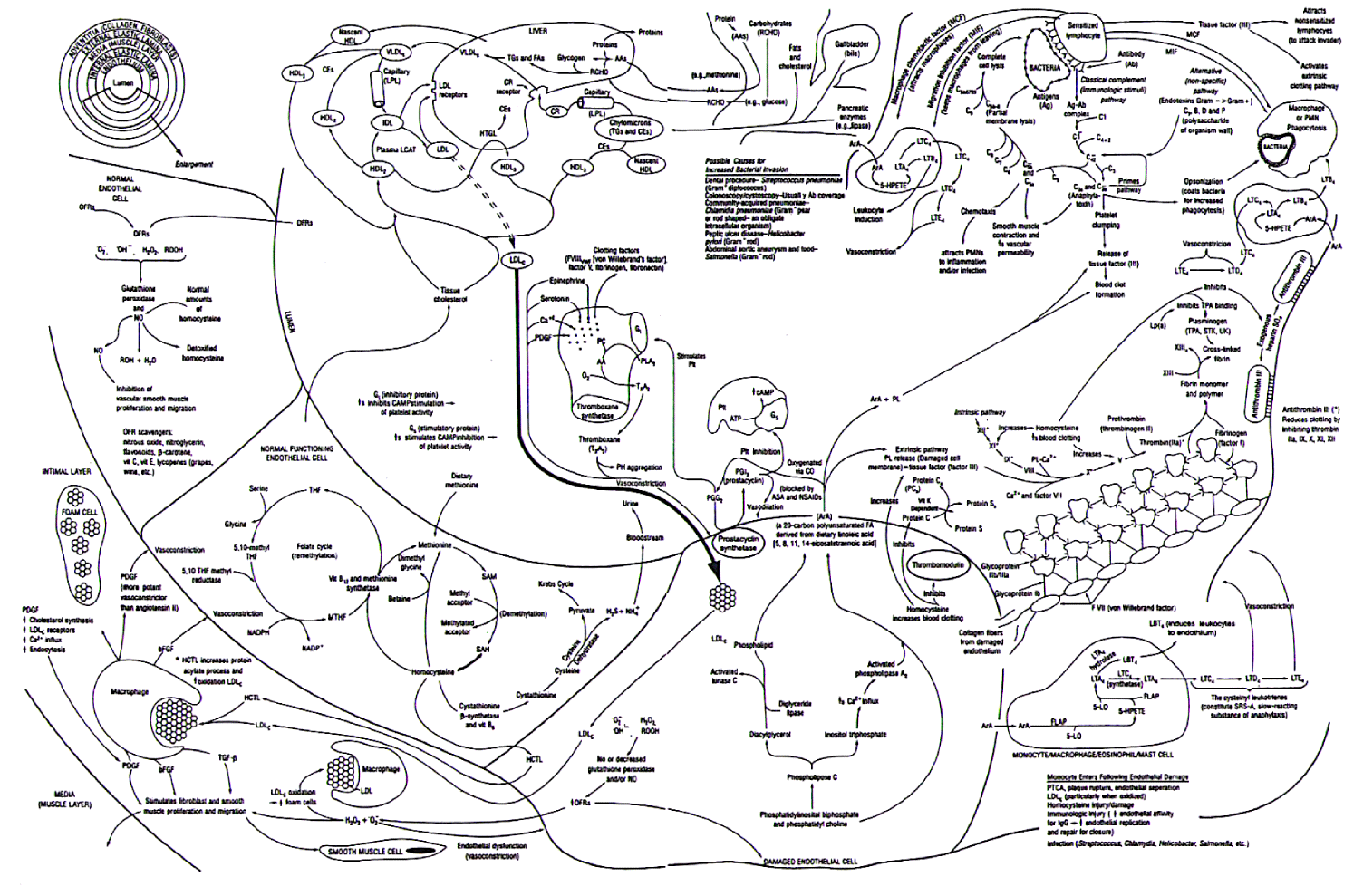
It is now commonly accepted that the Fleming Unified Theory of Vascular Disease (FUTVD) [2-4], shown in Figure 1, explains the mechanisms behind the inflammatory process that is associated with the impaired physiologic response we know as Coronary Artery Disease (CAD), resulting in Angina [5] and the Major Adverse Cardiac Events (MACE) of Myocardial Infarction (MI), ischemic cardiac failure, cerebrovascular disease (CVA, RIND and TIA), claudication and cancers.
Coronary artery disease is the result of this inflammatory change within the walls of the arteries, impairing the ability of the coronary arteries to regionally deliver the amount of blood required by the heart at any moment in time [6-8]. It is this inability of the coronary arteries to relax and increase blood flow, producing regional blood flow differences-resulting in angina [5] - which we must be able to measure to accurately diagnose CAD, and it is the change in this ability over time, which we must measure, to determine if CAD is progressing, regressing or stabilizing-either naturally or as the result of a treatment process-be that dietary, medical or otherwise.
For decades it has been presumed that measureable blood (serum markers) tests associated within the various components promulgating this inflammatory proces (lipids, homocysteine, lipoprotein (a), fibrinogen, c-reactive protein, interleukin-6, and potential infectious agents), would provide useful information as to the progression, regression or stabilization of this inflammatory process.
However, these blood tests do not represent what is happening at the tissue level-the true site of the inflammatory process-raising the question as to the relationship between the serum (blood) levels of these inflammatory components and the tissue level of these inflammatory components.
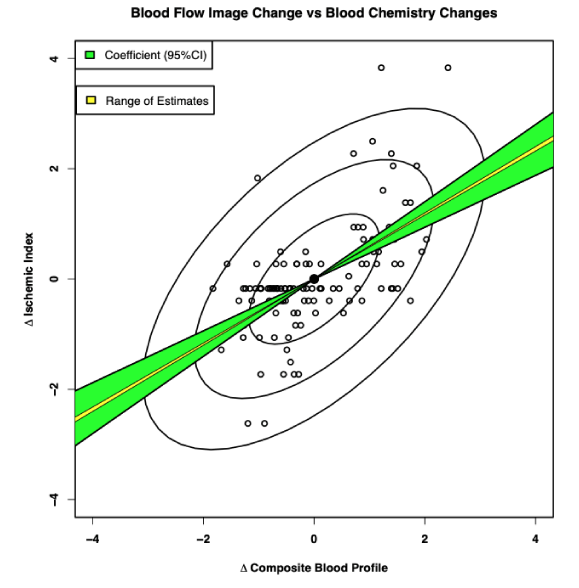
As shown in Figure 2, we have known for more than a decade, that measured changes in these serum blood tests (biomarkers) considered responsible for promoting inflammatory processes does not correlate with measured changes in the actual physiologic CAD [9], yet we have continued to focus on these blood tests with their poor correlations to CAD [10], rather than measuring true CAD itself [11-18].
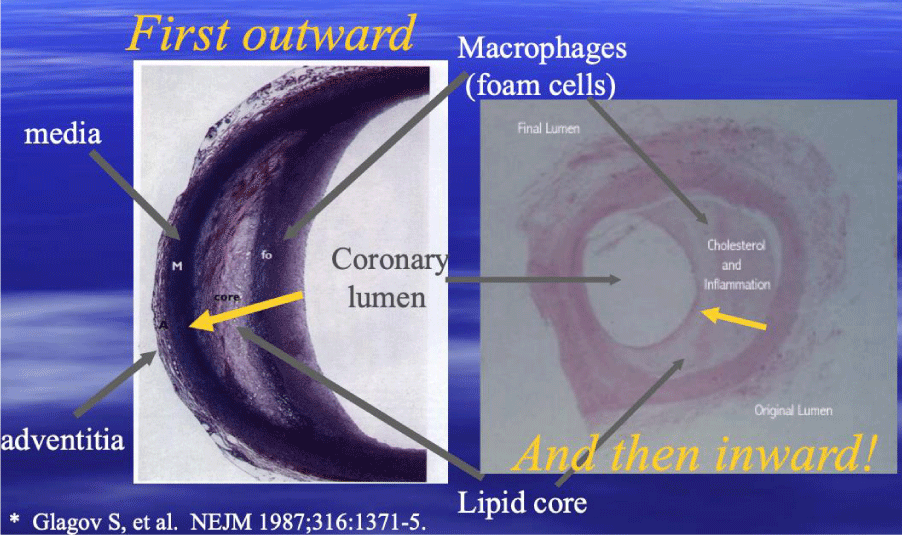
Given our improved understooding, that CAD is caused by an inflammatory buildup occuring within the walls of the arteries (Figures 3,4) resulting in impared regional blood flow (Figure 5) and only later narrowing the coronary lumen [19], the limitations in using cardiac catheterizations to measure changes in CAD, is readily apparent – coupled with the addition of other errors made with coronary arteriography [20-22].
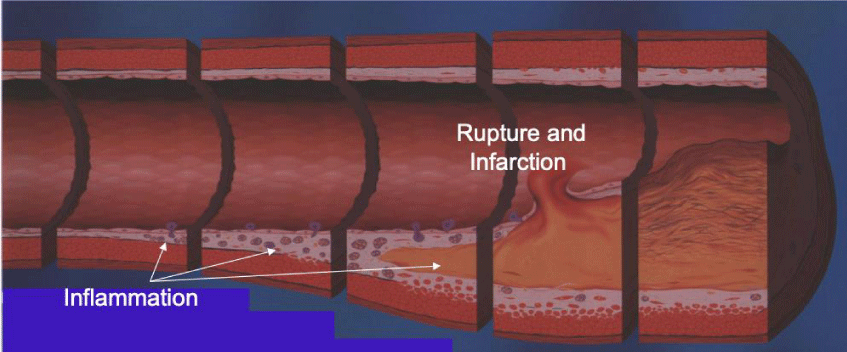
Consequently, coronary lumen narrowing-percent Diameter Stenosis (%DS)-occurs later in the inflammatory process, with 70% of all MIs occuring with less than 30% lumen narrowing. Interpretation using this approach to finding CAD, has resulted in errors in reading %DS, as well as the inability to determine the actual amount of inflammation (CAD) within the walls of the arteries.
Vulnerable inflammatory CAD-resulting from inflammatory damage to the wall of the coronary arteries-thus is first associated with an impairment in the ability of the arteries to dilate (relax) upon demand, to meet the increased regional blood supply required to meet cardiac demands/workload. It is now possible to quantify these changes using FMTVDM-the first utility patent capable of doing so [8,12-18].
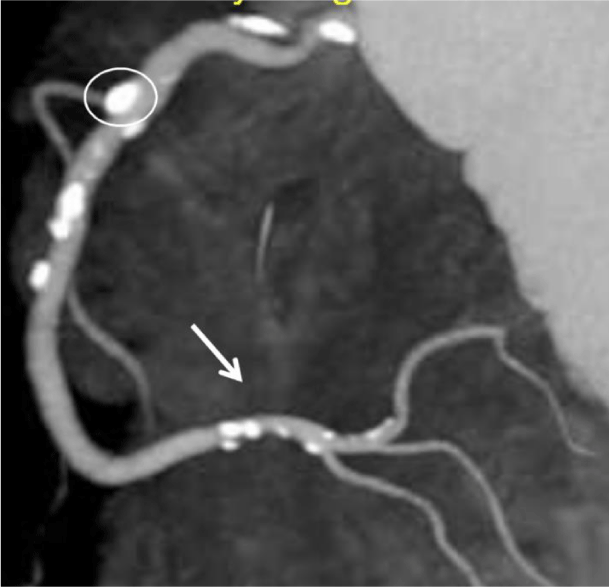
The process of inflammatory athero-sclerotic CAD, may be associated with calcium buildup (sclerotic) or not (atheroma)-depending upon the individual. As Glagov established [19], a considerable plaque buildup is required (Figure 3) before coronary lumen narrowing can be appreciated and this need not be associated with calcification. In fact, when plaques are visualized with Intravascular Ultrasound (IVUS), clinicians frequently differentiate plaques based upon visual appearance. Those lesions without sclerotic or calcific changes appear yellow in color and are more vulnerable to sudden rupture, while those with sclerosis and/or calcification have a grey or white appearance and are more stable-less likely to rupture. The presence of elevated CAC scores may provide insight into which lesions are less likely to suddenly rupture-providing insight into a subset of lesions-however, there is no information provided about lesions without calcium and no information in the literature supporting CAC use to measure regression of CAD.
Clearly once the calcium is present (Figure 6) there is demonstrable anatomic evidence of CAD; however, the use of coronary artery calcium scores (CCT/CAC) cannot be depended upon to (a) accurately diagnose non-sclerotic CAD, (b) actually measure the physiologic impact of CAD, or (c) to provide a true measure of the physiologic impact of CAD present.
Considering (1) the overall cost of CAD including the associated MACEs, (2) the lack of correlations associated with changes in blood tests and CAD, (3) the inability to accurately measure CAD using either coronary arteriography or CCT (aka CAC) scoring, (4) the tremendous costs associated with lipid reducing agents-which address only some but not all of the causes of inflammatory CAD, and (5) the ability of FMTVDM to quantitatively measure the physiologic effect of inflammatory CAD, this study is designed to determine what if any effect, lipid lowering medications, dietary and lifestyle changes have on inflammatory CAD.
By measuring the functional (physiologic) effect of cholesterol lowering medications and/or dietary modifications designed to alter the inflammatory factors resulting in CAD, we can determine the impact of both dietary recommendations and cholesterol lowering medications at both the serum and tissue level – where the inflammatory changes have a direct effect.
Breast Disease
In 2019, it was estimated that 1.8 million Americans would be diagnosed with cancer, including 268,600 women and 2,670 men with breast cancer, the most commonly diagnosed type of cancer [23].
For decades the medical sciences have focused on the ability to find cancers, by our ability to find something abnormal, either by (A) a visual qualitative imaging test (x-rays like mammograms [24,25], CTs [26], MRI [27], etc.), or by (B) an abnormally elevated blood test-which does not actually tell us if cancer is present any more than a calcium deposit does-resulting most likely from an increased protein or molecular compound being released from cells at higher than expected levels and thus measured at higher than normal levels in the blood.
We have also come to depend upon (C) biomarkers (e.g. BRCA1/2) suggestive of the potential for cellular and tissue change-but not determinant of change. Finally, we have come to depend upon our ability to diagnose cancer based upon our ability to find something (D) microscopically-including biomarkers (HER2, ER+, PR+) and abnormal cellular details as noted above. However, even this gold standard requires human qualitative interpretation with associated errors [28-30].
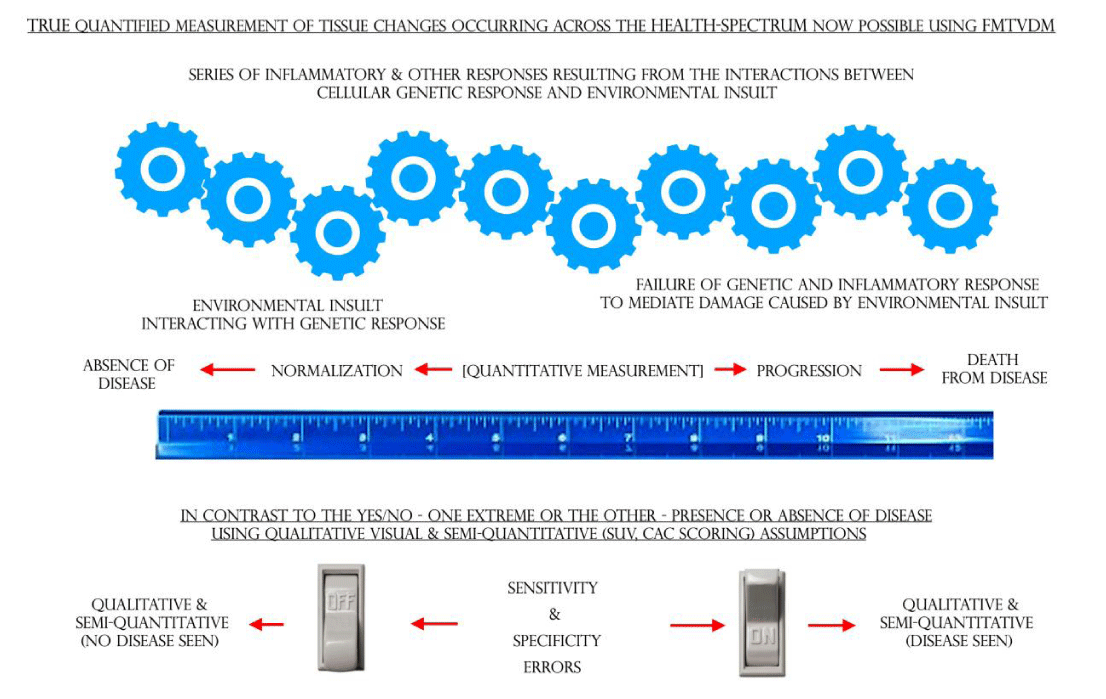
The changes that occur as normal cells respond to environmental insults resulting in a series of possible transformative changes, which may or may not result in cancer, can be measured from beginning to end as shown in Figure 7 [30-34].
Like inflammatory CAD, the transitional changes from “normal” breast tissue through the inflammatory changes into cancerous tissue, can be quantitatively measured (FMTVDM) using the same methods used to quantify physiologic inflammatory CAD.
Like CAD, measuring the consequential changes in regional blood flow and metabolism, that occur either naturally or as the result of treatment, is now possible using FMTVDM.
Rather than thinking about patients as a collection of diseases, a model promulgated by DRG and current procedural terminology (CPT) codes, the physician goal, pursuant to the Hippocratic Oath and the Declaration of Geneva, is to think in terms of the health of the patient [35]-viz. the patient’s health-spectrum.
As explained in detail elsewhere [34], the transitional changes associated with the potential development of cancer is the result of (A) enhanced genetic metabolic activity, which results in (1) the release of chemical mediators responding to the environmental factors-both positive and negative-and (2) augmentation of regional blood flow [36-39].
Nuclear imaging provides the only physiologic tool available for medical imaging of the body. As such, nuclear imaging provides the only true, real time, in vivo method for analyzing physiologic changes occurring within the patient.
While prior nuclear imaging procedures addressed qualitative imaging controls, they did not take into account quantitative imaging controls, leaving the interpretation of diagnostic studies flawed and without the accuracy, consistency and reliability required to (A) measure transitional changes in tissue or (B) the ability to measure the true impact of treatment upon cancer or other diseases such as coronary artery disease [40]. This has only recently been made possible [12,32].
Cancer has now become the number one killer of people worldwide. This fact alone should emphasize the importance of how we approach this global health problem. Cancer is no longer limited to industrialized countries, the poor or even the affluent. Cancer knows no geographic or socioeconomic barriers.
It is impossible to intelligently approach cancer from an absence of knowledge or limited approaches to finding, monitoring and determining the treatment response of a cancer. That which makes cancer, cancer, and that, which makes transitional changes in cells/tissue different from cancer and normal tissue, is what we must be able to measure if we are to address the health risk associated with the number one cause of morbidity and mortality worldwide. We cannot address cancer or its treatment, without the ability to measure these changes [41-44].
Study Design
The study will include: Initial and Final FMTVDM measurements of both the Heart and Breast.
Serum blood tests routinely used to compare with FMTVDM outcomes, and Dietary Treatments.
Treatments
While there are differing opinions, most agree that dietary and lifestyle factors play a MAJOR contributing role in the development and progression of inflammatory CAD. Absent a prior quantitative study measuring the physiologic outcome (FMTVDM), there is no clear consenses as to which dietary and lifestyle factors are most influential.
Prior studies conducted by the Principle Investigator have demonstrated that for three different treatment arms, a study of 120 individuals is sufficient to demonstrate statistical differences between groups. Thus, the proposed study would include 40 individuals per treatment arm of the study. These prior studies have also demonstrated statistical differences after 1-year of treatment.
To maximize the benefits of each treatment arm, sites will be selected, where the investigators (a) agree as to the specifics of the treatment approach and (b) do not deviate from the proposed protocol.
Participants should include men and women 21 plus years of age, non-pregnant, on no vitamins, minerals or medications. Non-smokers (including cigarettes, cigars, pipes, marijuana, vaping products, etc.), with investigator agreed to limits on alcohol consumption. They must agree not to become pregnant during the study.
Proposed Treatment Arms (n=40 per arm) of the study
1) Dietary and lifestyle.
A) Low Carbohydrate
B) Vegan
C) American Heart Association, or
D) another as of yet unidentified diet
2) Lipid Lowering medications.
A) Statin #1
B) Statin #2
C)PCSK9-Inhibitor
If further groups are added (e.g. a diet and drug combination, a diet and vitamin combination, etc.), this will become an independent “arm” of the study with an “n” of 40 participants.
Measured outcomes.
1) FMTVDM (Heart & Breast).
A) As this is an absolute quantifiable value, and cannot be altered, it will become the final deteminant of the physiologic impact of treatment upon inflammatory CAD.
B) Measurement will be at the beginning and end of the treatment period.
If participants drop out prior to completion of the 1-year of treatment, FMTVDM will be obtained at the time of drop out AND at the completion of 1-year-as this reflects real world outcomes.
2) Simultaneous Breast Cancer-Disease Imaging will be acquired per FMTVDM.
Measured Surrogate Blood Tests and Weight.
1) Measured at entry, 6-weeks, 3-mo Association nths, 6-months, 9-months - exit and 1-year from beginning of study.
The quantification of changes in both coronary artery disease and breast disease as measured by FMTVDM, will provide answers to the fundamental questions surrounding the impact of current dietary practices and medical regimens.
This knowledge will allow us to scientifically make recommendations to patients based upon measured outcomes and become the foundation for further research and understanding of individuals with heart disease and cancer.
FMTVDM is issued to first author. First author, authored the “Inflammation and Heart Disease” and “Angina” Theories.
- Inflammation and Heart Disease; Fleming Unified Theory of Vascular Disease. 1-655833842. Started 9-1-2011. Effective 9-16-2011, TX 7-451-244.
- Fleming RM (1999) Chapter 64. The Pathogenesis of Vascular Disease. Textbook of Angiology. John C. Chang Editor, Springer-Verlag New York, NY 787-798. Link: https://bit.ly/3bC2iiF
- Fleming RM (2000) The Fleming Unified Theory of Vascular Disease: A Link Between Atherosclerosis, Inflammation, and Bacterially Aggravated Atherosclerosis (BAA). Angiol 51: 87-89. Link: https://bit.ly/2VpZy1Y
- Fleming RM, Boyd L, Forster M (2000) Reversing Heart Disease in the New Millennium - The Fleming Unified Theory. Angiology 51: 617-629. Link: https://bit.ly/2XR6mHF
- Fleming RM (2003) Angina and coronary Ischemia are the result of coronary regional Blood Flow Differences. J Amer Coll Angiol 1:127-142.
- Stenosis Flow Reserve (SFR), Coronary Flow Reserve (CFR); Quadratic Coronary Flow Reserve (QCFR). 1-655833951. Started 9-1-2011.
- Fleming RM (1999) Chapter 29. Atherosclerosis: Understanding the relationship between coronary artery disease and stenosis flow reserve. Textbook of Angiology. John C. Chang Editor, Springer-Verlag, New York, NY 381-387.
- Fleming RM, Fleming MR, Chaudhuri TK (2019) True AI Implementation Through FMTVDM Proprietary Equations and QCA. Biomed J Sci Tech Res 20: 15154-15160. Link: https://bit.ly/2zjoaRx
- Fleming RM, Harrington GM (2008) "What is the Relationship between Myocardial Perfusion Imaging and Coronary Artery Disease Risk Factors and Markers of Inflammation?" Angiology 59: 16-25. Link: https://bit.ly/3ezuzYS
- Matthew R (2000) Storks Deliver Babies (p=0.008). Teaching Statistics 22: 36-38. Link: https://bit.ly/3eDqwuz
- Fleming RM (1999) Chapter 31. Nuclear Cardiology: Its Role in the Detection and Management of Coronary Artery Disease Textbook of Angiology. John C. Chang Editor, Springer-Verlag New York, NY 397-406.
- The Fleming Method for Tissue and Vascular Differentiation and Metabolism (FMTVDM) using same state single or sequential quantification comparisons. Patent Number 9566037.
- Fleming RM, Fleming MR, McKusick A, Chaudhuri TK (2018) Semi-quantification limitations: FMTVDM©℗ demonstrates quantified tumor response to treatment with both regional blood flow and metabolic changes. J Nucl Med 59: 1643-1644. Link: https://bit.ly/2VndyJK
- Fleming RM, Fleming MR, McKusick A, Chaudhuri T (2018) FMTVDM-TFM©℗: True Quantification requires Standardization of the tool being used to Measure, with a Known, Unchanging Standard to produce accurate, consistent and reproducible Quantified Measurements. J Nucl Card 26. Link: https://bit.ly/34PjFK8
- Fleming RM, Fleming MR, Chaudhuri TK (2019) True AI Implementation Through FMTVDM Proprietary Equations and QCA. Biomed J Sci Tech Res 20: 15154-15160. Link: https://bit.ly/3eAA3CB
- Fleming RM, Fleming MR, Harrington G, McKusick A, Chaudhuri T (2018) USVAH Study demonstrates statistically significant improvement in diagnosis and care of U.S. Veterans using FMTVDM-FHRWW ©℗ “Quantitative” Nuclear Imaging. The era of truly quantitative stress-first, stress-only imaging has begun! J Nucl Med Radiat Ther 9: 006. Link: https://bit.ly/2KnHHCG
- Fleming RM, Fleming MR, McKusick A, Chaudhuri T (2018) Multi Center Clinical Trial Confirms FMTVDM©℗ MPI in Seven Modern Clinical Laboratories in the U.S.A. and Asia. Artificial Intelligence (AI) with True Quantification. J Nucl Med Radiat Ther 9: 372. Link: https://bit.ly/34TY0Ah
- Fleming RM, Fleming MR, Chaudhuri TK, McKusick A (2019) FMTVDM provides first patented Quantitative Method to accurately Measure both Heart Disease and Breast Cancer on the “Health-Spectrum”. J Cardiovasc Med Cardiol 6: 019-020. Link: https://bit.ly/2VqddG8
- Glagov s, Weisenberg E, Zarins CK, Stankunavicius R, Kolettis GJ (1987) Compensatory Enlargement of Human Atherosclerotic Coronary Arteries. N Engl J Med 316: 1371-1375. Link: https://bit.ly/2yx2ZuC
- Fleming RM, Kirkeeide RL, Smalling RW, Gould KL (1991) Patterns in Visual Interpretation of Coronary Arteriograms as Detected by Quantitative Coronary Arteriography. J Am Coll Cardiol 18: 945- 951. Link: https://bit.ly/2VJzQEL
- Fleming RM (2000) Shortcomings of coronary angiography. Letter to the Editor. Cleve Clin J Med 67: 450.
- Fleming RM (2001) Coronary Artery Disease is More than Just Coronary Lumen Disease. Amer J Card 88: 599-600. Link: https://bit.ly/34QdXYg
- Cancer Stat Facts: Common Cancer Sites. Link: https://bit.ly/2Vmf3YO
- Ekpo EU, Alakhras M, Brennan P (2018) Errors in Mammography Cannot be Solved Through Technology Alone. Asian Pac J Cancer Prev 19: 291-301. Link: https://bit.ly/2RTGDdP
- Miller AB, Wall C, Baines CJ, Sun P, To T, Narod SA. et al. (2014) Twenty-five-year follow-up for breast cancer incidence and mortality of the Canadian National Breast Screening Study: randomized screening trial. BMJ 348: g366. Link: https://bit.ly/3avYn5C
- Krishna S, Murray CA, McInnes MD, Chatelain R, Siddaiah M, et al. (2017) CT imaging of solid renal masses: pitfalls and solutions. Clin Radiol 72: 708-721. Link: https://bit.ly/2VnYrjb
- Rominger M, Berg D, Frauenfelder T, Ramaswamy A, Timmesfeld N, et al. (2016) Which factors influence MRI-pathology concordance of tumour size measurements in breast cancer? Eur Radiol 26: 1457-1465. Link: https://bit.ly/2VG8uiG
- Raab SS, Grzybicki DM, Janosky JE, Zarbo RJ, Meier FA, et al. (2005) Clinical Impact and Frequency of Anatomic Pathology Errors in Cancer Diagnosis. Cancer 104: 2205-2213. Link: https://bit.ly/3eF2UpB
- Raab SS, Grzybicki DM (2010) Quality in cancer diagnosis. CA Cancer J Clin 60: 139-165. Link: https://bit.ly/3aqd3Dh
- Tosteson ANA, Yang Q, Nelson HD, Longton G, Soneji SS, et al. (2018) Second opinion strategies in breast pathology: a decision analysis addressing over-treatment, under-treatment and care costs. Breast Cancer Res Treat 167: 195-203. Link: https://bit.ly/34R51C5
- Tucker I (2018) AI cancer detectors. Link: https://bit.ly/2VpxOe3
- Fleming RM, Dooley WC, Chaudhuri TK (2017) The Development of FMTVDM-BEST Imaging: The Answer for Breast Cancer. Breast Enhanced Scintigraphy Test (BEST): Quantifying the Detection of Breast Cancer and its Treatment. J Nucl Med Radiat Ther 8: 350. Link: https://bit.ly/3eEmuCg
- Sheikh A (2018) Evolution of Quantification in Clinical Nuclear Medicine: A Brief Overview of Salient Uses and Upcoming Trends. J Nucl Med Radiat Ther 9: 375. Link: https://bit.ly/2VrrFOu
- Fleming RM, Fleming MR, Dooley WC, Chaudhuri TK (2020) The Importance of Differentiating Between Qualitative, Semi-Quantitative and Quantitative Imaging – Close Only Counts in Horseshoes. Eur J Nucl Med Mol Imaging 47: 753-755. Link: https://bit.ly/3an6Xn6
- Fleming RM, Fleming MR, Chaudhuri TK, McKusick A (2019) Ethics in Medicine – The Hippocratic Oath! Acta Scientific Med Sci.
- Wright GL, Maroulakou IG, Eldridge J, Liby TL, Sridharan V, et al. (2008) VEGT stimulation of mitochondrial biogenesis: requirement of AKT3 kinase. FASEB J 22: 3264-3275. Link: https://bit.ly/2xBG8OL
- Siemerink MJ, Klaassen I, Vogels IMC, Griffioen AW, Van Noorden CJ, et al. (2012) CD34 marks angiogenic tip cells in human endothelial cell cultures. Angiogenesis 15: 151-163. Link: https://bit.ly/2wUSXTT
- Senchukova MA, Nikitenko NV, Tomchuk ON, Zaitsev NV, Stadnikov AA (2015) Different types of tumor vessels in breast cancer: morphology and clinical value. Springerplus 4: 512. Link: https://bit.ly/2RQfF6M
- Luengo-Gil G, Gonzalez-Billalabeitia E, Perez-Henarejos SA, Navarro Manzano E, Chaves-Benito A, et al. (2018) Angiogenic role of miR-20a in breast cancer. PLoS ONE 13: e0194638. Link: https://bit.ly/2KlhNPR
- Fleming RM, Fleming MR, Chaudhuri TK, McKusick A, Dooley WC (2019) Nuclear Imaging: Physician Confusion Over True Quantification and Isotope Redistribution. J Clin Cases Rep 3: 32-42. Link: https://bit.ly/3cx6gcc
- Fleming RM, Dooley WC (2002) Breast Enhanced Scintigraphy Testing (B.E.S.T.) Distinguishes Between Normal, Inflammatory Breast Changes and Breast Cancer. A Prospective Analysis and Comparison with Mammography. Integr Cancer Ther 1: 238-245. Link: https://bit.ly/2wTCkb1
- Fleming RM, Fleming MR, Chaudhuri TK, Dooley WC, McKusick A (2019) Letter to the Editor: A response to Hruska’s case study on molecular breast imaging and the need for true tissue quantification. Breast Cancer Res 21: 15. Link: https://bit.ly/2RUZapZ
- Fleming RM, Fleming MR, Chaudhuri TK, McKusick A, Dooley WC (2019) Statistical demonstration that FMTVDM is superior to mammography. Ann Clin Radiol 2: 1012. Link: https://bit.ly/3cuYBLm
- Fleming RM, Fleming MR (2019) Flawed evidence underpins approval of new cancer drugs. BMJ 366: l5399. Link: https://bit.ly/2XO0E9v

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
