International Journal of Pharmaceutical Sciences and Developmental Research
Chronic (52-week) oral toxicity study of herbal tea of Moringa stenopetala and Mentha spicata leaves formulation in Wistar albino rats
Abdu Hassen Musa1*, Girmai Gebru2, Asfaw Debella3, Eyasu Makonnen4, Mesfin Asefa5, Samuel Woldekidan6, Boki Lengiso7 and Chala Bashea7
2Assistant Professor of Histology and Embryology, Department of Anatomy, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia
3Lead Researcher, Directorate of Traditional and Modern Medicine Research, Ethiopian Public Health Institute, Addis Ababa, Ethiopia
4Professor of Pharmacology, Department of Pharmacology and Clinical Chemistry, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia
5Assistant Professor of Pathology, Department of Pathology, Saint Paul’s Hospital Millennium Medical College, Addis Ababa, Ethiopia
6Senior Researcher, Directorate of Traditional and Modern Medicine Research, Ethiopian Public Health Institute, Addis Ababa, Ethiopia
7Senior Medical Laboratory Technologist, Directorate of National Reference Laboratory for Clinical Chemistry and Hematology, Ethiopian Public Health Institute, Addis Ababa, Ethiopia
Cite this as
Musa AH, Gebru G, Debella A, Makonnen E, Asefa M, et al. (2022) Chronic (52-week) oral toxicity study of herbal tea of Moringa stenopetala and Mentha spicata leaves formulation in Wistar albino rats. Int J Pharm Sci Dev Res 8(1): 013-022. DOI: 10.17352/ijpsdr.000038Copyright License
© 2022 Musa AH, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Background: Moringa stenopetala leaves have long been used to treat diabetes, hypertension, respiratory problems, and other diseases. The herbal formulation of Moringa stenopetala and Mentha spicata leaves was found to be more effective in lowering high blood pressure and blood sugar levels. Unlike its pharmacological properties, the long-term safety profile of this herbal formulation has not been investigated yet. Thus, this study investigated the long-term (chronic) oral toxicity of herbal tea of M. stenopetala and M. spicata leaves blended in rats.
Methods: Wistar albino rats were randomly distributed into four groups (n = 10/sex/group), and then randomly assigned to a control group and three test groups. The control group (G I) received distilled water. The test groups (G II-IV) received 559.36, 1118.72, and 2237.44 mg/kg of herbal tea of M. stenopetala and M. spicata leaves blend respectively, for 360 consecutive days. During the treatment period, in-life parameters (mortality, clinical symptoms, body weight, and food intake) were evaluated. On the 361st day, hematological, serum biochemical, gross morphological, and histological parameters were investigated.
Results: Throughout the 360-day treatment period, no herbal tea-related deaths, severe clinical symptoms, loss of body weight, or food intake were seen in any of the treated groups. Bodyweight, food consumption, organ weight, hematological, and serum biochemical findings showed no significant differences between the control and treated groups in both sexes. Macro-pathological and histopathological examinations of the major organs (liver, kidney, heart, pancreas, stomach, and spleen) revealed no herbal tea-related pathologic alterations.
Conclusion: The findings indicate that long-term (360-days) oral administration of the herbal tea of M. stenopetala and M. spicata leaves blend is well tolerated by rats. Hence, it would be safe/low toxic up to a dose of 2237.44 mg/kg/day in chronic exposure.
Abbreviations
BASO: Basophil; CK: Creatine kinase; EOSI: Eosinophil; EPHI: Ethiopian Public Health Institute; LYMP: Lymphocyte; LDH: Lactate Dehydrogenase; MONO: Monocyte; NEUT: Neutrophil; OECD: Organization for Economic Corporation and Development; PLT: Platelet; SPHMMC: Saint Paul’s Hospital Millennium Medical College; TMMRD: Traditional and Modern Medicine Research Directorate.
Introduction
Medicinal plants have been used in disease prevention and treatment since prehistoric times. Some medicinal plants have also been utilized as sources of novel pharmacological agents or modern medications. In many areas of the world, the utilization of medicinal plants has been the primary means of treating ailments. These plants play a significant role in the basic health care system of most developing countries, including Ethiopia [1,2].
In Ethiopia, nearly 80% of the population depend on traditional medicine for their basic health care, and over 95% of traditional medicinal preparations are of plant origin [3,4]. The flora of Ethiopia comprises more than 6500 species of higher plants, of which around 12 – 19% are endemic to the country, and of those higher plants over 732 species are used as medicinal plants. Leaves and roots are dominant and/or most used parts of these medicinal plants [4-6]. In Ethiopia, the leaves of Moringa stenopetala and Mentha spicata are well known for their medicinal and nutritional values [7,8], and their potential in the preparation of herbal recipes [9].
Moringa stenopetala is native to Southern Ethiopia and Northern Kenya. In southern Ethiopia, the leaves of M. stenopetala are widely used in the traditional health system for the treatment of diabetes, hypertension, malaria, asthma, and retained placenta [7,10]. The leaves were tested for a range of pharmacological activities such as antihyperglycemic, antihypertensive, antihyperlipidemic, and diuretic activities [11-13], owing to the presence of several beneficial phytomolecules including flavonoids, phenolic, glycosides, alkaloids, saponins, and glucosinolates [11,14]. Mentha spicata is native to temperate regions of Europe, and western and central Asia [15]. Mentha spicata is widely used as a flavoring agent in food products and as a therapeutic herb in the traditional system of medicine. In different parts of Ethiopia, the M. spicata leaves are used for the treatment of raised blood sugar and blood pressure, excessive flow of menstruation, and various digestive and respiratory tract disorders [8,16,17]. The leaves of M. spicata are reservoirs of several phytomolecules including flavonoids, phenols, glycosides, steroids, and carvone [18-20]. The leaves were also reported to possess various pharmacological activities such as antihyperglycemic, diuretic, antimicrobial, antioxidant, and anti-inflammatory properties [19,21,22].
Herbal formulations have recently gained a lot of interest around the world since herbal mixtures possess some benefits that aren't present in single herbs [23,24]. Such plant-derived formulations are commonly prepared and used for the treatment of a variety of chronic diseases, including, hypertension [24], kidney failure [25], Asthma [26], and diabetes mellitus [27,28]. The herbal formulation made from leaves of Moringa stenopetala and Mentha spicata has shown good antidiabetic and antihypertensive activities on rodent models of hypertension and hyperglycemia. These herbs’ leaves formulation has demonstrated better physicochemical (e.g., good organoleptic properties, excellent flow, and solubility properties, optimal total phenolic and flavonoid contents, ash value and moisture content as well as better particle size, angle of repose, bulk and tapped density) and microbial quality properties [9]. Furthermore, the leaf formulation of these herbs showed low toxic effects during short and mid-term (acute, subacute, and subchronic) oral exposure to rodents [9,29].
Even if it is safe/low toxic in the acute and subchronic exposure, chronic (long-term) usage may have detrimental consequences. A chronic toxicity test is performed to detect the toxicity potentials of a test substance after administration for a larger proportion of the animal’s lifespan. Thus, a long-term toxicity study maximizes the potential for identifying adverse effects that are undetectable on short and mid-term toxicity tests [30,31]. Many adverse effects have been reported with prolonged administration of a variety of herbal formulations, including, cell degeneration and congestion in the blood vessels, congestion and vacuolation in the liver [32], and cytotoxicity, hematological and biochemical alterations [33]. Thus, the long-term toxicity profiles of herbs or their formulations should be scientifically evaluated before their prolonged use and/or to assure patient safety. The present study was performed to assess the chronic toxicity potential of the herbal tea of M. stenopetala and M. spicata leaves blend after repeated-dose 360-day oral administration in rats.
Materials and methods
Plant materials collection, formulation and extraction
Moringa stenopetala leaves were taken from the Arba-Minch University experimental site in Arba-Minch town, Southern Region of Ethiopia (6°01'59" N, 37° 32'59" E). Fresh M. spicata leaves were collected from the botanical garden of Wondo Genet Medicinal and Aromatic Plants Research Center in Wondo Genet town, Sidama Region, Ethiopia (7° 1' N, 38° 35' E). A taxonomist authenticated the leaves of M. stenopetala (Batch No. 011) and M. spicata (Batch No. 012), and voucher specimens were placed in the Traditional and Modern Medicine Research Directorate (TMMRD) of the Ethiopian Public Health Institute (EPHI), Addis Ababa, Ethiopia.
Both plants' leaves were cleaned, shade dried, cut into small pieces, and crushed individually using an electric grinder with a mesh size of 3mm. The coarse powder of the individual plants was sieved with various mesh sizes (5 mm – 212 μm) to obtain a particle size of 212μm leaf granules. The leaf granules were weighed individually and then combined uniformly in an electric blender for 10 minutes in a ratio of 85:15 w/w (M. stenopetala and M. spicata , respectively) with particle sizes of 212 μm. Based on previous investigations, doses of 559.36, 1118.72, and 2237.44 mg/kg body weight of blended leaves of M. stenopetala and M. spicata were chosen for the current chronic toxicity study [9,13,34]. The total weight of the animals in each test group was used to determine the weight of the leaf formulation to be measured. In a funnel-placed glass beaker, the measured herbal formulation was kept on filter paper, and 100ml of boiled distilled water (94.5) was poured over it. The herbal tea infusion was then allowed to cool to room temperature before being given to test group rats [9,13]. The herbal tea infusion was given orally via an intragastric tube in a dosing volume of 2 ml/100 g body weight [31].
Experimental animals
Healthy test drug-naïve male and female Wistar albino rats (Rattus norvegicus, 8-9 weeks old with an average weight of 149.60 + 4.51 g) were obtained from the EPHI's Animal Breeding Unit for the current experiment. The rats were housed in plastic cages with stainless steel wired lids and wood shavings as bedding material (n = 5/sex/cage). The cages were kept in an environmentally controlled animal room (temperature 22 + 30 0C; relative humidity 30–50%; 12-hour light/dark cycle). Free access to drinking water and standard pelleted feeds were provided to the animals. Polypropylene bottles with stainless steel sipper tubes were used to deliver drinking water. Daily, the water bottles and bedding materials were cleaned. The animals were given a week to acclimate to the laboratory settings before being tested. Following the acclimation period, all rats were tattooed on the tail to identify them individually, weighed to determine their body weight ranges, and subjected to detail clinical observations using an individual clinical score sheet to allow for within-subject comparisons. The Institutional Review Board of the College of Health Sciences, at Addis Ababa University, reviewed and approved the experimental protocols (AAUMF-010/18/ANAT), in compliance with the OECD guideline [31]. The animals were cared for and utilized under the European Union Directive 2010/63/EU guidelines [35]. For the reporting of animal research, the ARRIVE guidelines were applied [36]. The animal experiments (i.e., treatments of rats, assessments of toxicological parameters, and data acquisitions) were carried out in the laboratories of EPHI and Saint Paul’s Hospital Millennium Medical College (SPHMMC), Addis Ababa, Ethiopia.
Experimental design
The chronic oral toxicity study was performed according to the OECD guideline test number 452, for testing of chemicals on chronic toxicity study in rodents [31] with a slight modification. Eighty Wistar rats (40 males and 40 females) were randomly distributed into four groups (n = 10/sex/group), and then randomly assigned to a control group and three test groups for experimentation. The control group of male and female rats (G-I) received a vehicle (distilled water). The test groups (both males and females in G-II, G-III, and G-IV) were treated with the herbal teas of M. stenopetala and M. spicata leaves blends (at concentrations of 559.36, 1118.72, and 2237.44 mg/kg body weight, respectively) once daily for a total of 360 days.
Mortality and clinical signs observations
All animals were inspected twice a day for mortality and symptoms of illness throughout the treatment period. General clinical assessments were made once a day, usually after dosing. Once a week, outside of their home cages, detailed clinical observations were performed on all animals. The presence of stereotypies (e.g., excessive grooming, and repetitive circling) and/or bizarre behaviors (e.g., self-mutilation, and walking backward), as well as changes in skin, fur, eyes, mucous membranes, autonomic activity, locomotion, posture, and responses to handling, were all documented.
Body weight and food consumption measurements
Each rat's body weight was measured before the first exposure, then once a week, and lastly at necropsy (on the 361st day). Once a week, food consumption was recorded per cage (n = 5/sex).
Hematological and serum biochemical analyses
After the last (360th day) treatment, all surviving rats fasted for 12 hours (food, but not water). Following the period of fasting, they were anesthetized with pentobarbital (150 mg/kg of body weight) via intraperitoneal injection [37]. Then, a 4 - 6 cc blood sample was taken from each anesthetized rat through cardiac puncture and deposited into two separate test tubes. The first blood sample was placed in a test tube with anticoagulant (di-potassium salt of ethylene-diamine tetra-acetic acid) and analyzed using an Automated Hematology Analyzer (Symex-XT, 1800i, Japan) to determine values of total white blood cell count (WBC), total red blood cell count (RBC), blood platelet (PLT) count, hemoglobin (HGB), hematocrit (HCT), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), relative lymphocyte count (LYMP%), relative monocyte count (MONO%), relative neutrophil count (NEUT%), relative eosinophil count (EOSI%) and relative basophil count (BASO%). The second blood sample was placed in a plane test tube and maintained at room temperature for one hour before being centrifuged to obtain serum using an electric centrifuge. The serum was then analyzed using an Automated Clinical Chemistry Analyzer (Huma Star 80, Germany) to assess the serum levels of alanine transaminase (ALT), alkaline phosphate (ALP), aspartate transaminase (AST), gamma-glutamyl transferase (GGT) creatine kinase (CK), lactate dehydrogenase (LDH), glucose, total cholesterol, creatinine, urea, total protein, direct bilirubin, amylase, albumin, high-density lipoproteins cholesterol (HDLC), triglyceride (TG), Na+, K+, Mg2+, and Cl-.
Gross necropsy and organ weight
On the day of necropsy (on the 361st day), all rats were inspected for gross morphological alterations or lesions in their physical appearance (e.g., emaciated, thin, or obese), skin, natural orifices, eyeballs, and eyelids. They were then dissected and lesions/changes in the subcutaneous tissue, major body cavities (e.g., excessive fluid/blood), and vital organs (e.g., changes in size, color, shape, and texture) were documented. The liver, kidney, heart, pancreas, stomach, and spleen were excised, cleaned of from any adherent tissues, weighed (absolute weight), and samples were taken for histological investigation.
Histopathological examinations
Tissue samples from the liver, kidney, heart, pancreas, stomach, and spleen were fixed in a 10% neutral buffered formalin solution, dehydrated in an ascending series of alcohol, cleared in two changes of xylene, infiltrated in two changes of melted paraffin wax, embedded in melted paraffin wax, and sliced with a thickness of 4 – 5 µm using a rotary microtome. The tissue slices were then mounted on slides, deparaffinized in three changes of xylene, rehydrated in decreasing concentrations of alcohol, rinsed in distilled water, stained with Harris hematoxylin, decolorized in acid alcohol, immersed in sodium carbonate solution, washed in running tap water, counterstained with eosin Y, dehydrated by an increasing concentration of alcohol, cleared in two changes of xylene, mounted with Dibutylphthalate polystyrene xylene, covered with coverslips, and then analyzed microscopically for pathological alterations [38,39], and photomicrographs were recorded by a digital camera (Nicon Coolpix-5000, Germany).
Statistical analysis
The Statistical Package for Social Sciences (SPSS) version 24 software (SPSS Inc., Chicago, IL, USA) was used to analyze the data collected in this study. For determining the significant difference between groups, the data were subjected to a one-way analysis of variance (ANOVA). Dunnett's post hoc test was used to determine the significant difference between the control and treated groups. The findings were presented as mean + standard deviation (M + SD). P < 0.05 was considered statistically significant.
Results
Mortality and clinical signs
In both sexes, no herbal tea-related death or severe symptoms of toxicity were observed in any of the dosage groups (559.16, 1118.72, or 2237.44 mg/kg/day) throughout the chronic treatment period. In addition, the treated group of rats showed no visible physical, physiological, or behavioral changes throughout the short and long-term observation period. All the clinical observations (like mild piloerection and mild diarrhea) were transient and/or common in the studied species of rat, as they were noted in the control group of rats.
Body weight and food consumption
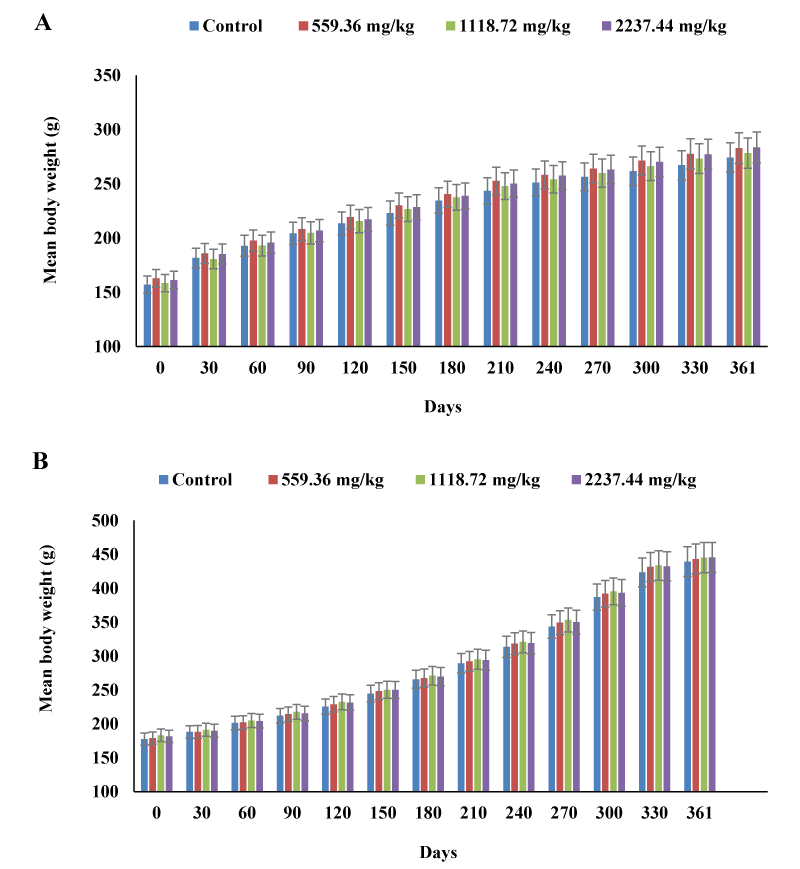
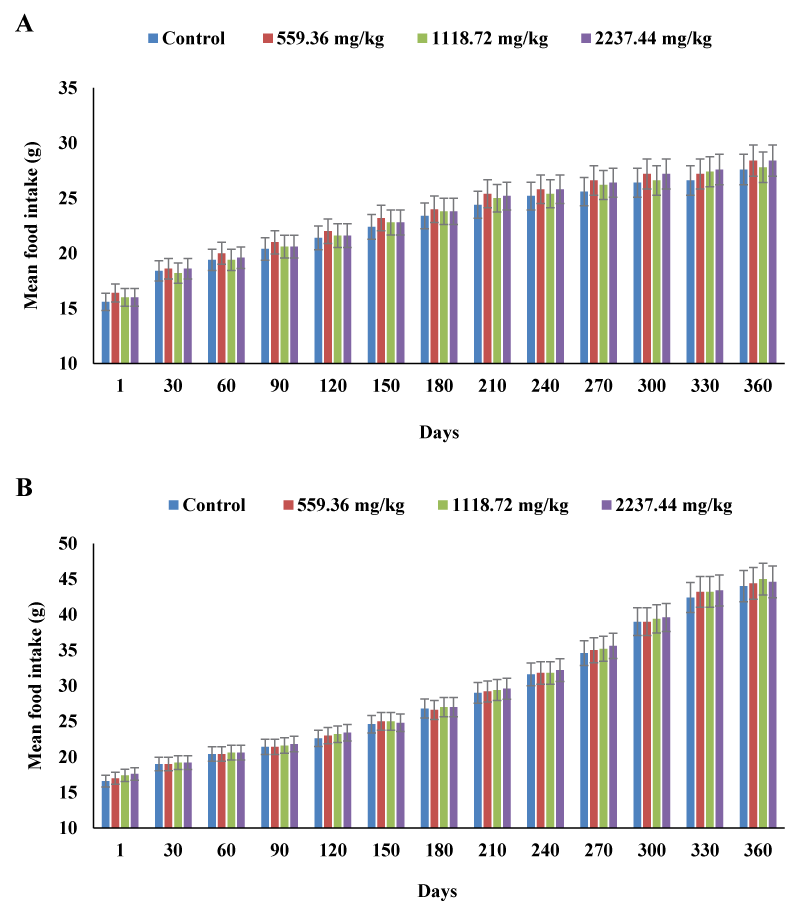
The herbal tea had no adverse effects on body weight or body weight gain in male and female rats. In all test groups, both male and female rats showed a steady increase in body weight throughout the administration period. The weekly body weight increment in the test groups was generally comparable with those of the control groups (Figure 1). When compared with their respective initial body weights, all study groups had gained weight by the end of the experiment. In females, the final weight gain increased by 2.6 % at 559.16 mg/kg, 2.4 % at 1118.72 mg/kg, and 4.4 % at 2237.44 mg/kg, when compared with the value of 117.20 + 7.29 g of the control group. The final weight gain in male test groups increased by 0.84 % at 559.16 mg/kg, 0.15 % at 1118.72 mg/kg, and 0.76 % at 2237.44 mg/kg, as compared with the data (261.80 + 6.22 g) of the control males. The treated groups in both sexes had gained more weight than the respective control groups. However, the differences were statistically insignificant for both sexes. Similarly, herbal tea had no negative effects on food consumption. Both male and female rats in the treated groups had a progressive increase in food consumption throughout the experiment (Figure 2). However, there was no statistically significant difference in the weekly food consumption between the control and treated groups in both sexes. Moreover, the gradual increase in body weight was associated with the normal increase in food consumption throughout the 56-weeks study period.
Hematological parameters
Table 1 shows data of the hematological parameters by dosage and sex groups measured after chronic administration of the herbal tea of M. stenopetala and M. spicata leaves blend. The results of hematological parameters in the treated group of females showed a decrease in RBC, HGB, HCT, MCH, MCHC, MCV, PLT, MONO, and LYMP, while an increase in WBC, NEUT, BASO, and EOSI, which are mainly in a non-dose dependent manner, in comparison with the control group values. In treated male groups, PLT, HCT, MCH, MCV, and LYMP showed an increment, while WBC, RBC, HGB, MCHC, BASO, and EOSI displayed a decrement in a non-dose dependent manner. The contents of MONO and NEUT in the treated males also showed a decrement except for herbal tea at a low dose level, which exhibited an increment in MONO and NEUT when compared with the values of the respective parameters of the control group. However, there were no statistically significant differences in the values of the aforementioned hematological parameters between the control and treated groups for both male and female rats. Moreover, the changes in these hematological parameters were minor and fall within the normal ranges for both sexes of rats.
Biochemical parameters
Serum levels of biochemical parameters in male and female rats are tabulated in Table 2. In the treated female groups, ALT, ALP, AST, HDLC, creatinine, direct bilirubin, amylase, Na+, Mg2+, and Cl- increased, while GGT, CK, LDH, TG, glucose, total cholesterol, urea, total protein, albumin, and K+ decreased in a non-dose dependent manner in comparison with the values the respective parameters in the control females. A non-dose dependent increase in ALT, AST, CK, HDLC, creatinine, K+, and Cl-, while a non-dose dependent decrease in ALP, GGT, LDH, TG, urea, total protein, direct bilirubin, total cholesterol, albumin, glucose, amylase, Mg2+, and Na+ were observed in the treated male groups as compared with the control group values. However, the serum biochemical analysis revealed no statistically significant differences in the values of the above-mentioned parameters between the control and treated groups in either sex. Moreover, the observed minor changes both in male and female test groups fall within the reference ranges for rats of the same age and sex.
Gross necropsy and organ weight
At necropsy, no herbal tea-associated macro-pathological lesions or changes were detected on the internal and external organs of the treated groups. Furthermore, the absolute weight of the liver, kidney, heart, pancreas, spleen, and stomach showed no statistically significant variations between the control and treated groups in both male and female rats, as presented in Table 3.
Histopathology of the major organs
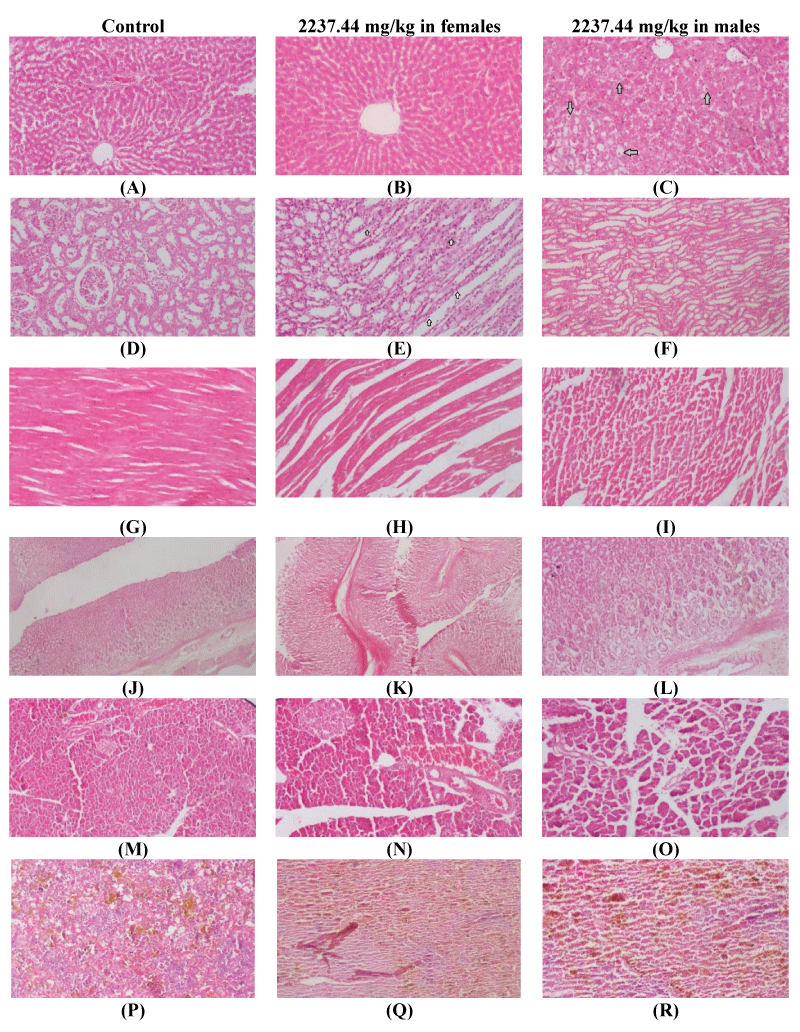
After repeated dose (360-days) treatment with the herbal tea of M. stenopetala and M. spicata leaves blend, the liver, kidney, heart, pancreas, stomach, and spleen were sampled and examined for any herbal tea-related microscopic lesions or changes. In histopathological examination, a few rats from all study groups had mild hydropic changes in the liver and mild inflammation of the kidney (Figure 3). Nevertheless, these hepatic and renal pathologic lesions did not exhibit dose-dependent changes, did not distort the general architecture of the organs, and were detected both in the vehicle and herbal tea-treated group rats. The general architectures and histologic components of the heart, pancreas, stomach, and spleen in all study group rats were normal.
Discussion
Herbal mixtures have gained widespread acceptance because it has been proven that herbal formulations including two or more distinct herbs generate better therapeutic results than individual herbs [23,24]. The herbal formulations are widely used for the management of chronic diseases such as kidney failure [25], Asthma [26], and diabetes mellitus [27]. The herbal formulation used in this study, which is prepared from the blended leaves of M. stenopetala and M. spicata , has shown to be more effective in lowering high blood pressure and blood sugar levels [9]. Moreover, the leaf formulation of these herbs showed low toxic effects during short and mid-term (acute and subchronic) oral exposure to rodents [9,29]. However, the long-term (chronic) toxicity profile of this herbal blend has not been evaluated yet. A chronic toxicity test is performed to detect the cumulative and latent toxicity potentials of a test substance or compound after administration for a larger proportion of the animal’s lifespan [30,31]. The present study evaluated the chronic (360-days) oral toxicity of herbal tea of M. stenopetala and M. spicata leaves blended in male and female rats.
Death and severe clinical symptoms are the most serious consequences of a test compound's toxicity [40]. Throughout the 360-day treatment period, oral administration of the herbal tea of M. stenopetala and M. spicata leaves blend caused no death or serious toxic symptoms in both male and female rats. All the clinical observations in the treated groups (like mild piloerection and mild diarrhea) were transient and/or common in the studied species of rat, as they were noted in the control group rats.
In toxicity studies, loss of body weight and food consumption are used as sensitive indicators of illness or to assess test substance-related effects [30]. During the chronic experimental period, all study group rats demonstrated gradual changes in body weight and food consumption. However, there were no significant differences in body weight between the control and test groups of both sexes. Similarly, food consumption showed no significant differences between the control and treated groups throughout the experiment. These findings suggest that long-term administration of the herbal tea may not induce adverse effects on the general health, normal growth, and dietary intake of rats at the doses tested. The current in-life findings are in line with previous chronic toxicity studies of the leaf extract of M. stenopetala in rats [41].
Hematology and serum biochemistry analyses are important components of the safety assessment processes for medicinal herbs and novel pharmaceuticals under development. These clinical pathology parameters are used to identify the extent of a test compound's harmful effect on blood tissue and various organs [30,42]. In the current study, values of the hemograms (e.g., WBC, RBC, PLT, HGB, HCT, MCH, and MCHC) and serum biochemicals (e.g., ALT, AST, ALP, creatinine, urea, total protein, glucose, amylase, and cholesterol) did not exhibit statistically significant variations between the control and treated groups in both male and female rats. All the observed clinical pathology changes were minor and fall within the normal reference ranges for both sexes, suggesting that chronic oral administration of the herbal tea up to the high dose of 2237.44 mg/kg/day may not induce adverse effects on blood parameters in rats. The present clinical pathology findings were agreed with previous long-term toxicity studies of leaves extract of Lignosus rhinocerotis [43], and the fruiting body of Lignosus rhinocerotis [44] in rats.
Anatomic pathological lesions induced by test chemicals on vital organs are considered the principal observations among toxicity indicators [30,40]. At the end of the treatment period, no herbal tea-related macro-pathological lesions or changes were seen in the major internal and external viscera of both male and female rats. Furthermore, no histopathological alterations were observed in the heart, pancreas, stomach, and spleen sections of male and female rats treated with different doses of herbal tea infusions. However, the liver and kidney sections of a few male and female rats from all study groups displayed minor histopathologic changes (i.e., mild hydropic changes of the liver and mild inflammations of the kidney).
These hepatic and renal pathologic lesions might be associated with active ingredients of the herbal tea or its metabolites. Similar hepatic and renal pathologic lesions were observed after chronic administration of the leaves extracts of M. stenopetala [41] and Stachytarpheta cayennensis [45] in rats. Nevertheless, the hepatic and renal pathologic lesions in this study were not induced by the herbal tea, since they were not accompanied by significant alterations of the liver test enzymes (e.g., ALT, AST, ALP) and renal biomarkers (e.g., urea and creatinine), did not show dose-dependent changes (in terms of frequency and severity), and they were also detected in the tissue sections of the control rats. Thus, the current pathologic lesions might be spontaneous or background lesions that are observed in the population of Wistar albino rats, and/or they may represent normal anatomic variability within a population of the study rats [46,47].
Conclusion
In the present chronic toxicity study, oral administration of the herbal tea of M. stenopetala and M. spicata leaves blend caused no death or severe toxic symptoms in male and female rats throughout the experimental period of 360-days. Body weight, food consumption, organ weight, hematological, and biochemical findings did not show herbal tea-related significant alterations. There were no herbal tea-associated pathologic changes in the general histologic architecture of the major organs. These results demonstrate that chronic (360-day) oral administration of the herbal tea of M. stenopetala and M. spicata leaves formulation is safe/low toxic to rats up to the maximum dose of 2234.77 mg/kg/day. However, further preclinical (e.g., reproductive and developmental) studies and clinical trials are required to obtain comprehensive toxicological data and to approve its safety in humans.
Data availability statement
The data supporting the reported results are found within this manuscript and details are available upon request to the corresponding author.
Authors’ contributions
AHM: Conceptualization, data curation, formal analysis, investigation, methodology, project administration, validation, visualization, writing—original draft preparation, writing—review, and editing. AD: Conceptualization, data curation, formal analysis, methodology, project administration, resources, supervision, validation, visualization, writing—review, and editing. GG: Conceptualization, data curation, formal analysis, methodology, project administration, supervision, validation, visualization, writing—review, and editing. EM and MA: Data curation, supervision, methodology, resources, validation, visualization, writing—review, and editing. SW, CB, and BL: Investigation, resources, writing—review, and editing. All authors have read and agreed to the published version of the manuscript.
The authors appreciate and thank the Ethiopian Public Health Institute (EPHI) and the Saint Paul’s Hospital Millennium Medical College (SPHMMC), Addis Ababa, Ethiopia for the provision of their laboratories, materials, and chemicals.
- Mashabela NM, Otang-Mbeng W. Evaluating the Effect of Fertilizers on Physiological Growth, Chemical, Bioactive Components and Secondary Metabolites in Vigna Unguiculata (L.). Plant Biotechnol Persa. 2022; 4: 37-49.
- James PB, Wardle J, Steel A, Adams J. Traditional, complementary and alternative medicine use in Sub-Saharan Africa: a systematic review. BMJ Glob Health. 2018 Oct 31;3(5):e000895. doi: 10.1136/bmjgh-2018-000895. PMID: 30483405; PMCID: PMC6231111.
- Fassil H. Beyond Plants Professionals & Parchments: The role of home-based medicinal plant use and traditional health knowledge in primary health care in Ethiopia. Ethnobot Res Appl. 2005; 3: 037-047.
- Awulachew MT. Hand Book of Common Ethiopian Traditional Medicinal Plants: Their Parts and Uses for Human and Animal Treatments. J Dis Med Plants. 2021. 7: 48-60.
- Abebe AH. Important Medicinal Plants in Ethiopia: A Review in Years 2015–2020.” Herbs and Spices - New Processing Technologies [Working Title]. IntechOpen 1-17.2021.
- Admasu M, Yohannes M. Ethiopian Common Medicinal Plants: Their Parts and Uses in Traditional Medicine -Ecology and Quality Control. Intech Open. 2020. 5-22.
- Mekonnen Y, Gessesse A. Documentation on the uses of Moringa stenopetala and its possible antileishmanial and antifertility effects. SINET: Ethiop J Sci. 1998; 21: 287-295.
- Belayneh A, Asfaw Z, Demissew S, Bussa NF. Medicinal plants potential and use by pastoral and agro-pastoral communities in Erer Valley of Babile Wereda, Eastern Ethiopia. J Ethnobiol Ethnomed. 2012; 8: 1-10.
- Debella A. Moringa stenopetala leaves and herbal adjuvants blend medicinal tea granule package for supportive care of pre-diabetes and pre-hypertension. Paper presented at: Dissemination Workshop of Multisectoral Interdisciplinary Research Findings on Moringa stenopetala , February 25-28, 2021; Arba-Minch University, Southern region, Ethiopia. 2021; 7: 50-51.
- Edwards S, Tadesse M, Demissew S, Hedberg I. Flora of Ethiopia and
Eritrea, Vol. 2, part1: Magnoliaceae to Flacourtiaceae. Uppsala, Sweden 2000. - Sileshi T, Makonnen E, Debella A, Tesfaye B. Antihyperglycemic and subchronic toxicity study of Moringa stenopetala leaves in mice. J Coastal Life Med. 2014; 2: 214-221.
- Geleta B, Makonnen E, Debella A, Tadele A. In vivo antihypertensive and antihyperlipidemic effects of the crude extracts and fractions of Moringa stenopetala (Baker.) Cufod. leaves in rats. Front Pharmacol. 2016; 7: 1-10.
- Fekadu N, Basha H, Meresa A, Degu S, Girma B, Geleta B. Diuretic activity of the aqueous crude extract and hot tea infusion of Moringa stenopetala (Baker f.) Cufod. leaves in rats. J Exp Pharmacol. 2017 Jun 21;9:73-80. doi: 10.2147/JEP.S133778. PMID: 28684924; PMCID: PMC5484571.
- Mengistu M, Abebe Y, Mekonnen Y, Tolessa T. In vivo and in vitro hypotensive effect of aqueous extract of Moringa stenopetala . Afr Health Sci. 2012 Dec;12(4):545-51. PMID: 23515422; PMCID: PMC3598298.
- Lawrence BM (Ed.). Mint: the genus Mentha. CRC Press 2006.
- Kassahun BM, Egata DF, Lulseged T, Yosef WB, Tadesse S. Variability in Agronomic and Chemical Characteristics of Spearmint (Mentha spicata L.) Genotypes in Ethiopia. Int J Adv Biol Biom Res. 2014; 2: 2704-2711.
- Banjaw DT, Dikir W, Gebre A, Geja W, Tsegaye D, Molla Y. Aromatic and medicinal plants in Wondo Genet Agricultural Research Center Botanical Garden, South Ethiopia. Med Aromat Plants (Los Angel). 2016; 5: 278.
- Shahbazi Y. Chemical Composition and In Vitro Antibacterial Activity of Mentha spicata Essential Oil against Common Food-Borne Pathogenic Bacteria. J Pathog. 2015;2015:916305. doi: 10.1155/2015/916305. Epub 2015 Aug 16. PMID: 26351584; PMCID: PMC4553199.
- Snoussi M, Noumi E, Trabelsi N, Flamini G, Papetti A, De Feo V. Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules. 2015 Aug 7;20(8):14402-24. doi: 10.3390/molecules200814402. PMID: 26262604; PMCID: PMC6332415.
- Brahmi F, Khodir M, Mohamed C, Pierre D. Chemical Composition and Biological Activities of Mentha Species, In: Aromatic and Medicinal Plants - Back to Nature. IntechOpen.2017; 2017: 48-55.
- Tyagi AK, Malik A. Antimicrobial potential and chemical composition of Mentha piperita oil in liquid and vapor phase against food spoiling microorganisms. Food Control. 2011; 22: 1707–1714.
- Bayani M, Ahmadi-Hamedani M, Jebelli Javan A. Study of Hypoglycemic, Hypocholesterolemic and Antioxidant Activities of Iranian Mentha spicata Leaves Aqueous Extract in Diabetic Rats. Iran J Pharm Res. 2017 Winter;16(Suppl):75-82. PMID: 29844778; PMCID: PMC5963648.
- Li P, Su W, Yun S, Liao Y, Liao Y, Liu H, Li P, Wang Y, Peng W, Yao H. Toward a scientific understanding of the effectiveness, material basis and prescription compatibility of a Chinese herbal formula Dan-hong injection. Sci Rep. 2017 Apr 10;7:46266. doi: 10.1038/srep46266. PMID: 28393856; PMCID: PMC5385876.
- Sholikhah EN, Mustofa M, Nugrahaningsih DAA, Yuliani FS, Purwono S, Sugiyono S, Widyarini S, Ngatidjan N, Jumina J, Santosa D, Koketsu M. Acute and Subchronic Oral Toxicity Study of Polyherbal Formulation Containing Allium sativum L., Terminalia bellirica (Gaertn.) Roxb., Curcuma aeruginosa Roxb., and Amomum compactum Sol. ex. Maton in Rats. Biomed Res Int. 2020 Mar 24;2020:8609364. doi: 10.1155/2020/8609364. PMID: 32309440; PMCID: PMC7136774.
- Maurya H, Kumar T. Formulation, standardization, and evaluation of polyherbal dispersible tablet. Int J App Pharm. 2019; 11:158-167.
- Saleem U, Usman M, Anwar F, Akhtar MF, Ahmad B. Pharmacological and toxicological evaluation of two anti-asthmatic polyherbal formulations. An Acad Bras Cienc. 2020 Oct 12;92(3):e20191562. doi: 10.1590/0001-3765202020191562. PMID: 33053107.
- Liyanagamage DSNK, Jayasinghe S, Attanayake AP, Karunaratne V. Acute and Subchronic Toxicity Profile of a Polyherbal Drug Used in Sri Lankan Traditional Medicine. Evid Based Complement Alternat Med. 2020 Jul 13;2020:2189189. doi: 10.1155/2020/2189189. PMID: 32765627; PMCID: PMC7388592.
- Abdillah S, Gita Lestari A, Monika P, Sumaryono W, Hisyam K. Preliminary analysis of the antidiabetic activity of Indonesian polyherbal formulation. J Nat Remedies. 2021; 21: 219-222.
- Musa AH, Debella A, Gebru G, Makonnen E, Asefa M, Admas A, et al. Subchronic toxicity study of herbal tea of Moringa stenopetala (Baker f.) Cudof. and Mentha spicata L. leaves formulation in Wistar albino rats. Toxicol Rep. 2022; 9: 797–805.
- Jacobson-Kram D, Keller KA, (eds.). Toxicological testing handbook: Principles, applications, and data interpretation, 2nd ed., Informa Healthcare USA, Inc.270 Madison, Avenue New York, NY 2006.
- Organization for Economic Co-Operation and Development (2009) Guidelines for the Testing of Chemicals: Chronic Toxicity Study in Rodents Test No. 452. 2009: 1-10.
- Teh BP, Ahmad N, Ibnu Rasid EN, Zolkifli NA, Sastu Zakaria UR, Mohamed Yusoff N, Zulkapli A, Japri N, Lee JC, Muhammad H. Herbal-Based Formulation Containing Eurycoma longifolia and Labisia pumila Aqueous Extracts: Safe for Consumption? Pharmaceuticals (Basel). 2021 Feb 10;14(2):142. doi: 10.3390/ph14020142. PMID: 33579048; PMCID: PMC7916751.
- Ghayas S, Hannan A, Rizwani GH. Phytochemical, Antioxidant, Toxicological, and Pharmaceutical Evaluation of Polyherbal Formulation: Irochel. Dose Response. 2022 Feb 7;20(1):15593258211073412. doi: 10.1177/15593258211073412. PMID: 35153638; PMCID: PMC8829781.
- Abed AL-Ani, KAM. Primary phytochemical identification and some biochemical parameters study of ethanolic extract of Mentha spicata leaves in mice. J Chem Pharm Res. 2016; 8: 818–822.
- European Union (2010) Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Official Journal of the EU L 276/33. 2010: 1-47.
- Percie du Sert N, Hurst V, Ahluwalia A. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. BMC Vet Res. 2020; 16: 242.
- American Veterinary Medical Association. The AVMA Guidelines for the euthanasia of animals: 2020 Edition. American Veterinary Medical Association. 1931 N, Meacham Road, Schaumburg, IL 60173. Version. 2020; 2020: 22–63.
- Slaoui M, Fiette L. Histopathology procedures: from tissue sampling to histopathological evaluation. Methods Mol Biol. 2011;691:69-82. doi: 10.1007/978-1-60761-849-2_4. PMID: 20972747.
- Suvarna SK, Layton C, Bancroft JD. Bancroft’s theory and practice of histological techniques, 8th ed., Elsevier Limited, Nottingham and Sheffield, UK 2019.
- Nohynek GJ (2002) Presenting toxicology results: How to evaluate data and write reports. Taylor & Francis. 2002: 44-156.
- Bayu F, Afework M, Geleta B, Ergete W, Makonnen E. Effect of Chronic Administration of Aqueous Leaves Extract of Moringa stenopetala on Blood Parameters and Histology of Liver and Kidney in Rats. Ethiop J Health Sci. 2020 Mar;30(2):259-268. doi: 10.4314/ejhs.v30i2.14. PMID: 32165816; PMCID: PMC7060390.
- Everds NE. Evaluation of clinical pathology data: correlating changes with other study data. Toxicol Pathol. 2015 Jan;43(1):90-7. doi: 10.1177/0192623314555340. Epub 2014 Oct 31. PMID: 25361750.
- Moodley JS, Krishna SBN, Pillay K, Sershen G, Patrick G. Green synthesis of silver nanoparticles from Moringa oleifera leaf extracts and its antimicrobial potential. Adv Nat Sci: Nanosci Nanotechnol. 2018; 9: 1-9.
- Li IC, Yang BH, Lin JY, Lin S, Chen CC. Nutritional and 13-Week Subchronic Toxicological Evaluation of Lignosus rhinocerotis Mycelium in Sprague-Dawley Rats. Int J Environ Res Public Health. 2021 Jan 31;18(3):1271. doi: 10.3390/ijerph18031271. PMID: 33572641; PMCID: PMC7908145.
- Olayode OA, Daniyan MO, Olayiwola G. Biochemical, hematological and histopathological evaluation of the toxicity potential of the leaf extract of Stachytarpheta cayennensis in rats. J Tradit Complement Med. 2019 May 16;10(6):544-554. doi: 10.1016/j.jtcme.2019.05.001. PMID: 33134130; PMCID: PMC7588336.
- McInnes EF. Background lesions in laboratory animals: A Color Atlas. Saunders, Elsevier, Edinburgh 2012.
- McInnes EF (ed). Pathology for toxicologists: Principles and practices of laboratory animal pathology for study personnel (1st Ed). Edited by Elizabeth McInnes. Published by John Wiley & Sons Ltd. 2017; 2017: 1-22.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
