International Journal of Veterinary Science and Research
Microbial quality and handling practices of raw cow milk in North Shewa Zone, Oromia, Ethiopia
Tadesse Birhanu1, Tesfaye Debelu1*, Bosenu Abera2, Alemnesh Yirda2 and Sanago Satalo3
2Salale University, College of Agriculture and Natural Resource, Department of Animal Science, P.O. Box 245, Fitche, Ethiopia
3Jinka University, College of Agriculture and Natural Resources, Department of Animal Science, P.O. Box 165, Jinka, Ethiopia
Cite this as
Birhanu T, Debelu T, Abera B, Yirda A, Satalo S (2022) Microbial quality and handling practices of raw cow milk in North Shewa Zone, Oromia, Ethiopia. Int J Vet Sci Res 8(2): 057-063. DOI: 10.17352/ijvsr.000114Copyright License
© 2022 Birhanu T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Background: Milk is universally accepted as a complete diet that plays a key role to ensure food security. However, the quality of raw cow milk is affected at various stages due to inadequate dairy infrastructure and limited knowledge of hygienic handling practices. Thus, the study aimed to evaluate the microbial quality of raw cow milk and handling practices in the North Shewa zone, Oromia, Ethiopia.
Methodology: Purposive sampling was used to select study districts and participants based on the available dairy production potential, milk market, and collection center. Four hundred participants were selected; 50 raw cow milk samples were collected from households and collection spots in study areas.
Results: Microbial analysis indicated that the mean values of total cell counts were higher in households (4.836±0.206 in log10 CFU/ml) than 4.391±0.15 at collection spots. Similarly, the mean values of Staphylococcus aurous were 2.470±0.038 at the household level and 2.249±0.093 at collection spots. The mean values of E.coli, yeast count and mould count were however, higher at collection spots (5.414±0.100, 3.372±0.091, 2.670±0.037) than household level (3.343±0.122, 2.944±0.074, 2.479±0.039), respectively.
Conclusions: The higher total counts and mean values of microbial loads of economic and public health significance; like Staphylococcus aurous and E. coli at household level and collection spots indicated poor hygienic raw cow milk handling practices. Therefore, awareness should be created among the dairy cattle producers and milk collectors to minimize economic losses through milk spoilage with soil microbes and the risk of consumer safety due to zoonotic pathogens.
Abbreviations
APC: Aerobic Plate Count EcC: E.coli Count; GDP: Gross Domestic Product LB: Lactose Broth; MC: Mold Count; MSA: Mannitol Salt Agar PCA: Plate Count Agar; SaC: Staphylococci aureus Count SPC: Standard Plate Count; TAMBC: Total Aerobic Mesophilic Bacterial Count TCC: Total Coliform Count; VRBA: Violet Red Bile Agar YC: Yeast Count
Background
Ethiopia is endowed with the largest livestock population in Africa, comprising approximately 59.5 million heads of cattle, 30.7 million sheep, and 30.2 million goats. Livestock provides 16% of the total GDP (equivalent to 30% of agricultural GDP) and generates 14% of the country’s foreign exchange [1]. Dairy production, among the livestock production sector, is a critical issue in Ethiopia; because it is among the main sources of food and income. However, the livestock sector has not been fully exploited and promoted in the country [2,3]. Cow milk and milk products generate income for the farm households regularly and provide highly nutritious food for people of all ages, including infants and lactating mothers; reducing the problem of malnutrition among the rural households [4].
Despite the high livestock population, the productivity of the dairy production and benefits obtained from the sector are still very low. For instance, the annual per capita consumption of milk in Ethiopia is very low, (19 liters/person/year) as compared to other African countries like Kenya (120 liters/person/year) and Sudan (180 liters/person/year) [5].
Milk value chains have several outlets through which milk products flow from the producer to the consumer; which affects the quality of milk and transaction costs as well as the potential risk of contamination with pathogens. Subsequently, the safety of dairy products is a major concern around the world, particularly in developing countries where poor handling practices of dairy products from the consumer health point of view may lead to the transmission of various foodborne diseases [6,7].
Recently, an enormous number of smallholder dairy farms have been operating in the North Shewa zone, Oromia, Ethiopia. However, productivity is hindered by several challenges such as shortage of feed, the burden of disease, inadequate veterinary service, and lack of infrastructure coupled with limited knowledge on the hygienic handling practices of raw milk; which could result in public health risks and economic losses [8]. In addition, there is no well-documented information about the microbial quality and handling practices of raw cow milk in the area [9]. Thus, the study was designed to elucidate fundamental information on the microbial quality and assess handling practices of raw cow milk in the North Shewa zone, Oromia, Ethiopia.
Materials and methods
Study areas and design
A cross-sectional study design was conducted in the Kuyu, Girar Jarso, and Wuchale districts of the North Shewa zone, Oromia, Ethiopia. The zone is located 112 km north of Addis Ababa, the capital city of Ethiopia, on the asphalt road connecting Addis Ababa to Gojam. It is located between 9o05’ and 10o23’N latitude and 37o57’ and 39o28’E longitude. The zone has 13 rural districts, one administrative town (Fitche), 18 towns, and 267 rural and 24 urban kebeles. Its altitude ranges from about less than 1000m to over 3540m. Its annual rainfall is from 600 - 2000mm. The average minimum temperature is 10oC and the maximum temperature is 32oC [10]. The major livestock species managed in the areas include cattle, small ruminants, and equines. The subsector contributes to the subsistence requirement of the population in terms of milk, milk products, and meat [8].
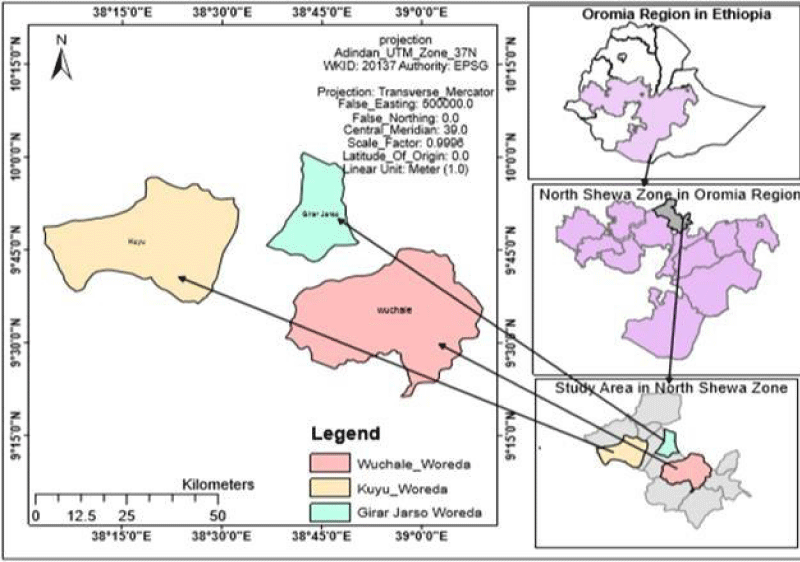
Livestock production is the dominant activity in the zone next to crop production; which accounts 1,676,748 cattle, 118,0430 sheep, 32,4274 goats, 106,472 horses, 8,035 mules, 270881 donkeys, 102,367 poultry and 142,210 beehives (147,268 cultural, 9,101 intermediate and 3,286 modern) in the zone. It is called the milk belt area where 206,000,000 liters of milk per year is produced and more than 60% is supplied to Addis Ababa; because there are a remarkable number of dairy cattle population in the zone; comprising 352,216 local and 1,324,532 crossbreeds. There are 1470 smallholder dairy cattle producers and 30 collectors in the zone [8]. Figure 1 below depicts the map of the north Shewa zone and study districts.
Study participants
The study participants were smallholder dairy cattle producers and collectors in the three selected districts (Kuyu, Girar Jarso, and Wuchale), North Shewa Zone, and Oromia, Ethiopia.
Sampling and sample size determination
A cross-sectional study design was conducted from March 2018 to December 2019. The purposive sampling technique was used to select study districts and study participants based on the potential of dairy cattle production and accessibility to transportation. Three peasant associations were randomly selected from each district. The sample size was determined according to Bartlett, et al. [11]. A list of 1500 smallholder dairy cattle producers and collectors (1470 market-oriented smallholder dairy cattle producers and 30 collectors) was considered as a sampling frame (N).
Where, n = the sample size of the study; N = total number of smallholders and collectors in each peasant association/kebele; e = maximum variability or margin of error of 5% (0.05); 1 = the probability of the event occurring. Therefore, a total of 400 study participants were selected at 5% standard error with a 95% confidence interval.
Data collection
Pre-tested structured questionnaires initially designed and developed in English and translated into the local language (Afan Oromo) were used and then back to English to check for consistency and clarity. Then, face-to-face interviews were carried out about hygienic raw milk handling practices, housing conditions, milk handling practices, milk collection, milk marketing, transportation systems, and types of utensils used for milking and storage and transportation of milk. Both qualitative and quantitative data and structured and semi-structured questionnaires were used. About 18 key informants (veterinarians, para veterinarians (agricultural development agents), and heads of districts’ livestock development agencies) were selected for in-depth interviews using the purposive sampling technique based on their rich experience.
Sample collection and processing
Three peasant associations were purposively selected from each study district based on dairy production potential, milk market, access to milk supply, and collection center. Moreover, collection centers, informal merchants, and dairy cooperative unions in each district were included and interviewed in the survey. A total of 50 milk samples were collected; during collection, 254 ml of raw milk samples were collected aseptically in sterile sample bottles from milking equipment at the household level and bucket at the collection unit in a cold icebox (4°C) to restrict microbial growth and multiplication. The sample was transported to Food Microbiology Laboratory, Ethiopian Public Health Institute, Addis Ababa, for microbiological analysis as early as possible within four hours of collection. Then, serial dilutions of the milk samples were prepared soon following the standard procedures [12].
Microbial identification and enumeration
Microbial quality tests were conducted for the enumeration of bacterial and fungal loads from raw cow milk samples using Standard Plate Count (SPC), Total Aerobic Mesophilic Bacterial Count, Total Coliform Counts, spore-forming bacteria, yeast, and mold using appropriate media [12].
Total Aerobic Mesophilic Bacterial Count (TAMBC): 254 ml of milk sample collected from each dairy farmer and collection unit was dispensed into sterile test tubes in 0.1% peptone water (Himedia), as a 1:9 portion of the peptone water for initial dilution. A serial dilution was made by transferring 1ml of the previous dilution to 10-7. A plate count agar (Himedia) media was used to grow the bacteria at an incubation temperature of 30°C for 72 hours in an inverted direction and the colonies were counted using a colony counter [12].
Total Coliform Count (TCC): 254 ml of milk sample collected from each dairy farmer and the collection unit was dispensed into sterile test tubes containing 9 ml of 0.1% peptone water and thoroughly mixed using a whirling mixer. Subsequent serial decimal dilutions were prepared in a similar manner using 0.1% peptone water. Duplicate appropriate decimal dilutions were surface plated on Violet Red Bile Agar (VRBA) (Himedia, India) and incubated at 45°C for 24 hours. After complete incubation, typically dark red colonies on uncrowned plates were considered Coliform for colony counts [13]. This was followed by a confirmatory test by transferring four to five colonies from each spread plate to tubes of Lactose Broth (LB) (Pharma, US) with inverted Durham tubes. Gas production after 24 hours of incubation at 32°C was considered sufficient evidence of the presence of coliform [12].
Staphylococci aureus Count (SaC): Two ml of milk sample was thoroughly diluted, mixed, and plated on Mannitol Salt Agar (MSA) plates. The dried plates were incubated for 45 - 48 hours at 35°C. Typical Staphylococci colonies appeared as golden yellow, smooth, circular, convex, and moist were counted. For confirmation, a catalase test, coagulase test, and DNase test were conducted and, finally, hemolysis was detected [12].
E. coli Count (ECC): Ten ml of raw cow milk was homogenized in a sterile sample container with saline solution. Decimal dilutions were prepared (10-1 to 10-6). Each dilution was used to inoculate MacConkey agar using a spread plate method. Plates were incubated at 37oC for 24 hours. After incubation, the plates were examined for suspicious colonies of E. coli. Pink to red colonies was suspicious of E. coli. For further confirmation, the indole test was conducted as follows: suspicious colonies were cultured on the nutrient broth and incubated for 24 hours at 37°C. Then, 0.3 ml of Kovac’s reagent was added and checked for dark red color at the top of the test tubes; which are peculiar characteristics of a generic E. coli [12]. Finally, the pure colonies were carefully enumerated.
Yeast and Mold Count (YMC): Samples of raw milk were diluted following similar methods as for aerobic plate count (APC) but dilutions were surface plated on potato dextrose agar at a pH of 5.6±0.2. The dried plates were incubated at 25°C for seven days. Colonies with a yellow-white and slightly dark color were counted as yeasts and molds [12].
Data management and analysis
The data entry, organization, and summarization were performed on an excel spreadsheet (Microsoft office® excel 2007). For the analysis, SPSS version 23 (Armonk, NY: IBM Corp) software was used. Descriptive statistics were employed to describe the frequency, mean values, and proportion (percentages) of respondents; the microbial loads were calculated using a logarithm function. A p-value of < 0.05 and a 95% confidence interval were used to determine the statistical significance of an estimate.
Results
Housing and cleaning practices
In the present study, the majority (79.8%) of the study participants were keeping their cows under better husbandry practices like housing, health care, and barn hygiene; 84.5% of whom were providing a separate house to their dairy cow. For 72% of the study participants, the dairy cows’ barn floors were the earthen floor, while 18.5% of them constructed a barn with a concrete floor. Also, 49.2% of the study participants had the practice of daily cleaning their dairy cow barn (Table 1).
Hygienic milking and milk utensils hygiene
The majority of the respondents (75.8%), have replied that milkers wash hands before milking and between milking two or more cows; however, the remaining 24.2% of them did not practice hand washing both before and between milking. On the other hand, 64% of the respondents have the trend of washing cow’s udder before milking; while 34% of them didn’t wash cow udders. The study has also shown, that 5.3% of the respondents practice teat dipping using antiseptics at the time of milking, while 94.7% did not. For cleaning milk utensils; 45.5%, 28.7%, and 26.8% of the respondents used river, pond, and pipeline water, respectively. Respondents also indicated that there was a trend of smoking milk utensils using olive leaf and stem; among which 47% of them fumigate the milk utensils before and after use (Table 2).
Milk marketing and transportation
The present study has revealed that the majority (72.2%) of the respondents sold milk to milk processors and collectors. Nearly half of the respondents (48.3%) sold milk due to a lack of modern milk processing facilities. Most of the respondents (76.5%) used plastic containers, while the rest 23.5% of them used aluminum-coated metallic utensils for holding and transporting milk. The majority (88%) of the respondents used foot transportation to supply milk for collectors in the study areas (Table 3). This situation is similar to the location in the study areas.
Microbial quality of raw cow milk
The study has revealed the load of EcC as 20% at households and 12% at collection spots; whereas SaC it has shown a microbial load of 20% and 6% at households and collection spots respectively. Table 4 below shows the microbial load test values of raw cow milk evaluated during the study.
The mean values of TAMBC in the study were 4.391±0.15 for households and 4.836±0.206 for collection spots with an overall mean of 4.513±0.170. The TAMBC were 3.762±0.161 log10 CFU mL-1 at the household level and 5.721±0.055 log10 CFU mL-1 at collection spots with the overall mean of 4.188±0.166 log10 CFU mL-1. There was a statistically significant difference between TCC and the sources of milk (p<0.05). In this study, the mean values of EcC were 3.343±0.122 for households and 5.414±0.100 for collection spots with an overall mean of 3.573±0.133. The study has also shown EcC mL-1 of milk samples collected from household milk producers was lower than the raw milk samples obtained from collection spots; however, the difference in EcC between the sources of milk was not statistically significant (p>0.05). The mean values of SaC were 2.470±0.038 for household milk producers and 2.249±0.093 for collection spots with an overall mean of 2.396±0.059. It has been connoted that SaC per milliliter of milk samples collected from household milk producers was higher than the raw milk samples obtained from collection spots, but the difference was not statistically significant (p>0.05). Similarly, the mean values of YC were 2.944±0.074 for household milk producers and 3.372±0.091 for collection spots with an overall mean of 3.106±0.082. The mean values of MC were also 2.479±0.039 for household milk producers and 2.670±0.037 for collection spots with an overall mean of 2.577±0.038. However, there was no statistically significant difference between MC and the sources of milk (p>0.05) (Table 5).
Discussion
The study was the 1st of its kind to analyze the microbial quality and handling practices of raw cow milk in the milk belt areas of the north Shewa zone, Oromia, Ethiopia. Both qualitative and quantitative research methods were employed to perform the study. Poor housing conditions, the unhygienic and poor health status of the lactating cow, unhygienic milking methods, milk storage, and transportation equipment, and unclean water used for washing milk equipment were identified as the major contributing factors to the contamination of raw cow milk. This finding was in line with the finding of Gurmessa [14,15] and Amenu et al. [16]; which identified the health status of a lactating cow and, unclean water and milking utensils as the most decerning factors which deteriorate the microbial quality of raw cow milk. It was also analogous to other findings that were conducted in the central highland of Ethiopia [17,18]. However, it disagrees with the finding of Sintayehu and Haile who reported the highest microbial load of 96.2% from traditionally fermented milk-‘Irgo’, collected from Hawassa town, southern Ethiopia [3].
Similarly, the result of the present study has shown that the mean values of TAMBC and Staphylococcus aureus were higher at the household level as compared to the collection spots; 6.30 and 5.30 log10 CFU mL-1, respectively. This in turn is lower than the upper acceptable limit given by the East African Community Standard [19]. However, it was lower than the findings of Amentie et al. [18] in rural areas of Babile district, eastern Ethiopia. This difference might be due to inadequate dairy infrastructure and lack of vehicles with cooling systems coupled with limited knowledge of the hygienic milk handling practices [15,20,21].
The study was also shown the higher TCC and EcC of raw cow milk obtained from collection spots than at the household level regardless of the location of the study areas. This finding also agrees with the finding of [20,22-25] which declared the higher microbial load of raw cow milk obtained from the bulk tank. It also agrees with the finding of Gebretsadik et al. [26] who reported a significantly lower proportion of contamination of milk collected from dairy farms as compared to milk from vendors and cafeterias. This might be due to cross-contamination of milk during transportation, poor environmental sanitation, lack of sanitation of storage containers, and lack of temperature control throughout the value chain. In addition, since the raw milk samples obtained from collection spots were taken from the bulk tank, contaminated milk from a single household may deteriorate the microbial quality of the entire milk in the tank; and hence, increase the microbial load of the milk samples obtained from collection spots. Thus, it is logical to see increased microbial load in the milk sample obtained from collection spots. The mean values of yeast and mold counts were also higher at collection spots than at the households’ level which was higher than the upper acceptable limit of 2.1 and 1.7 log10 FUmL-1, respectively [16]. This was also in agreement with other findings that were conducted in the eastern highland of Ethiopia [19,27-29]. The presence of high numbers of yeast and mold in milk indicates that the milk has been contaminated with soil, specks of dust, air, and other contaminants due to poor handling practices of raw milk. Moreover, milk was collected on dusty roadsides without shade and cooling systems in study areas.
The result of the key informant interview was triangulated with qualitative data on why most milk producers and collectors used equipment made up of plastic materials and poor milk handling practices in the study areas. This result was in line with the finding of Amenu et al. [16] in southern Ethiopia.
Taken all together, the study indicated a knowledge gap in hygienic handling practices of raw cow milk and a higher microbial load of milk collected from collection spots as compared to milk collected from households. However, no significant difference was observed either in the knowledge gap of dairy cow producers or microbial load over locations in the study areas.
Limitation of the study
The scope of the study was limited to the three districts so the study couldn’t represent the whole North Shewa zone. A cross-sectional study design was employed in this study it is impossible to see a causal relationship by using this design [21].
Conclusion
The microbial analysis indicated that the mean values of TAMBC and SaC were higher at the household level than at collection spots, and both were lower than the upper acceptable limit. However; the mean values of TCC, EcC, YC, and MC were higher at collection spots than at the household level. The study has also shown poor handling practices of raw cow milk, which was primarily due to inadequate dairy infrastructure and limited knowledge on the hygienic handling practices of raw cow milk. Therefore, awareness should be created among milk producers and collectors concerning hygienic handling practices of raw cow milk. Moreover, milk collectors should use a cooling system to maintain bulk tank temperature to limit microbial load and subsequent milk spoilage and consumer safety.
The authors would like to acknowledge Salale University for the financial and logistic support to conduct this study. The study participants and all individuals who rendered help during the study period are highly acknowledged.
- Shapiro BL, Gebru G, Desta S, Negassa A, Nigussie K, Aboset G, Mechale H. Ethiopia livestock sector analysis. ILRIProject Report. Nairobi, Kenya: International Livestock Research Institute (ILRI); 2017.
- Federal Democratic Republic of Ethiopia Central Statistical Agency, Agricultural sample survey report on livestock and livestock characteristics, Volume II. Addis Ababa, Ethiopia; 2019.
- Addo KK, Mensah GI, Aning KG, Nartey N, Nipah GK, Bonsu C, Akyeh ML, Smits HL. Microbiological quality and antibiotic residues in informally marketed raw cow milk within the coastal savannah zone of Ghana. Trop Med Int Health. 2011 Feb;16(2):227-32. doi: 10.1111/j.1365-3156.2010.02666.x. Epub 2010 Nov 10. PMID: 21070512.
- Addo KK, Mensah GI, Aning KG, Nartey N, Nipah GK, Bonsu C, Akyeh ML, Smits HL. Microbiological quality and antibiotic residues in informally marketed raw cow milk within the coastal savannah zone of Ghana. Trop Med Int Health. 2011 Feb;16(2):227-32. doi: 10.1111/j.1365-3156.2010.02666.x. Epub 2010 Nov 10. PMID: 21070512.
- D'Haene E, Desiere S, D'Haese M, Verbeke W, Schoors K. Religion, Food Choices, and Demand Seasonality: Evidence from the Ethiopian Milk Market. Foods. 2019 May 16;8(5):167. doi: 10.3390/foods8050167. PMID: 31100898; PMCID: PMC6560392.
- Food and Agriculture Organization (FAO).Considerations of Food Safety and Consumer Protection. Site visited on 25/8/2013.
- Felleke G. Milk and Dairy Products, Post-harvest Losses and Food Safety in Sub-Saharan Africa and the Near East. A Review of the Small Scale Dairy Sector – Ethiopia. FAO Prevention of Food Losses Program; FAO, Rome, Italy; 2003.
- Zelalem Y, Bernard F. Handling and microbial load of cow’s milk and irgo- fermented milk collected from different shops and producers in central highlands of Ethiopia. Ethiopian Journal of Animal Production. 2006; 6 (2): 67-82.
- North Shewa zone Livestock and Fishery Development Agency (NSZLFDA). Daiy Production and Processing in North Shewa zone, Oromia, Fitche, Ethiopia; 2019
- Ethiopia Topographic Maps East View Cartographic web site accessed on August 14, 2021.
- Bartlett JE, Kotrlik JW, Higgins CC. Organizational Research: Determining Appropriate Sample Size in Survey Research. Information Technology Learn. Performance Journal. 2001; 19 (1).
- BS EN ISO 6887-1. Microbiology of the food chain-preparation of test samples, initial suspension and decimal dilutions for microbiological examination. Part 1: General rules for the preparation of the initial suspension and decimal dilutions; 2017.
- Hussien B, Hailu Y and Eshetu M. Physicochemical Properties and Microbiological Quality of ititu (Traditionally Fermented Cow Milk) in Selected District of Borena Zone, Oromia Regional State, Ethiopia. Open Journal of Animal Sciences. 2010; 11: 125-138.
- Yousef AE, Carlstrom C. Food Microbiology Laboratory Manual .a John Weiley and Son, Inc., Hoboken, New Jersey.Canada, 2003.
- Gurmessa T. Microbiological quality and impact of hygienic practices on raw cow's milk obtained from pastoralists and market in Yabello District, Borana zone, Ethiopia. Global Journal of Food Science and Technology. 2015; 3 (2): 2408-5472.
- Amenu K, Wieland B, Szonyi B, Grace D. Milk handling practices and consumption behavior among Borana pastoralists in southern Ethiopia. J Health Popul Nutr. 2019 Feb 7;38(1):6. doi: 10.1186/s41043-019-0163-7. PMID: 30732649; PMCID: PMC6367795.
- Amentie T, Kebede A, Mekasha Y, Eshetu M. Microbiological Quality of Raw Cow Milk across the Milk Supply Chain in Eastern Ethiopia, East African Journal of Sciences. 2016; 10 (2): 119-132.
- Zelalem Y. Microbial Properties of Ethiopian Marketed Milk and Milk Products and Associated Critical Points of Contamination: An Epidemiological Perspective, Addis Ababa, Ethiopia. 2009; 298-232.
- EACS. Raw Cow Milk-Specification. East African Community (EACS, 2007) standard 2007; 1-19.
- Oliver SP, Jayarao BM, Almeida RA. Foodborne pathogens in milk and the dairy farm environment: food safety and public health implications. Foodborne Pathog Dis. 2005 Summer;2(2):115-29. doi: 10.1089/fpd.2005.2.115. PMID: 15992306.
- Myles Hollander Douglas A. Wolfe, Eric Chicken. nonparametric statistical methods (2nd edition) John Wiley & Sons, Inc, 1999.
- Lencho G, Seblewongel A (2018) Assessment of dairy farmers’ hygienic milking practices and awareness on cattle milk-borne zoonoses in Bishoftu, Ethiopia. Journal of Veterinary Medicine and Animal Health. 2018, 10 (2) : 45-54.
- Fernandes R. Microbiology Handbook Dairy Products. 100pp. FSA (2006). Milk Hygiene on the Dairy Farm. Practical guides for milk producers, England, 2009.
- Khan AA, Massod FA, Bhat BA. Bacteriological quality and safety of raw milk in Kashmir valley, Wayamba Journal of Animal Science. 2011; 2102-5789.
- Kang'ethe EK, Onono JO, McDermott B, Arimi M. Isolation of E. coli O157:H7 from milk and cattle faeces from urban dairy farming and non dairy farming neighbour households in Dagoretti Division, Nairobi, Kenya: prevalence and risk factors. East Afr Med J. 2007 Nov;84(11 Suppl):S65-75. doi: 10.4314/eamj.v84i11.9578. PMID: 18338724.
- Berhe G, Wasihun AG, Kassaye E, Gebreselasie K. Milk-borne bacterial health hazards in milk produced for commercial purpose in Tigray, northern Ethiopia. BMC Public Health. 2020 Jun 9;20(1):894. doi: 10.1186/s12889-020-09016-6. PMID: 32517771; PMCID: PMC7285727.
- Tadele A, Mitiku E, Yoseph M and Ameha K. Milk postharvest handling practices across the supply chain in Eastern Ethiopia. Journal of Advanced Veterinary and Animal Research. 2016; 3(2); 112-126
- International Organization for Standardization (ISO). Microbiology of the food chain Horizontal method for the enumeration of microorganisms: Colony count at 30°C by the surface plating technique. 1st edition 2013; 2.
- McLandsborough LA. Food Microbiology Laboratory. United States of America: CRC Press, 2005.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
