Journal of Biology and Medicine
Flowable placental connective tissue matrices for tendon repair: A review
Nicole M Protzman1, Yong Mao2, Raja Sivalenka3, Desiree Long3, Anna Gosiewska3*, Robert J Hariri3 and Stephen A Brigido3
2Laboratory for Biomaterials Research, Department of Chemistry and Chemical Biology, Rutgers University, 145 Bevier Rd., Piscataway, New Jersey 08854, USA
3Vice President Research & Development, Degenerative Diseases, Celularity Inc., 170 Park Ave., Florham Park, NJ 07932, USA
Cite this as
Protzman NM, Mao Y, Sivalenka R, Long D, Gosiewska A, et al. (2022) Flowable placental connective tissue matrices for tendon repair: A review. J Biol Med 6(1): 010-020. DOI: 10.17352/jbm.000030Copyright License
© 2022 Protzman NM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Tendon injuries are associated with considerable pain and disability. Owing to the hypovascularity and hypocellularity of the tissue, natural tendon healing is slow and ineffective. Traditional conservative and surgical treatment options fail to address the underlying pathology. As a result, the healed tendon is mechanically incompetent and prone to degeneration and rupture. Therefore, new biological methods have been suggested to enhance tendon repair and regeneration. Flowable Placental Connective Tissue Matrices (FP-CTMs) represent a promising means to promote tendon healing. Like non-flowable placental scaffolds, FP-CTMs possess the innate healing properties of the placenta and provide structural and biochemical extracellular matrix components. Unlike their non-flowable counterparts, FP-CTMs have the added benefits of minimal invasiveness and the capacity to fill irregular spaces. FP-CTMs can enhance tendon repair by providing a three-dimensional extracellular matrix for cellular attachment and proliferation while decreasing inflammation and limiting adhesion formation. The present report reviews tendon biology, pathology, healing, and current treatment modalities, followed by a comprehensive literature review, evaluating the clinical application of FP-CTMs for tendon repair. Recent research suggests that the use of FP-CTMs in tendon repair is safe and efficacious and further indicates that FP-CTMs can modulate the tendon repair environment and improve clinical outcomes. However, the existing clinical evidence is limited to retrospective case series with no control group. Therefore, additional work must be performed to better understand the clinical applications and therapeutic benefits of FP-CTM in tendon repair compared with conventional treatments.
- The hypocellularity and hypovascularity of tendons make the healing process slow and ineffective.
- Current conservative and surgical treatments fail to address the underlying pathology, and as a result, the healed tendon is mechanically incompetent and prone to degeneration and rupture.
- Flowable Placental Connective Tissue Matrices (FP-CTMs) may enhance tendon repair by providing a three-dimensional extracellular matrix for cellular attachment and proliferation while decreasing inflammation and limiting adhesion formation.
- Although FP-CTMs represent a distinct group of scaffolds, each commercial product is sourced from various placental tissues and subjected to different processing procedures, impacting the biological and mechanical properties of the tissue, which may impact tenocyte-CTM interactions and clinical outcomes.
- Available evidence shows that the use of FP-CTMs for tendon repair is safe and efficacious and has further demonstrated significant improvements in pain and function. However, the clinical research is limited to retrospective case series. Therefore, additional research is needed to evaluate the appropriate clinical applications and associated outcomes.
Abbreviations
AATB, American Association of Tissue Banks; A-CTM, AmnioFill® Connective Tissue Matrix; ADLs, Activities of Daily Living; AM, Amniotic Membrane; B-CTM, BioRenew® Connective Tissue Matrix; bFGF, basic Fibroblast Growth Factor; DNA, Deoxyribonucleic Acid; ECM, Extracellular Matrix; FP-CTM, Flowable Placental Connective Tissue Matrix; GAGs, Glycosaminoglycans; HCT/P, Human Cell, Tissue, or Cellular or Tissue-based Product; I-CTM, Interfyl® Connective Tissue Matrix; IL-6, InterLeukin-6; IL-1β, Interleukin 1 beta; LE, Lower Extremity; LOE, Level Of Evidence; NSAIDs, Non-Steroidal Anti-Inflammatory Drugs (NSAIDs); NRS, Numerical Rating Scale; PDGF, Platelet-Derived Growth Factor; SMR2T™, Selective Membrane of Reparative and Reconstructive Tissue; TGF-β, Transforming Growth Factor-Beta; µdHACM, micronized dehydrated Human Amnion/Chorion Membrane; VAS, Visual Analog Scale; VEGF, Vascular Endothelial Growth Factor.
Introduction/background
Tendon disorders have become increasingly common and account for a substantial portion of musculoskeletal injuries [1-4]. The number of people affected by tendinopathies and subsequent tendon tears is rapidly increasing [5]. This is attributed to the combined effects of population growth and aging as well as increased participation in extreme/competitive sports. The magnitude of treating these injuries presents a major clinical and financial burden to modern medicine [6].
After the injury, the mechanical competence of the native tendon is never restored. The healed tissue is burdened by the formation of adhesions, which disrupt the Extracellular Matrix (ECM) and increase the risk of further degeneration and rupture [7]. Even after surgical repair, the tendon is predisposed to re-rupture [8]. Thus, researchers and clinicians are interested in new methods to augment tendon repair. While exogenous scaffolds have shown promise [9-11], Flowable Placental Connective Tissue Matrices (FP-CTMs) have gained increasing interest [12]. Like non-flowable scaffolds, FP-CTMs provide structural and biochemical ECM components. In contrast with their non-flowable counterparts, FP-CTMs offer the added benefits of minimal invasiveness and the capacity to fill irregular spaces. Clinicians are considering the use of FP-CTMs for the treatment of tendinopathy/tendinitis [13-15] as well as the surgical repair and reconstruction of periarticular soft tissues [12].
Within, tendon biology, pathology, healing, and current treatment modalities are presented, followed by a review of the clinical application of FP-CTMs for tendon repair. A comprehensive literature search was performed to identify the available clinical evidence. Specifically, the PubMed database was queried for the terms: “placental tissue matrix,” “micronized,” “human,” and “tendon.” Inclusion criteria included studies reporting on the use of FP-CTMs for tendon repair, clinical outcomes, and human subjects. Exclusion criteria included animal data, basic science studies, review articles, and non-English language literature. A date range was not defined. This review uniquely demonstrates the gaps in the literature as well as potential directions for future work relative to the application of FT-CTMs for tendon repair.
Tendon structure and function
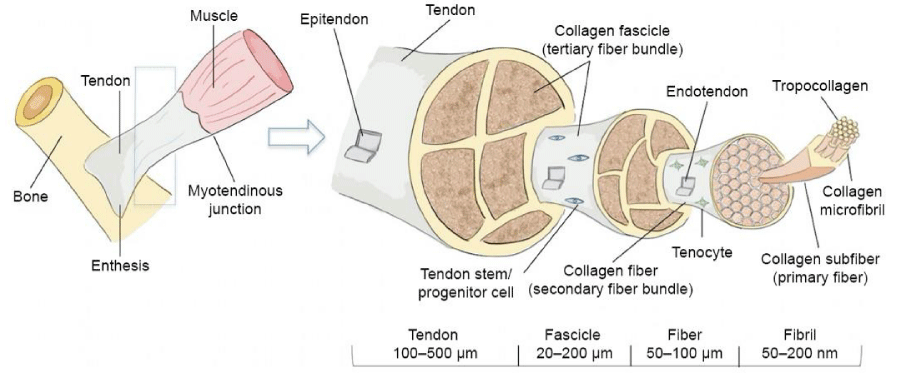
Tendons are comprised of dense fibrous connective tissue, which connects muscle to bone. The interface where the tendon attaches to the muscle is known as the myotendinous junction, and the interface where it attaches to the bone is known as the osteotendinous junction or enthesis (Figure 1). The primary function of a tendon is to transmit forces produced by a muscular contraction to the skeletal system to enable movement. Tendons have higher tensile strength compared with muscle, which enables them to withstand significant amounts of tension and protect the muscle from external forces [16].
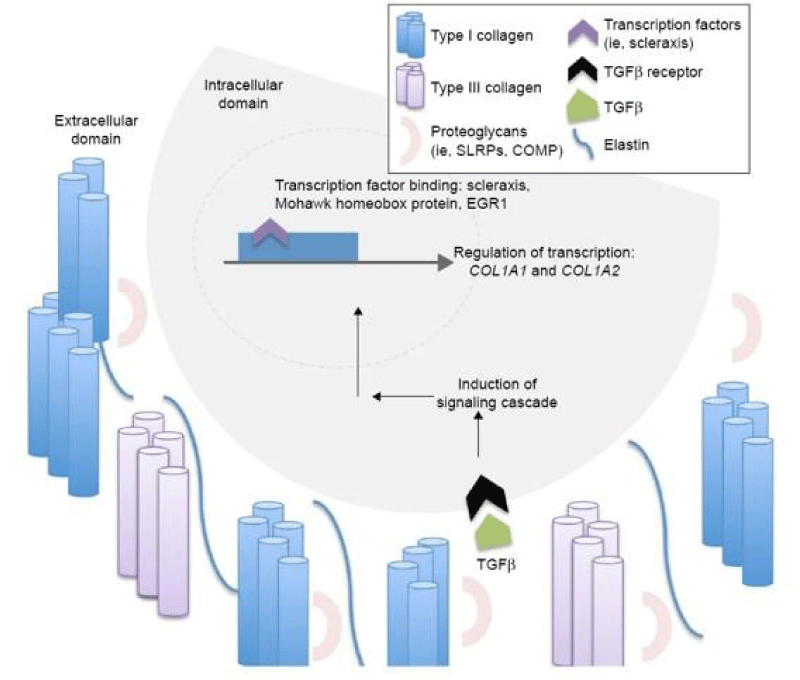
The ECM of tendons is composed of collagen, elastin, proteoglycans, and glycoproteins (Figure 2) [17]. Collagen is the most abundant molecule in the ECM, accounting for 60% to 85% of the dry weight of the tissue [18]. Type I collagen fibers are organized along the long-axis of the tissue, which affords tendons excellent uniaxial mechanical strength [19], although there are small amounts of type II collagens in the epitenonium/endotenonium and type III collagens in the fibrocartilaginous areas of the osteotendinous junction [16]. The organization of the tendon is hierarchical; collagen fibrils join together to form collagen fibers, which join together to form bundles, the bundles join together to form fascicles, and finally, the fascicles join together to form the whole tendon (Figure 1). The epitenon and endotenon are connective tissue sheaths, which permit smooth movements against adjacent structures and provide blood vessels, lymphatics, and nerves. The epitenon surrounds each tendon, and the endotenon encloses fibers. The elastic fibers contain elastin, which is responsible for the extensibility of the tendon [20]. Interspersed throughout the collagen hierarchy, there are non-collagenous matrix components, commonly grouped into proteoglycans, glycoproteins, and glycoconjugates [19]. Proteoglycans are responsible for the viscoelastic properties of the tendon. There are two predominant proteoglycans in tendons: decorin, a member of the small leucine-rich proteoglycan (SLRP) family accounting for approximately 80% of the total proteoglycan content in tendons, and versican, a large aggregating proteoglycan [21]. Various glycoproteins have been identified in tendon tissue, including collagen oligomeric matrix component (COMP) [22], tenascin-C [23], and tenomodulin [23].
There are two specialized fibroblast cells in tendon tissue: tenoblasts and tenocytes. They comprise 90% to 95% of the cells within the tendon. Chondrocytes, synovial cells, and vascular cells make up the remaining 5% to 10% [24]. Tenoblasts are immature tendon cells with an ovoid shape. As they mature, they transform into tenocytes with an elongated spindle shape [16]. Tenocytes are responsible for maintaining and synthesizing the components of the ECM and are intimately involved in tendon repair. Tenocytes respond to mechanical loading through the modulation of the ECM [25]. Loading, therefore, is essential to tendon homeostasis, but can also readily promote remodeling or degeneration [26]. Exposure to elevated mechanical stresses can place tendon tissues at risk of damage, and overloading is widely considered a causative factor in the onset of tendon injuries [26].
Tendon pathology
Tendon injuries are common disorders of the musculoskeletal system that are associated with considerable pain and disability, affecting both the athletic and general populations [27-31]. Tendon injuries often occur secondary to overuse, traumatic injury, or intrinsic age-related degeneration. Tendon injuries can be classified into one of two groups: acute or chronic. Acute injuries occur instantaneously, whereas chronic injuries develop over time.
The terminology and definitions relating to tendon injury are ever-changing [32]. Generally, the term “tendinopathy” describes a broad spectrum of tendon pathologies that are associated with pain, swelling, and impaired function [33-35]. It is often used to describe a chronic tendon injury in the absence of a partial or complete tear. The term “tendinitis” describes tendon pathology that has an inflammatory component [32]. The term “tendinosis” describes a tendon with impaired tendon healing, devoid of inflammatory cells. Tendon “tears” or “ruptures” refer to the separation of the tendon from the tissue to which it is attached. Spontaneous tendon tears occur without prior symptoms.
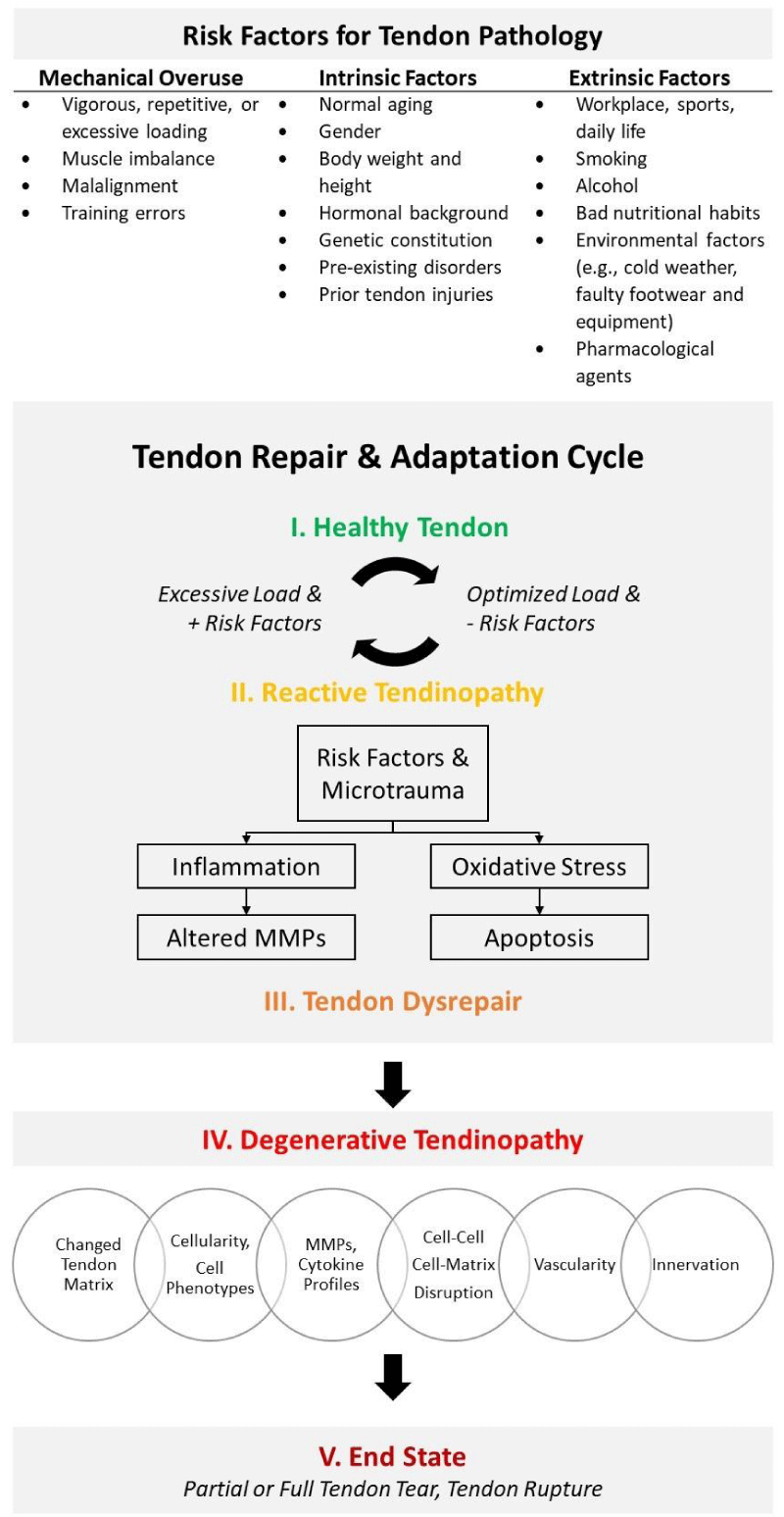
Tendinopathy has a multifactorial etiology, arising from biological and lifestyle-related factors as well as the use of pharmacologic agents. Like others, Steinmann and colleagues [36] grouped risk factors for chronic tendon pathology into three buckets: 1) mechanical overuse; 2) intrinsic factors (i.e., acting from within the body); and 3) extrinsic factors (i.e., acting on the body) (Figure 3). The onset of tendinopathy is often associated with a mechanical event, such as overuse or overloading. However, there is variation in how much load an individual can endure before developing tendinopathy. Likewise, the treatments required for recovery vary by individual. Identifying and understanding the risk factors may assist in understanding the progression of this multifaceted disease.
In a 2009 review, Cook and Purdam introduced a continuum model of tendinopathy [37]. This model suggests that in response to an excessive load (i.e., volume, intensity, frequency), tendons enter a pathologic continuum that consists of three continuous stages: 1) reactive tendinopathy; 2) tendon disrepair (i.e., failed to heal); and 3) degenerative tendinopathy. According to Cook and Purdam, adding or removing load moves the tendon forward or backward along the continuum. Building on the work of Cook and Purdam, Steinmann and colleagues [36] suggest that risk factors can act as triggers, predisposing the tendon to injury and impairing proper tendon healing. In their model, a tendon in the early reactive tendinopathy stage still possesses the capacity for healing. However, as risk factors increase, the tendon enters a state of disrepair, and eventually, tendon degeneration occurs. The tendon undergoes structural and compositional changes with alterations in biology and biomechanics. Cell-to-cell and cell-to-matrix communication are distorted, disrupting the organization of the ECM, making the tendon prone to further injury and tendon rupture [36]. The rate of progression through the various stages of tendinopathy is unknown and is likely influenced by various risk factors, such as age, gender, biomechanics, activity level, and nutritional habits [36].
Incidence of tendon tears
Although there are almost 4,000 tendons in the human body, certain tendons are more prone to rupture than others (Table 1).
The rotator cuff experiences more tendon tears than any other tendon in the human body [38]. Rotator cuff tears are multifactorial in origin, often a combination of age-related and degenerative changes from micro- and macro-trauma [39]. For example, 13% of individuals in their fifties [40] and 50% of individuals in their eighties experience a rotator cuff tear [41]. The Achilles tendon, the largest and strongest tendon in the human body, is the most common tendon to rupture in the lower extremity [42]. Rupture of the Achilles tendon most commonly affects middle-aged men, who participate in sports [43-45]. The third most common group of tendons to rupture are the flexor and extensor tendons of the hand and wrist. Injury to the flexor and extensor tendons of the hand and wrist most commonly occurs in males between the ages of 20-29 years of age. Twenty-five percent of these injuries are work-related, most commonly occurring in construction and extraction occupations [46].
Tendon healing
The natural process of tendon healing is slow and ineffective. The hypovascular and hypocellular nature of tendons limits their intrinsic capacity for healing [47]. Existing degenerative pathology and repetitive injury cause excessive inflammation, further impairing the healing process [48]. Even when a tendon successfully progresses through the healing cascade, the result is a mechanically and histologically inferior tendon compared with its native counterpart [11].
Tendon healing is controlled by tenocytes and their surrounding ECM [50] and is orchestrated through three multifaceted, overlapping stages: inflammatory, proliferative, and remodeling [49] (Table 2).
The initial inflammatory stage begins with a vascular and cellular response to injury. Vascular permeability increases, followed by an influx of red blood cells, white blood cells, and platelets. The invading inflammatory cells secrete a variety of cytokines and growth factors throughout the healing cascade. Initially, proinflammatory InterLeukin-6 (IL-6) and Interleukin 1 Beta (IL-1β) are released, and later, in the reparative stage, Transforming Growth Factor Beta (TGF-β), Platelet-Derived Growth Factor (PDGF), Vascular Endothelial Growth Factor (VEGF), and basic Fibroblast Growth Factor (bFGF) are released [50]. During the inflammatory stage, macrophages and tenocytes are recruited to the site of injury. Macrophages digest necrotic materials, and tenocytes are activated and begin proliferating. The release of angiogenic factors also initiates the formation of a vascular network in the healing tissue [51]. In normal healing, the inflammatory phase lasts between 3 and 7 days.
During the proliferative stage, also known as the reparative stage, tenocytes synthesize ECM components, including proteoglycans and collagen. The collagen is primarily type III collagen in random organization [52]. The formation of granular tissue, neovascularization, and epithelialization are the notable characteristics of the proliferative stage, which lasts for several weeks.
The remodeling stage begins 6 to 8 weeks after injury and can take more than a year to complete [52]. During the remodeling phase, the collagen reorganizes along the longitudinal axis of the tendon, thereby restoring tendon stiffness and tensile strength. In addition, tenocytes synthesize and degrade the ECM, replacing the mechanically inferior type III collagen with type I collagen. The ECM continues to mature as collagen fibril crosslinking occurs, and the tissue gains biomechanical strength. However, successful restoration of native tendon structure and function does not occur due to the formation of scar tissue.
Previous reports have suggested that there are two cellular mechanisms of tendon healing, known as intrinsic and extrinsic healing [49]. Intrinsic healing occurs with the proliferation and migration of tenocytes from the epitenon and endotenon, preventing the formation of adhesions [53]. Extrinsic healing, on the other hand, occurs with the invasion of cells from outside the tendon [53]. Although these mechanisms were once believed to be independent of one another, researchers now believe that they must be balanced to optimize tendon healing [53].
Treatment of tendon injuries
The treatment of tendon injuries is conservatively focused. The goal is pain management, rather than treatment of the underlying pathology [35]. The mainstays of conservative treatment include rest or activity modification, physical therapy, immobilization, Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), and/or corticosteroid injections [35,54,55]. Although both NSAIDs and corticosteroid injections provide short-term pain relief, long-term NSAIDs are discouraged, due to the associated risks, including gastrointestinal toxicity, renal damage, and increased cardiovascular risk [56,57], and corticosteroid injections have shown no intermediate or long-term benefit [58,59]. Additionally, there is evidence to suggest that corticosteroids may predispose tendons to rupture, especially in weight-bearing joints [60]. Surgical intervention is often not recommended until conservative treatment options have been exhausted [61]. Even with surgical repair, however, clinical outcomes are less than optimal with reported re-tear rates as high as 94% [8].
For this reason, many biological treatments have been suggested for the management of tendon injuries (Table 3) [61]. Flowable placental connective tissue matrices represent a promising solution. The flowable ECM-like material is designed to fill the deficits within the tendon, facilitate cellular attachment and proliferation, attenuate the inflammatory response, and limit adhesion formation [12].
FP-CTMs
Flowable placental connective tissue matrices are sourced from various placental tissues, such as the amniotic membrane, the chorionic membranes, the umbilical cord, or a combination of these sources. Placental tissues possess anti-inflammatory [62-65], anti-bacterial [66-70], anti-viral [68,69], anti-fibrotic [62,68,71], and immunomodulatory properties [72,73] that are innate to healing (Table 4). In addition to their well-recognized biological properties, placental tissue also has notable mechanical properties, including elasticity, stiffness, and tensile strength [74]. The placenta has a collagen-rich ECM and contains key bioactive molecules, such as fibronectin, laminin, Glycosaminoglycans (GAGs), and elastin, which contribute to its biomechanical properties [75]. The presence of elastin and type III collagen gives the tissue its elasticity; the presence of elastin and interstitial collagens (types I and II), its stiffness; and the lattice-like orientation of collagen bundles, its mechanical strength. The structure of the placental ECM is presumed to promote cell attachment, proliferation, differentiation, epithelialization, and other aspects of healing [76-78]. Despite extensive research documenting the inherent ability of placental tissue to aid in healing, the acquisition and processing of the tissue are constantly evolving, and differences in tissue source and processing have the potential to influence both the biological and mechanical properties of the tissue.
Following stringent donor screening and selection, the tissue is procured and processed. Manufacturers sterilize and preserve the tissue to minimize the risk of disease transmission and to allow prolonged storage, respectively. Tissue preservation is often achieved using one of several preservation methods, most commonly through cryopreservation, drying, or lyophilization [66]. Although these conventional preservation techniques render the amniotic epithelial cells nonviable, the sterilization process is incomplete as evidenced by immunogenic responses in non-decellularized AM [79, 80]. Consequently, removing the cellular content from natural tissue-derived matrices has been suggested to promote healing, and integration with host tissues, and to avoid rejection [81].
Endogenous cells are a contaminant and have the potential to induce host reactivity, including an immune reaction and inflammation, leading to implant rejection. Decellularization is a process whereby endogenous cells, cell debris, and Deoxyribonucleic Acid (DNA) remnants are removed to prevent an immune response while retaining the natural structural and chemical elements of the ECM [82]. Decellularization occurs through mechanical, enzymatic, and chemical means, although it is not rigidly defined [81]. As with the preservation of tissue, decellularization can also affect the structures and entities within the ECM. Therefore, a successful preservation-decellularization protocol must be designed to delicately balance the removal of cellular material as well as retain the innate properties and functional characteristics of the ECM [81-83]. This balance, however, is particularly elusive and is dependent upon tissue source and application.
Commercially available FP-CTMs
Commercially available FP-CTMs are sourced from various placental tissues and are subjected to different processing methodologies (Table 5). The products listed in Table 5 are processed from human tissue according to the American Association of Tissue Banks (AATB) standards and are regulated as a Human Cell, Tissue, or Cellular or Tissue-based Product (HCT/P) by the US FDA under section 361 of the Public Health Service act as HCT/P (21 CFR, Part 127.10a). Indications for use include supplementation or replacement of inadequate or damaged integumental tissue, providing a framework for incorporation by the recipient’s tissues and cells. AmnioFill® (A-CTM) is a minimally manipulated, non-viable cellular tissue matrix allograft that retains ECM proteins, growth factors, cytokines, and other specialty proteins of the placenta [84,85] (Figure 4A). AmnioFix® is a micronized dehydrated human amnion/chorion membrane allograft that also retains ECM proteins, growth factors, cytokines, and other specialty proteins of the placenta. Both products are manufactured using a selective membrane of reparative and reconstructive tissue (SMR2T™) technology and patented PURION® processing. According to the manufacturer, the PURION® process involves the gentle separation of placental tissue layers, cleaning, and tissue reassembly, followed by dehydration of the tissue [86]. During this process, blood components are removed, and the ECM remains intact [86]. BioRenewTM (B-CTM) is an all-natural placental tissue treatment that retains powerful growth factors, cytokines, collagens, tissue inhibitors of metalloproteinases, and bioactive molecules known to modulate the immune system [87,88] (Figure 4B). Interfyl® (I-CTM) is an allogenic decellularized particulate human FP-CTM consisting of natural human structural and biochemical ECM components [12, 62] (Figure 4C & 4D). Contrary to the other FP-CTMs, the decellularization process removes residual cells, cell debris, growth factors, and cytokines, while retaining an ECM structure with high collagen content and key bioactive molecules, such as fibronectin, laminin, GAGs, and elastin [75].
in vitro research supporting FP-CTMs in tendon repair
In 2022, Moreno and colleagues [64] conducted an in vitro investigation to evaluate the effects of micronized dehydrated Human Amnion/Chorion Membrane (µdHACM, MiMedx, Marietta, GA) on the inflammatory environment and hypervascularity associated with tendinopathy. Treatment with µdHACM was found to neutralize proinflammatory cytokines and proteases and regulate angiogenesis. These findings suggest that µdHACM may reduce the influx of inflammatory cells, attenuate inflammation, and improve ECM restoration, which may give rise to a more structurally sound tendon. While encouraging, additional in vivo study is necessitated.
Our research group recently published an in vitro investigation, evaluating the direct interaction between tenocytes and FP-CTMs. The study compared three human FP-CTMs to determine which interacted more favorably with human tenocytes [20]. The FP-CTMs included 1) A-CTM, a minimally manipulated, non-viable cellular particulate; 2) B-CTM, a liquid matrix; and 3) I-CTM, a decellularized flowable particulate. Readouts included tenocyte adhesion and proliferation, cell migration, phenotype maintenance, and inflammatory response. In line with the hypothesis, the study demonstrated that I-CTM, the decellularized FP-CTM, provided a more cell-friendly matrix to support tenocyte function. Although tenocyte attachment was significantly higher on A-CTM, I-CTM supported greater tenocyte proliferation. In addition, I-CTM significantly increased tenocyte migration, whereas A-CTM was comparable to the control. The presence of I-CTM also prevented the loss of the tenocyte phenotype and attenuated the inflammatory response [20]. These findings demonstrate that the direct interaction of tenocytes with FP-CTMs positively modulates the tendon repair environment.
Collectively, the results from these two in vitro reports show promise for the use of FP-CTMs in the treatment of tendon injuries. However, the latter report also suggests that the decellularization of placental tissue may enhance the regulation of inflammatory processes by human tenocytes. Although decellularization is performed to reduce immune response, the process of removing the cellular components can affect the structures and entities within the ECM, disrupting its functional characteristics [1,6,17]. However, it appears as though the decellularization of I-CTM balances the removal of cellular content with the retention of necessary regulatory proteins. This is evident based on I-CTM attenuating the inflammatory response beyond that of other non-decellularized FP-CTMs [62]. More research is needed to determine if the observations are in fact due to decellularization or to other differences in tissue processing. Moreover, the clinical translation of these findings remains to be understood.
in vivo application of FP-CTMs
A comprehensive literature search was conducted to identify studies evaluating the clinical application of FP-CTM for tendon repair [13-15] (Table 6).
In 2015, Lullove published a pilot study reviewing ten patients who received a flowable placental tissue matrix injection (Human Regenerative Technologies LLC, Redondo Beach, CA) to treat tendon or muscular injuries of the lower extremity [13]. These included posterior tibial tendonitis, peroneal tendonitis, anterior tibial tendonitis, extensor muscles of the foot, plantar musculature of the foot, excluding the plantar fascia, and Achilles tendonitis. Ultrasound guidance was used to target the site of injury when administering the injection. Outcome measures included pain and ultrasound evaluation of the tendon/muscle at four and six weeks. By week four, 8/10 patients reported no pain, and by week five, all patients were pain-free. No adverse events or side effects were reported. No standard of the care treatment group was included for comparison.
In 2017, Gellhorn and Han reported a case series to evaluate the use of µdHACM allograft injection (MiMedx, Marietta, GA) for the treatment of tendinopathy or arthritis [15]. Forty patients were included; 20 were treated for tendon pathology and 20 for joint pathology. All patients received an ultrasound-guided injection of µdHACM. Outcomes measures included pain and function, which were measured at 1, 2, and 3 months after the injection. Statistically significant reductions in pain and statistically significant improvements in function were observed. Localized pain at the injection site was common, but no other adverse events or side effects were reported. From this, the authors conclude that µdHACM injection is clinically effective in reducing pain and improving function.
And most recently, in 2020, a retrospective case series was published by Spector and colleagues evaluating µdHACM allograft injection (MiMedx, Marietta, GA) as a treatment for Achilles tendinopathy. Patients were seen twice after the injection within a 45-day observation window. The examined outcome variables included changes in the reported level of pain and treatment-associated adverse events. In the 45-day period, 66% of patients reported complete symptom resolution, while the remaining patients reported symptom improvement without complete resolution. After injection, there were two patients who reported muscle tightness.
Summary & next steps
Placental tissues offer vast clinical utility due to their unique structure, low immunogenicity, and biological properties. The amniotic membrane (AM) of the placenta was first used as a biomaterial for surgical reconstruction in 1910 as a substrate for skin transplantation [89]. However, only fresh AM was available at that time, which was difficult to obtain and carried a risk of disease transmission. Several decades later, with the introduction of better tissue processing techniques and preservation methods, placental tissue has regained popularity and is now used in several specialties, including wound and skin care [12], ophthalmology [90], gynecology [66], and orthopedics [91].
In this report, the literature was reviewed to better understand the clinical application of FP-CTMs in the setting of tendon repair. While the use of FP-CTMs is expanding [91-94], there is a paucity of clinical data evaluating its application in the treatment of tendon pathology. Three studies were identified that used an FP-CTM injection for the treatment of tendonitis/tendinopathy [13-15]. Although the results from these studies demonstrate the safety and efficacy of FP-CTM injection, all three studies are retrospective case series (level IV) that reviewed the results of a single investigator. To systematically evaluate the safety and efficacy of FP-CTMs in tendon healing, a prospective clinical trial is needed.
Prior evidence suggests that differences in processing methodology could influence the direct interaction between tenocytes and FP-CTMs [4,62], potentially causing variation in clinical outcomes. Two of the three clinical case series evaluated the injection of AmnioFix® for the treatment of tendinopathy [14,15], whereas the pilot study from 2015 evaluated the use of PX50® (Human Regenerative Technologies LLC, Redondo Beach, CA). PX50® and DX100® are the flowable forms of BioRenew® PTMTM therapy [95]. All three studies reported statistically significant reductions in pain [13-15], and the two studies evaluating AmnioFix® reported statistically significant improvements in function [14,15]. Both products evaluated are thought to retain ECM proteins, growth factors, collagens, and bioactive molecules found in human placental tissues [84,85,87]. To date, no clinical research studies have evaluated the use of a decellularized FP-CTM to augment tendon healing. This is an important next step, as in vitro evidence suggests that decellularization of the placental tissue may improve tenocyte function and phenotype maintenance and also attenuate the inflammatory response [62].
Two clinical applications have been suggested for the use of FP-CTM in tendon repair [12-15]. The first is for the treatment of tendinitis/tendinopathy with an ultrasound-guided injection [13-15]. The second is an injection directly into the tendon or tendon sheath during the surgical repair of ruptured tendons. At this time, the available clinical evidence is limited to the former method. Additional clinical research is needed to evaluate the outcomes associated with the surgical repair of the ruptured tendon using FP-CTMs.
Although the existing data appears to support the clinical use of FP-CTMs for tendon repair [13-15,62, 91], additional clinical research is needed to more fully assess the safety and efficacy of FP-CTMs in this application. Once the safety and efficacy have been demonstrated, additional randomized, controlled trials are needed to determine if this treatment strategy improves clinical outcomes both in the treatment of tendinosis and tendon tears.
Conclusion
Clinical psychologists significantly have more knowledge about ASD than Medical social workers. Medical Social workers knowledge about ASD is limited (11.51 out of a maximum total score of 19) in this study.
Recommendations
There is the need to factor in teachings and tutorials on ASD in the curriculum development for students of Medical social work while in training and introduction of continuing education for Medical social workers in practice that would incorporate aspects teaching on ASD and related disorders. Also, during the training of Medical Social Workers and Clinical psychologists, clinical rotations in facilities that care for autistic children should be encouraged. More future research especially in Nigeria where is a paucity of information on issues related to ASD is advocated.
- Butler DL, Juncosa N, Dressler MR. Functional efficacy of tendon repair processes. Annu Rev Biomed Eng. 2004;6:303-329. doi: 10.1146/annurev.bioeng.6.040803.140240. PMID: 15255772.
- Praemer AF, Furner S, Rice DP. Musculoskeletal conditions in the united states. 2 ed. Chicago, IL: American Academy of Orthopaedic Surgeons; 1999. 182 p.
- Praemer AF, Furner S, Rice DP. Musculoskeletal conditions in the united states. 1 ed. Chicago, IL: American Academy of Orthopaedic Surgeons; 1992. 199 p.
- McQuilling JP, Burnette M, Kimmerling KA, Kammer M, Mowry KC. A mechanistic evaluation of the angiogenic properties of a dehydrated amnion chorion membrane in vitro and in vivo. Wound Repair Regen. 2019;27(6):609-621. doi: 10.1111/wrr.12757. PMID: 31425636; PMCID: PMC6900065.
- Yelin EH, Cisternas M. Musculoskeletal disease prevalence: United States Bone and Joint Initiative; 2014-2022. 4th. Available from: https://www.boneandjointburden.org/fourth-edition/viiib0/musculoskeletal-disease-prevalence.
- Yelin EH, Cisternas M. Aggregate expenditures: United States Bone and Joint Initiative; 2014-2022. 4th. Available from:https://www.boneandjointburden.org/fourth-edition/viiid2/aggregate-expenditures.
- Reddy GK, Stehno-Bittel L, Enwemeka CS. Matrix remodeling in healing rabbit achilles tendon. Wound Repair Regen. 1999;7(6):518-527. doi: 10.1046/j.1524-475x.1999.00518.x. PMID: 10633012.
- Galatz LM, Ball CM, Teefey SA, Middleton WD, Yamaguchi K. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86(2):219-224. doi: 10.2106/00004623-200402000-00002. PMID: 14960664.
- Alshomer F, Chaves C, Kalaskar DM. Advances in tendon and ligament tissue engineering: Materials perspective. J Mater. 2018;2018:1-17. doi: 10.1155/2018/9868151.
- Hortensius RA, Ebens JH, Harley BA. Immunomodulatory effects of amniotic membrane matrix incorporated into collagen scaffolds. J Biomed Mater Res A. 2016;104(6):1332-1342. doi: 10.1002/jbm.a.35663. PMID: 26799369; PMCID: PMC5030101.
- Liu Y, Ramanath HS, Wang DA. Tendon tissue engineering using scaffold enhancing strategies. Trends Biotechnol. 2008;26(4):201-209. doi: 10.1016/j.tibtech.2008.01.003. PMID: 18295915.
- Brigido SA, Carrington SC, Protzman NM. The use of decellularized human placenta in full-thickness wound repair and periarticular soft tissue reconstruction: An update on regenerative healing. Clin Podiatr Med Surg. 2018;35(1):95-104. doi: 10.1016/j.cpm.2017.08.010. PMID: 29156171.
- Lullove E. A flowable placental tissue matrix allograft in lower extremity injuries: A pilot study. Cureus. 2015;7(6):e275. doi: 10.7759/cureus.275. PMID: 26180699; PMCID: PMC4494536.
- Spector JE, Hubbs B, Kot K, Istwan N, Mason D. Micronized dehydrated human amnion/chorion membrane injection in the treatment of chronic achilles tendinitis. J Am Podiatr Med Assoc. 2021;111(6). doi: 10.7547/19-170. PMID: 34137854.
- Gellhorn AC, Han A. The use of dehydrated human amnion/chorion membrane allograft injection for the treatment of tendinopathy or arthritis: A case series involving 40 patients. PM R. 2017;9(12):1236-1243. doi: 10.1016/j.pmrj.2017.04.011. PMID: 28483683.
- Bordoni B VM. Anatomy, tendons. Treasure Island, FL: StatPearls Publishing; 2022. Available from: https://www.ncbi.nlm.nih.gov/books/NBK513237/
- Yoon JH, Halper J. Tendon proteoglycans: Biochemistry and function. J Musculoskelet Neuronal Interact. 2005;5(1):22-34. PMID: 15788868.
- Kastelic J, Galeski A, Baer E. The multicomposite structure of tendon. Connect Tissue Res. 1978;6(1):11-23. doi: 10.3109/03008207809152283. PMID: 149646.
- Screen HR, Berk DE, Kadler KE, Ramirez F, Young MF. Tendon functional extracellular matrix. J Orthop Res. 2015;33(6):793-799. doi: 10.1002/jor.22818. PMID: 25640030; PMCID: PMC4507431.
- Svärd A, Hammerman M, Eliasson P. Elastin levels are higher in healing tendons than in intact tendons and influence tissue compliance. Faseb j. 2020;34(10):13409-13418. doi: 10.1096/fj.202001255R. PMID: 32794252.
- Samiric T, Ilic MZ, Handley CJ. Characterisation of proteoglycans and their catabolic products in tendon and explant cultures of tendon. Matrix Biol. 2004;23(2):127-140. doi: 10.1016/j.matbio.2004.03.004. PMID: 15246111.
- Smith RK, Gerard M, Dowling B, Dart AJ, Birch HL, Goodship AE. Correlation of cartilage oligomeric matrix protein (comp) levels in equine tendon with mechanical properties: A proposed role for comp in determining function-specific mechanical characteristics of locomotor tendons. Equine Vet J Suppl. 2002(34):241-244. doi: 10.1111/j.2042-3306.2002.tb05426.x. PMID: 12405694.
- Jo CH, Lim HJ, Yoon KS. Characterization of tendon-specific markers in various human tissues, tenocytes and mesenchymal stem cells. Tissue Eng Regen Med. 2019;16(2):151-159. doi: 10.1007/s13770-019-00182-2. PMID: 30989042; PMCID: PMC6439073.
- Kannus P. Structure of the tendon connective tissue. Scand J Med Sci Sports. 2000;10(6):312-320. doi: 10.1034/j.1600-0838.2000.010006312.x. PMID: 11085557.
- Andarawis-Puri N, Flatow EL, Soslowsky LJ. Tendon basic science: Development, repair, regeneration, and healing. J Orthop Res. 2015;33(6):780-784. doi: 10.1002/jor.22869. PMID: 25764524; PMCID: PMC4427041.
- Kjaer M. Role of extracellular matrix in adaptation of tendon and skeletal muscle to mechanical loading. Physiol Rev. 2004;84(2):649-698. doi: 10.1152/physrev.00031.2003. PMID: 15044685.
- Chan JJ, Chen KK, Sarker S, Hasija R, Huang HH, Guzman JZ, Vulcano E. Epidemiology of achilles tendon injuries in collegiate level athletes in the united states. Int Orthop. 2020;44(3):585-594. doi: 10.1007/s00264-019-04471-2. PMID: 31907586.
- Florit D, Pedret C, Casals M, Malliaras P, Sugimoto D, Rodas G. Incidence of tendinopathy in team sports in a multidisciplinary sports club over 8 seasons. J Sports Sci Med. 2019;18(4):780-788. PMID: 31827363; PMCID: PMC6873129.
- Leong HT, Fu SC, He X, Oh JH, Yamamoto N, Hang S. Risk factors for rotator cuff tendinopathy: A systematic review and meta-analysis. J Rehabil Med. 2019;51(9):627-637. doi: 10.2340/16501977-2598. PMID: 31489438.
- Arnold MJ, Moody AL. Common running injuries: Evaluation and management. Am Fam Physician. 2018;97(8):510-516. PMID: 29671490.
- Chang MK, Tay SC. Flexor tendon injuries and repairs: A single centre experience. J Hand Surg Asian Pac Vol. 2018;23(4):487-495. doi: 10.1142/s2424835518500479. PMID: 30428796.
- Charnoff J, Ponnarasu S, Naqvi U. Tendinosis. Treasure Island (FL): StatPearls Publishing; 2022. Available from: https://www.ncbi.nlm.nih.gov/books/NBK448174/.
- Millar NL, Silbernagel KG, Thorborg K, Kirwan PD, Galatz LM, Abrams GD, Murrell GAC, McInnes IB, Rodeo SA. Tendinopathy. Nat Rev Dis Primers. 2021;7(1):1. doi: 10.1038/s41572-020-00234-1. PMID: 33414454.
- Riley G. Chronic tendon pathology: Molecular basis and therapeutic implications. Expert Rev Mol Med. 2005;7(5):1-25. doi: 10.1017/s1462399405008963. PMID: 15796783.
- Riley G. Tendinopathy--from basic science to treatment. Nat Clin Pract Rheumatol. 2008;4(2):82-89. doi: 10.1038/ncprheum0700. PMID: 18235537.
- Steinmann S, Pfeifer CG, Brochhausen C, Docheva D. Spectrum of tendon pathologies: Triggers, trails and end-state. Int J Mol Sci. 2020;21(3). doi: 10.3390/ijms21030844. PMID: 32013018; PMCID: PMC7037288.
- Cook JL, Purdam CR. Is tendon pathology a continuum? A pathology model to explain the clinical presentation of load-induced tendinopathy. Br J Sports Med. 2009;43(6):409-416. doi: 10.1136/bjsm.2008.051193. PMID: 18812414.
- Ensor KL, Kwon YW, Dibeneditto MR, Zuckerman JD, Rokito AS. The rising incidence of rotator cuff repairs. J Shoulder Elbow Surg. 2013;22(12):1628-1632. doi: 10.1016/j.jse.2013.01.006. PMID: 23466172.
- Longo UG, Franceschi F, Ruzzini L, Rabitti C, Morini S, Maffulli N, Denaro V. Histopathology of the supraspinatus tendon in rotator cuff tears. Am J Sports Med. 2008;36(3):533-538. doi: 10.1177/0363546507308549. PMID: 18006676.
- Tempelhof S, Rupp S, Seil R. Age-related prevalence of rotator cuff tears in asymptomatic shoulders. J Shoulder Elbow Surg. 1999;8(4):296-299. doi: 10.1016/s1058-2746(99)90148-9. PMID: 10471998.
- Tashjian RZ. Epidemiology, natural history, and indications for treatment of rotator cuff tears. Clin Sports Med. 2012;31(4):589-604. doi: 10.1016/j.csm.2012.07.001. PMID: 23040548.
- Shamrock AG, Varacallo M. Achilles tendon rupture. Treasure Island (FL): StatPearls Publishing; 2022. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430844/.
- Raikin SM, Garras DN, Krapchev PV. Achilles tendon injuries in a united states population. Foot Ankle Int. 2013;34(4):475-480. doi: 10.1177/1071100713477621. PMID: 23386750.
- Järvinen TA, Kannus P, Maffulli N, Khan KM. Achilles tendon disorders: Etiology and epidemiology. Foot Ankle Clin. 2005;10(2):255-266. doi: 10.1016/j.fcl.2005.01.013. PMID: 15922917.
- Thomopoulos S, Parks WC, Rifkin DB, Derwin KA. Mechanisms of tendon injury and repair. J Orthop Res. 2015;33(6):832-839. doi: 10.1002/jor.22806. PMID: 25641114; PMCID: PMC4418182.
- de Jong JP, Nguyen JT, Sonnema AJ, Nguyen EC, Amadio PC, Moran SL. The incidence of acute traumatic tendon injuries in the hand and wrist: A 10-year population-based study. Clin Orthop Surg. 2014;6(2):196-202. doi: 10.4055/cios.2014.6.2.196. PMID: 24900902; PMCID: PMC4040381.
- Liu CF, Aschbacher-Smith L, Barthelery NJ, Dyment N, Butler D, Wylie C. What we should know before using tissue engineering techniques to repair injured tendons: A developmental biology perspective. Tissue Eng Part B Rev. 2011;17(3):165-176. doi: 10.1089/ten.TEB.2010.0662. PMID: 21314435; PMCID: PMC3098959.
- Millar NL, Murrell GA, McInnes IB. Inflammatory mechanisms in tendinopathy - towards translation. Nat Rev Rheumatol. 2017;13(2):110-122. doi: 10.1038/nrrheum.2016.213. PMID: 28119539.
- Sharma P, Maffulli N. Biology of tendon injury: Healing, modeling and remodeling. J Musculoskelet Neuronal Interact. 2006;6(2):181-190. PMID: 16849830.
- Manning CN, Havlioglu N, Knutsen E, Sakiyama-Elbert SE, Silva MJ, Thomopoulos S, Gelberman RH. The early inflammatory response after flexor tendon healing: A gene expression and histological analysis. J Orthop Res. 2014;32(5):645-652. doi: 10.1002/jor.22575. PMID: 24464937; PMCID: PMC4076155.
- Gelberman RH, Chu CR, Williams CS, Seiler JG, 3rd, Amiel D. Angiogenesis in healing autogenous flexor-tendon grafts. J Bone Joint Surg Am. 1992;74(8):1207-1216. PMID: 1383229.
- Voleti PB, Buckley MR, Soslowsky LJ. Tendon healing: Repair and regeneration. Annu Rev Biomed Eng. 2012;14:47-71. doi: 10.1146/annurev-bioeng-071811-150122. PMID: 22809137.
- Zhao X, Jiang S, Liu S, Chen S, Lin ZY, Pan G, He F, Li F, Fan C, Cui W. Optimization of intrinsic and extrinsic tendon healing through controllable water-soluble mitomycin-c release from electrospun fibers by mediating adhesion-related gene expression. Biomaterials. 2015;61:61-74. doi: 10.1016/j.biomaterials.2015.05.012. PMID: 25996412.
- Kane SF, Olewinski LH, Tamminga KS. Management of chronic tendon injuries. Am Fam Physician. 2019;100(3):147-157. PMID: 31361101.
- Childress MA, Beutler A. Management of chronic tendon injuries. Am Fam Physician. 2013;87(7):486-490. PMID: 23547590.
- Moore RA, Tramèr MR, Carroll D, Wiffen PJ, McQuay HJ. Quantitative systematic review of topically applied non-steroidal anti-inflammatory drugs. Bmj. 1998;316(7128):333-338. doi: 10.1136/bmj.316.7128.333. PMID: 9487165; PMCID: PMC2665568.
- Wilcox CM, Cryer B, Triadafilopoulos G. Patterns of use and public perception of over-the-counter pain relievers: Focus on nonsteroidal antiinflammatory drugs. J Rheumatol. 2005;32(11):2218-2224. PMID: 16265706.
- Gaujoux-Viala C, Dougados M, Gossec L. Efficacy and safety of steroid injections for shoulder and elbow tendonitis: A meta-analysis of randomised controlled trials. Ann Rheum Dis. 2009;68(12):1843-1849. doi: 10.1136/ard.2008.099572. PMID: 19054817; PMCID: PMC2770107.
- Coombes BK, Bisset L, Vicenzino B. Efficacy and safety of corticosteroid injections and other injections for management of tendinopathy: A systematic review of randomised controlled trials. Lancet. 2010;376(9754):1751-1767. doi: 10.1016/s0140-6736(10)61160-9. PMID: 20970844.
- Brinks A, Koes BW, Volkers AC, Verhaar JA, Bierma-Zeinstra SM. Adverse effects of extra-articular corticosteroid injections: A systematic review. BMC Musculoskelet Disord. 2010;11:206. doi: 10.1186/1471-2474-11-206. PMID: 20836867; PMCID: PMC2945953.
- Kim SE, Kim JG, Park K. Biomaterials for the treatment of tendon injury. Tissue Eng Regen Med. 2019;16(5):467-477. doi: 10.1007/s13770-019-00217-8. PMID: 31624702; PMCID: PMC6778558.
- Mao Y, John N, Protzman NM, Kuehn A, Long D, Sivalenka R, Junka RA, Gosiewska A, Hariri RJ, Brigido SA. A decellularized flowable placental connective tissue matrix supports cellular functions of human tenocytes in vitro. J Exp Orthop. 2022;9(1):69. doi: 10.1186/s40634-022-00509-4. PMID: 35849201.
- Shimmura S, Shimazaki J, Ohashi Y, Tsubota K. Antiinflammatory effects of amniotic membrane transplantation in ocular surface disorders. Cornea. 2001;20(4):408-413. doi: 10.1097/00003226-200105000-00015. PMID: 11333331.
- Moreno SE, Massee M, Koob TJ. Dehydrated human amniotic membrane regulates tenocyte expression and angiogenesis in vitro: Implications for a therapeutic treatment of tendinopathy. J Biomed Mater Res B Appl Biomater. 2022;110(4):731-742. doi: 10.1002/jbm.b.34951. PMID: 34611976.
- Hao Y, Ma DH, Hwang DG, Kim WS, Zhang F. Identification of antiangiogenic and antiinflammatory proteins in human amniotic membrane. Cornea. 2000;19(3):348-352. doi: 10.1097/00003226-200005000-00018. PMID: 10832697.
- Mamede AC, Carvalho MJ, Abrantes AM, Laranjo M, Maia CJ, Botelho MF. Amniotic membrane: From structure and functions to clinical applications. Cell Tissue Res. 2012;349(2):447-458. doi: 10.1007/s00441-012-1424-6. PMID: 22592624.
- King AE, Paltoo A, Kelly RW, Sallenave JM, Bocking AD, Challis JR. Expression of natural antimicrobials by human placenta and fetal membranes. Placenta. 2007;28(2-3):161-169. doi: 10.1016/j.placenta.2006.01.006. PMID: 16513165.
- Niknejad H, Peirovi H, Jorjani M, Ahmadiani A, Ghanavi J, Seifalian AM. Properties of the amniotic membrane for potential use in tissue engineering. Eur Cell Mater. 2008;15:88-99. doi: 10.22203/ecm.v015a07. PMID: 18446690.
- Mencucci R, Paladini I, Menchini U, Gicquel JJ, Dei R. Inhibition of viral replication in vitro by antiviral-treated amniotic membrane. Possible use of amniotic membrane as drug-delivering tool. Br J Ophthalmol. 2011;95(1):28-31. doi: 10.1136/bjo.2010.179556. PMID: 20843855.
- Ramuta T, Šket T, Starčič Erjavec M, Kreft ME. Antimicrobial activity of human fetal membranes: From biological function to clinical use. Front Bioeng Biotechnol. 2021;9:691522. doi: 10.3389/fbioe.2021.691522. PMID: 34136474; PMCID: PMC8201995.
- Mamede KM, Sant'anna LB. Antifibrotic effects of total or partial application of amniotic membrane in hepatic fibrosis. An Acad Bras Cienc. 2019;91(3):e20190220. doi: 10.1590/0001-3765201920190220. PMID: 31531535.
- Wassmer CH, Berishvili E. Immunomodulatory properties of amniotic membrane derivatives and their potential in regenerative medicine. Curr Diab Rep. 2020;20(8):31. doi: 10.1007/s11892-020-01316-w. PMID: 32519069; PMCID: PMC7283202.
- Gleason J, Guo X, Protzman NM, Mao Y, Kuehn A, Sivalenka R, Gosiewska A, Hariri R, Brigido SA. Decellularized and dehydrated human amniotic membrane in wound management: Modulation of macrophage differentiation and activation. J Biotechnol Biomater. In press;12(8):1000288.
- Fénelon M, Catros S, Meyer C, Fricain JC, Obert L, Auber F, Louvrier A, Gindraux F. Applications of human amniotic membrane for tissue engineering. Membranes (Basel). 2021;11(6). doi: 10.3390/membranes11060387. PMID: 34070582; PMCID: PMC8227127.
- Bhatia M, Pereira M, Rana H, Stout B, Lewis C, Abramson S, Labazzo K, Ray C, Liu Q, Hofgartner W, Hariri R. The mechanism of cell interaction and response on decellularized human amniotic membrane: Implications in wound healing. Wounds. 2007;19(8):207-217. PMID: 26110364.
- Chen B, Jones RR, Mi S, Foster J, Alcock SG, Hamley IW, Connon CJ. The mechanical properties of amniotic membrane influence its effect as a biomaterial for ocular surface repair. Soft Matter. 2012;8(32). doi: 10.1039/c2sm26175h.
- Malhotra C, Jain AK. Human amniotic membrane transplantation: Different modalities of its use in ophthalmology. World J Transplant. 2014;4(2):111-121. doi: 10.5500/wjt.v4.i2.111. PMID: 25032100; PMCID: PMC4094946.
- Shayan Asl N, Nejat F, Mohammadi P, Nekoukar A, Hesam S, Ebrahimi M, Jadidi K. Amniotic membrane extract eye drop promotes limbal stem cell proliferation and corneal epithelium healing. Cell J. 2019;20(4):459-468. doi: 10.22074/cellj.2019.5423. PMID: 30123991; PMCID: PMC6099140.
- Barton K, Budenz DL, Khaw PT, Tseng SC. Glaucoma filtration surgery using amniotic membrane transplantation. Invest Ophthalmol Vis Sci. 2001;42(8):1762-1768. PMID: 11431440.
- Lee JW, Park WY, Kim EA, Yun IH. Tissue response to implanted ahmed glaucoma valve with adjunctive amniotic membrane in rabbit eyes. Ophthalmic Res. 2014;51(3):129-139. doi: 10.1159/000357097. PMID: 24503529.
- Aamodt JM, Grainger DW. Extracellular matrix-based biomaterial scaffolds and the host response. Biomaterials. 2016;86:68-82. doi: 10.1016/j.biomaterials.2016.02.003. PMID: 26890039; PMCID: PMC4785021.
- Gholipourmalekabadi M, Mozafari M, Salehi M, Seifalian A, Bandehpour M, Ghanbarian H, Urbanska AM, Sameni M, Samadikuchaksaraei A, Seifalian AM. Development of a cost-effective and simple protocol for decellularization and preservation of human amniotic membrane as a soft tissue replacement and delivery system for bone marrow stromal cells. Adv Healthc Mater. 2015;4(6):918-926. doi: 10.1002/adhm.201400704. PMID: 25651408.
- Balestrini JL, Gard AL, Liu A, Leiby KL, Schwan J, Kunkemoeller B, Calle EA, Sivarapatna A, Lin T, Dimitrievska S, Cambpell SG, Niklason LE. Production of decellularized porcine lung scaffolds for use in tissue engineering. Integr Biol (Camb). 2015;7(12):1598-1610. doi: 10.1039/c5ib00063g. PMID: 26426090; PMCID: PMC4666745.
- Koob TJ, Lim JJ, Massee M, Zabek N, Denoziere G. Properties of dehydrated human amnion/chorion composite grafts: Implications for wound repair and soft tissue regeneration. J Biomed Mater Res B Appl Biomater. 2014;102(6):1353-1362. doi: 10.1002/jbm.b.33141. PMID: 24664953.
- Lei J, Priddy LB, Lim JJ, Massee M, Koob TJ. Identification of extracellular matrix components and biological factors in micronized dehydrated human amnion/chorion membrane. Adv Wound Care (New Rochelle). 2017;6(2):43-53. doi: 10.1089/wound.2016.0699. PMID: 28224047; PMCID: PMC5286550.
- MiMedx. Purion process 2022. Available from: https://www.mimedx.com/purion-process/.
- Allograft tissue information & preparation instructions. DX100 7.39rev4 F1 ed: Skye Biologics Inc.; 2017.
- Irvin J, Danchik C, Rall J, Babcock A, Pine M, Barnaby D, Pathakamuri J, Kuebler D. Bioactivity and composition of a preserved connective tissue matrix derived from human placental tissue. J Biomed Mater Res B Appl Biomater. 2018;106(8):2731-2740. doi: 10.1002/jbm.b.34054. PMID: 29437272; PMCID: PMC6220977.
- Davis J. Skin transplantation with a review of 550 cases at the johns hopkins hospital. Johns Hopkins Med. 1910;15:307.
- Walkden A. Amniotic membrane transplantation in ophthalmology: An updated perspective. Clin Ophthalmol. 2020;14:2057-2072. doi: 10.2147/OPTH.S208008. PMID: 32801614; PMCID: PMC7383023.
- Brigido SA, Carrington S, Protzman NM, Mao Y, Pashuck E T, Kohn J, Bhatia M. The use of an acellular connective tissue matrix in hindfoot and ankle fusions: Understanding the cellular bench top data with a consecutive patient series: A pilot study. Clin Res Foot Ankle. 2018;6(3):276. doi: 10.4172/2329-910x.1000276.
- Willett NJ, Thote T, Lin AS, Moran S, Raji Y, Sridaran S, Stevens HY, Guldberg RE. Intra-articular injection of micronized dehydrated human amnion/chorion membrane attenuates osteoarthritis development. Arthritis Res Ther. 2014;16(1):R47. doi: 10.1186/ar4476. PMID: 24499554; PMCID: PMC3978824.
- Hanselman AE, Tidwell JE, Santrock RD. Cryopreserved human amniotic membrane injection for plantar fasciitis: A randomized, controlled, double-blind pilot study. Foot Ankle Int. 2015;36(2):151-158. doi: 10.1177/1071100714552824. PMID: 25249320.
- Zelen CM, Poka A, Andrews J. Prospective, randomized, blinded, comparative study of injectable micronized dehydrated amniotic/chorionic membrane allograft for plantar fasciitis--a feasibility study. Foot Ankle Int. 2013;34(10):1332-1339. doi: 10.1177/1071100713502179. PMID: 23945520.
- Kuebler D, Scarpone M. Biorenew® placental tissue matrix for treatment of tendon and ligament soft tissue injuries. Skye Biologics; 2016.
- Lipman K, Wang C, Ting K, Soo C, Zheng Z. Tendinopathy: Injury, repair, and current exploration. Drug Des Devel Ther. 2018;12:591-603. doi: 10.2147/dddt.S154660. PMID: 29593382; PMCID: PMC5865563.
- Shearn JT, Kinneberg KR, Dyment NA, Galloway MT, Kenter K, Wylie C, Butler DL. Tendon tissue engineering: Progress, challenges, and translation to the clinic. J Musculoskelet Neuronal Interact. 2011;11(2):163-173. PMID: 21625053; PMCID: PMC3689273.
- Kelly MP, Perkinson SG, Ablove RH, Tueting JL. Distal biceps tendon ruptures: An epidemiological analysis using a large population database. Am J Sports Med. 2015;43(8):2012-2017. doi: 10.1177/0363546515587738. PMID: 26063401.
- Clayton RA, Court-Brown CM. The epidemiology of musculoskeletal tendinous and ligamentous injuries. Injury. 2008;39(12):1338-1344. doi: 10.1016/j.injury.2008.06.021. PMID: 19036362.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
