International Journal of Radiology and Radiation Oncology
Effect of intraoperative radiotherapy for patients with low-risk early-stage breast cancer
Diego Carreño1*, Camila Quirland1, Pedro Ferrer2, Luz María González3 and Badir Chahuán4
2Institute of Nutrition and Food Technology, University of Chile, Santiago, Chile
3Oncological Institute, Arturo López Pérez Foundation, Santiago, Chile
4Reconstructive and Oncological Breast Surgery Unit, Oncological Institute, Arturo López Pérez Foundation, Santiago, Chile
Cite this as
Carreño D, Quirland C, Ferrer P, González LM, Chahuán B (2021) Effect of intraoperative radiotherapy for patients with low-risk early-stage breast cancer. Int J Radiol Radiat Oncol 7(1): 001-005 DOI: 10.17352/ijrro.000043Introduction: External Beam Radiation Therapy (EBRT), a type of whole breast radiation therapy, has been widely used in patients with early-stage breast cancer, however it is associated with longer treatment periods and higher toxicity. Intraoperative Radiotherapy (IORT) has emerged as an alternative, reducing treatment times, however, there is uncertainty about its effectiveness and safety.
Objective: To assess the effects of IORT on low-risk early-stage breast cancer in comparison to EBRT.
Methods: We searched in Epistemonikos, a database of systematic reviews, to identify the available evidence evaluating the benefit of IORT on these patients. We extracted data of primary studies from relevant systematic reviews. Data were reanalyzed and a meta-analysis, using evidence of randomized trials, was conducted. Results are reported trough a summary of findings table using the GRADE approach.
Results and conclusions: We identified eight systematic reviews including seven primary studies overall, of which two were randomized trials. We concluded that IORT slightly reduces adverse events and probably increases the local recurrence rate in comparison with EBRT.
Introduction
Breast cancer is the most common cancer and the second cause of cancer death among women in the Americas. Every year more than 462,000 new cases and almost 100,000 deaths from breast cancer occur in the region. In women from Latin America and the Caribbean, this health problem accounts for 27% of new cases and 16% of cancer deaths. Its incidence in the Americas is expected to increase by 2030, with an estimation of 572,000 new cases and 130,000 deaths [1]. In Chile, breast cancer is the most prevalent in women, accounting for 54,227 new cases and 28,584 deaths in 2020. In 75-year-old women or younger, the risk of developing this disease and their mortality is of 16% and 8%, respectively [2].
The breast-conserving therapy combined with post-operative radiotherapy is an equivalent treatment strategy to mastectomy for patients with early-stage breast cancer. External beam radiation therapy (EBRT), a type of whole breast radiation therapy, has become a gold standard as adjuvant therapy after breast-conserving surgery, reducing the risk of local recurrence and improving survival. The results of this intervention are associated with dosedependence, higher incidence of cardiotoxicity and lung cancer, which increases over time after exposure [3].
As an alternative to whole breast radiation therapy, the clinical practice includes the accelerated partial breast irradiation, a localized form of radiation concentrated on the tumor bed, site where most recurrence occur, which reduces the exposure to radiation from nearby organs and minimizes late adverse events [4,5].
Intraoperative radiotherapy (IORT) is one of the validated techniques of accelerated partial breast irradiation, whose greatest advantage over EBRT is that it limits the treatment to one session, which is applied to the tumor bed during surgery [4].
Despite the above, there is still uncertainty about the superiority and safety of the use of IORT over EBRT. Hence, the aim of this review is to assess the comparative effects of IORT over EBRT.
Methods
We searched in Epistemonikos, a database harboring documents from multiple information sources, including MEDLINE, EMBASE, Cochrane, among others, to identify systematic reviews and their included primary studies evaluating the effects of IORT in patients with low-risk early-stage breast cancer in comparison to EBRT [6]. The resulting summary of the body of evidence is presented as an evidence matrix [7]. The FRISBEE methodology was used to extract, review, and analyze the available evidence [8]. The outcomes included in the summary of findings table are those considered critical for decision-making, according to the opinion of the authors, and in general they are in agreement with the systematic reviews identified. Data of relevant outcomes were extracted from the identified reviews, including local recurrence, mortality, grade 3-4 adverse events related to skin radiotherapy toxicity, complications associated with the intervention, health-related quality of life, and physician reported cosmesis (breast appearance). When the available evidence came from more than one primary study, data were reanalyzed using meta-analysis. Results are reported through a summary of findings table following the GRADE approach.
Results
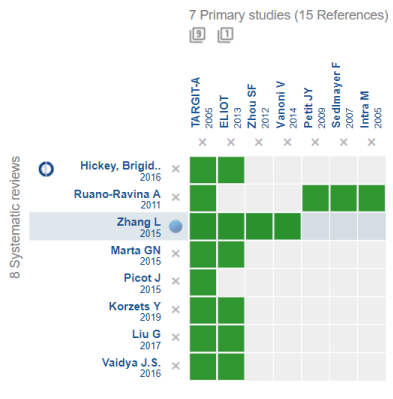
We identified eight systematic reviews [3-5,9-13] including seven primary studies overall [14-22], of which two were randomized trials [14,15]. Figure 1 shows these results in the matrix of evidence elaborated using Epistemonikos database. All further analysis and the reported results are based on the extracted data from the randomized trails, since they represent the body of evidence with the highest level of certainty. Additionally, observational studies did not provide any additional relevant information.
The randomized trials, named TARGIT-A and ELIOT, included patients aged 45 years or older, all of them were women with early-stage breast cancer, harboring tumors of a size of 2.5 cm or less, and that were suitable for breast-conserving surgery. Respect to TNM classification, the largest proportion was T1c and T1ab, and N0 and N1, respectively. In both trials, the analysis of hormone receptor markers showed a higher proportion of estrogen receptor (around 90%). They compared IORT versus EBRT, with doses between 20 at 25 Gray delivered in one full dose and 40 at 45 Gray delivered in 25 fractioned doses, respectively. The TARGIT-A trial used radiotherapy with boost and the ELIOT trial used radiotherapy with or without boost [14,15].
The average follow-up of the trials was 49.3 months with a range between 29 and 69.6 months. Overall, the studies included 4,756 patients. Both measured the outcomes of local recurrence (4,860 patients), overall survival (4,756 patients), complications associated to intervention (4,327 patients), and only the TARGIT-A trial measured the outcome of adverse events (3,451 patients) and physician reported cosmesis (105 patients). Regarding health-related quality of life measurement, no review allowed the extraction of data that could be incorporated into a meta-analysis, therefore, the information on this outcome is presented as a narrative synthesis.
Relevant outcomes, together with their absolute and relative effects, are reported in table. Our results releveled that IORT reduces de risk of grade 3-4 toxicities to the skin in a 69% (95% CI 0.10 - 0.95) when comparing to EBRT. The sources of the data related to this outcome were leveled with highest certainty of evidence. Mortality and complications showed a similar tendency than adverse events; however, these results were not statistically significant. In contrast, local recurrence shows more favorable results for EBRT than IORT, represented by an increase in the risk of recurrence of 206% when treating the patient with the intervention in evaluation. Results of cosmesis were also more favorable for EBRT than IORT, but the treatment effect was not statistically significant. Regarding quality of life, no studies statistically evaluated this outcome; however, the quality-of-life parameters evaluated in the TARGIT-A study were generally favorable for the intraoperative radiotherapy arm Table 1.
Discussion
The principal aim of the present study was to evaluate the effect of IORT on low-risk earlystage breast cancer in comparison to EBRT. Following the GRADE approach, our results show that the risk of local recurrence in patients could probably be three times higher in patients undergoing IORT than EBRT. The benefit in mortality is uncertain and could probably lead to a decrease for both intraoperative and external radiotherapy. Adverse events associated with radiotherapy decrease slightly in patients undergoing IORT compared to those treated with EBRT. In the same way, IORT may slightly decrease complications associated with radiation therapy and the achievement of cosmesis compared to EBRT. Additionally, IORT probably increases quality of life in comparison with external radiotherapy.
The results obtained apply to patients with initial breast cancer, postmenopausal, with tumor of 2.0 cm or smaller, a favorable molecular histological subtype (luminal A), and suitable for breast-conserving surgery. Although some studies included patients from other molecular subtypes, the results in terms of reported local recurrence for them are considerably higher for the intervention, therefore, the use of IORT in this context is not recommended [14].
In addition to the clinical benefits, IORT is associated with lower costs [12]. This could be due to a shorter time of treatment, which implies a reduction in the cost of professional fees and the period of medical rest (less time for medical leave of absence), an early return to work, and a decrease in costs of hospital stay and the transfer of patients who lives far from radiotherapy centers, among others. However, it is important to consider the additional costs associated to its implementation, because it requires the acquisition of the portable linear accelerator and the training of the medical and physical personnel in this technique. Regarding adherence to treatment, given that IORT is performed during surgery, the results obtained with the treatment do not depend on the patient’s commitment to the treatment, as it could be for EBRT. These considerations should be noted for further analyses in the local context for implementation decisions.
Results from relevant systematic reviews are consistent with those presented in this article. Regarding international clinical guidelines recommendations, the use of IORT in women with early-stage breast cancer vary according to the age range of the patients. The American Society of Radiation Oncology (ASTRO) and the European Society for Radiotherapy and Oncology (ESTRO) suggest that patients receiving accelerated partial irradiation on the breast should be at least 50 and 60 years old, respectively. According to these recommendations, some studies have shown favorable results, even when they include patients aged 40 years and older, however, further analysis and follow-up of these studies is required to extend the indication to this population [23].
Conclusion
Our results revealed that IORT does not show an absolute superiority when compared to EBRT, in terms of the outcomes reported in this article. Thus, although IORT is associated with clinical benefits by reducing the incidence of grade 3-4 toxicity to the skin, it does not reduce the risk of local recurrences in these patients in comparison to EBRT. Further research is needed to elucidate which alternative constitute the best option for the treatment of these patients and to clarify the subgroups of patients who may benefit the most with one alternative or the other.
We thank to Epistemonikos foundation for their contribution in the development on this article, since it is part of the Epistemonikos Evidence Synthesis project [6].
- Sánchez J (2020) Cáncer de mama. Pan American Health Organization / World Health Organization. Link: https://bit.ly/3oRXKtY
- World Health Organization (2020) Chile: Globocan 2020. Link: https://bit.ly/2Myyhss
- Korzets Y, Fyles A, Shepshelovich D, Amir E, Goldvaser H (2019) Toxicity and clinical outcomes of partial breast irradiation compared to whole breast irradiation for early-stage breast cancer: a systematic review and meta-analysis. Breast Cancer Res Treat 175: 531-545. Link: https://bit.ly/3pMBtPE
- Zhang L, Zhou Z, Mei X, Yang Z, Ma J, et al. (2015) Intraoperative Radiotherapy Versus Whole-Breast External Beam Radiotherapy in Early-Stage Breast Cancer: A Systematic Review and Meta-Analysis. Medicine (Baltimore) 94: e1143-e1143. https://bit.ly/36LEWql
- Hickey BE, Lehman M, Francis DP, See AM (2016) Partial breast irradiation for early breast cancer. The Cochrane database of systematic reviews 7. Link: https://bit.ly/39NeOgE
- Epistemonikos. Database of the best Evidence-Based Health Care. Link: https://bit.ly/36JTeHY
- Epistemonikos. Interactive matrix: Effect of intraoperative radiotherapy for patients with low-risk early-stage breast cancer. Link: https://bit.ly/2Ll9yr3
- Rada G (2014) Quick evidence reviews using Epistemonikos: A thorough, friendly and current approach to evidence in health. Medwave 14. Link: https://bit.ly/3atgeMc
- Marta GN, Macedo CR, Carvalho HdA, Hanna SA, da Silva JLF, et al. (2014) Accelerated partial irradiation for breast cancer: Systematic review and metaanalysis of 8653 women in eight randomized trials. Radiother Oncol 114: 42-49. Link: https://bit.ly/3oQi5A2
- Liu GC, Dong ZY, Huang BQ, Liu YL, Tang Y, et al. (2017) Efficacy and safety of accelerated partial breast irradiation: a meta-analysis of published randomized studies. Oncotarget 8: 59581-59591. Link: https://bit.ly/2O41yvz
- Vaidya JS, Bulsara M, Wenz F, Coombs N, Singer J, et al. (2016) Reduced Mortality With Partial-Breast Irradiation for Early Breast Cancer: A Meta-Analysis of Randomized Trials. Int J Radiat Oncol Biol Phys 96: 259-265. Link: https://bit.ly/3ay31lv
- Picot J, Copley V, Colquitt JL, Kalita N, Hartwell D, et al. (2015) The INTRABEAM (R) Photon Radiotherapy System for the adjuvant treatment of early breast cancer: a systematic review and economic evaluation [Article]. Health Technology Assessment 19: 1. Link: https://bit.ly/3cJv2t8
- Ruano-Ravina A, Cantero-Muñoz P, Eraso Urién A (2011) Efficacy and safety of intraoperative radiotherapy in breast cancer: A systematic review. Cancer Lett 313: 1525. Link: https://bit.ly/3azbgxD
- Veronesi UP, Orecchia RP, Maisonneuve PD, Viale GP, Rotmensz NM, et al. (2013). Intraoperative radiotherapy versus external radiotherapy for early breast cancer (ELIOT): a randomised controlled equivalence trial. Lancet Oncol 14: 1269-1277. Link: https://bit.ly/2MTHR9o
- Vaidya JSP, Wenz FP, Bulsara MP, Tobias JSP, Joseph DJP, et al. (2014) Risk-adapted targeted intraoperative radiotherapy versus whole-breast radiotherapy for breast cancer: 5-year results for local control and overall survival from the TARGIT-A randomised trial. Lancet 383: 603613. Link: https://bit.ly/2LjXkio
- Zhou SF, Shi WF, Meng D, Sun CL, Jin JR, et al. (2012) Interoperative Radiotherapy of Seventy-two Cases of Early Breast Cancer Patients During Breast-conserving Surgery. Asian Pacific Journal of Cancer Prevention 13: 1131-1135. Link: https://bit.ly/3jlGA7e
- Vanoni V, Bou Selman S, Mussari S, Menegotti L, Rosa M, et al. (2014) External Beam Radiation Therapy Versus Intraoperative Radiation Therapy for BreastConserving Therapy: A Large Single-Institution Matched-Pair Evaluation. International Journal Radiation Oncology, Biology, Physics 90: S261-S261. Link: https://bit.ly/3ttcCmh
- Petit JY, Veronesi U, Rey P, Rotmensz N, Botteri E, et al. (2009) Nipple-sparing mastectomy: risk of nipple-areolar recurrences in a series of 579 cases. Breast Cancer Res Treat 114: 97-101. Link: https://bit.ly/3oLSZ5r
- Sedlmayer F, Fastner G, Merz F, Deutschmann H, Reitsamer R, et al. (2007) IORT with Electrons as Boost Strategy during Breast Conserving Therapy in Limited Stage Breast Cancer: Results of an ISIORT Pooled Analysis. Strahlenther Onkol 183: 32-34. Link: https://bit.ly/2LjjzoC
- Intra M, Leonardi C, Luini A, Veronesi P, Gennari R, et al. (2005) Full-Dose Intraoperative Radiotherapy With Electrons in Breast Surgery: Broadening the Indications. Arch Surg 140: 936-939. Link: https://bit.ly/2MTYYru
- Veronesi U, Orecchia R, Luini A, Galimberti V, Gatti G, et al. (2005) Full-dose intraoperative radiotherapy with electrons during breast-conserving surgery:experience with 590 cases. Ann Surg 242: 101-106. Link: https://bit.ly/3aysdZ2
- Mussari S, Sabino della Sala W, Busana L, Vanoni V, Eccher C, et al. (2006) Full-Dose Intraoperative Radiotherapy with Electrons in Breast Cancer: First Report on Late Toxicity and Cosmetic Results from a Single-Institution Experience. Strahlenther Onkol 182: 589-595. Link: https://bit.ly/36LtyKX
- Correa C, Harris EE, Leonardi MC, Smith BD, Taghian AG, et al. (2017) Accelerated Partial Breast Irradiation: Executive summary for the update of an ASTRO Evidence-Based Consensus Statement. Practical Radiation Oncology 7: 73-79. Link: https://bit.ly/3oO9wG0
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
