International Journal of Pharmaceutical Sciences and Developmental Research
Is it possible to perform molecular hybridization between acetaminophen and nitric oxide donor molecule?
João Gabriel Gouvêa-Silva1,2, Leonardo Ribeiro Bernardo2, Ygor Jessé Ramos1* and Antônio José Calixto de Souza2
2Catholic University of Santos, Santos, SP, Brazil
Cite this as
Gouvêa-Silva JG, Bernardo LR, Ramos YJ, De Souza AJC (2021) Is it possible to perform molecular hybridization between acetaminophen and nitric oxide donor molecule? Int J Pharm Sci Dev Res 7(1): 001-002. DOI: 10.17352/ijpsdr.000033Opinion
Paracetamol (Acetaminophen) belongs to the class of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), having an analgesic, antipyretic and mild anti-inflammatory action due to a weak inhibitory action on cyclooxygenase isoenzymes, COX-1 and COX-2. These isoenzymes are responsible for the synthesis of prostanoids that have several physiological effects, such as: vasodilation; increased renal blood flow; stimulation of gastric protective mucus production; inhibition of gastric acid secretion responsible for reducing acidity in gastric tissue, among other effects. Despite its beneficial effects, the excessive use of NSAIDs is related to several adverse effects, which can alter the homeostasis of the cardiovascular, renal and gastric system [1].
The selective inhibitors of COX-2, known as COXIBS, were an alternative to control pain without having the adverse effects due to the inhibition of COX-1. Although, several reports related cardiovascular complications with long-term use of COXIBS, suggesting that specific benefit/risk ratio should be considered in the prescription of this drug class [2]. Despite the notorious beneficial effects of anti-inflammatories drugs, their mechanism of action creates an inherent risk with its use, bringing the possibility of proposing hybrid chemical structures that can mitigate adverse effects [3].
The Nurse's Health [1] analyzed the effect of NSAIDs on 51,630 normotensive nurses aged between 44 and 69 years, pointing that women who have used acetaminophen frequently had a 20% increase in the risk of developing high blood pressure. In hypertensive patients, the long-term use of this drug class can increase up to 6 mmHg arterial pressure mean, interfering with several classes of antihypertensive drugs [4]. In addition to the cardiovascular adverse effects, the mechanism of action of acetaminophen impairs gastric defense mechanisms, providing gastric lesions.
Nitric Oxide (NO) is an intercellular messenger that plays an important role in pathological and physiological regulations. Its antimicrobial, bactericidal, antiviral, vasodilatory and cytoprotective activity are considered fundamental for physiological homeostasis [5-7]. These beneficial effects makes nitric oxide a potential ally in new drugs developments, making molecular structures capable of donating nitric oxide into study targets, such as those derived from furoxan and benzofuroxan.
Nitric oxide donor molecules were capable of decreasing the gastric membrane damage in a group of rodents submitted to ketoprofen therapy associated with an nitric oxide donor when compared to the group submitted to ketoprofen only therapy [8]. The association of furoxan or benzofuroxan derivatives with other pharmacological groups as a strategy to enhance their pharmacological effect or mitigate adverse effects has been an interesting and actual strategy. There are reports of molecular hybridization with drugs belonging to the class of β-adrenergic antagonists, H2 antagonists, calcium channel modulators and non-steroidal anti-inflammatory drugs [8].
The hybridization of NSAIDs and nitric oxide donors structures have already been proposed with the objective of making safer NSAIDs [9-11]. Acetaminophen and a nitric oxide donor hybrid have been synthesized using an esterification reaction [12] and a study synthesized a hybrid between acetaminophen and a benzofuroxane derivate [13]. Hybridization of diclofenac and benzofuroxan derivatives have already been proposed too [14].
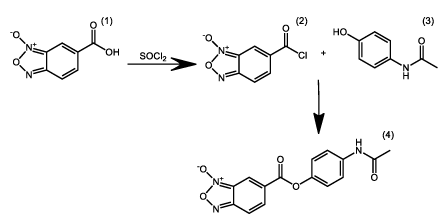
Therefore, it is feasible to believe that the molecular hybridization of paracetamol, N-(4-hydroxyphenyl)acetamide, with benzofuroxan (6-carboxybenzo [c] [1,2,5] oxadiazole 1-oxide (1) would promote a considerable reduction in its adverse effects. The formation of the hybrid (4) would be possible with esterification reactions between the carboxylic acids of acetaminophen (3) and benzofuroxan acyl chloride (2), after submitting it to the reaction with acyl chloride (SOCl2), as shown in Figure 1.
The reaction between carboxylic acids of acetaminophen (3) and benzofuroxan acyl chloride (2) is possible due to the nucleophilic capability of the acetaminophen hydroxyl group to react with the acyl chloride, a powerful acid. The deesterification of the hybrid can be fullfiled by esterases present on the metabolism, allowing both acetaminophen and benzofuroxan to perform their respective roles.
However, increased biochemical markers indicating increase in the oxidative stress in tissues submitted to therapy with nitric oxide donors have already been reported [6]. The association of acetaminophen with nitric oxide donors presented to be less hepatotoxic than acetaminophen isolated [12]. This association didn’t show significant effects on vascular system, despite the nitric oxide vasodilatory effects [12].
The feasibility of synthesizing this hybrid would bring new perspectives on the use of NSAIDs, providing greater safety in pain treatment of patients associated, or not, with other pathologies. The chemistry behind the synthesis of this hybrid is simple, making it achievable. However, the nitric oxide toleration needs to be better understood to grant more reliability and prevent any short or long terms health problems.
Nitric oxide donors may be the solution to prevent the majority cases of acetaminophen intoxications. It is possible to believe that the associations between NSAIDs with nitric oxid donors have the potential to make a revolution in pain treatment, since these associations have shown to be “improved” forms of NSAIDs.
- Dedier J, Stampfer MJ, Hankinson SE, Willett WC, Speizer FE, et al. (2002) Nonnarcotic Analgesic Use and the Risk of Hypertension in US Women. Hypertension 40: 604-608. Link: https://bit.ly/3v72twf
- Fosslien E (2005) Cardiovascular complications of non-steroidal anti-inflammatory drugs. Ann Clin Lab Sci 35: 347-385. Link: http://bit.ly/3v2vZDq
- Sehajpal S, Prasad DN, Singh RK (2018) Prodrugs of Non-steroidal Anti-inflammatory Drugs (NSAIDs): A Long March towards Synthesis of Safer NSAIDs. Mini Rev Med Chem 18: 1199-1219. Link: http://bit.ly/3sSEaQO
- Batlouni M (2010) Anti-Inflamatórios Não Esteroides: Efeitos Cardiovasculares, Cérebro Vasculares e Renais. Arquivos Brasileiros de Cardiologia 94: 556-563. Link: http://bit.ly/30jrBBQ
- Filho RF, Zilberstein B (2000) Óxido nítrico: o simples mensageiro percorrendo a complexidade. Metabolismo, síntese e funções. Revista da Associação Médica Brasileira 46: 265-271. Link: http://bit.ly/3t4WwOR
- Villa AL, Ceneviva R, Viaro F, Ramalho F, Campos AD, et al. (2006) The cytoprotective effect of a nitric oxide donor drug on gastric mucous membrane of rats treated with ketoprofen, a non-steroidal anti-inflammatory drug. Arq Gastroenterol 43: 233-237. Link: http://bit.ly/3v67Qfa
- Kumar S, Singh RK, Bhardwaj TR (2017) Therapeutic role of nitric oxide as emerging molecule. Biomed Pharmacother 85: 182-201. Link: http://bit.ly/3sZOwhO
- Cerecetto H, Porcal W (2005) Pharmacological Properties of Furoxans and Benzofuroxans: Recent Developments. Mini Rev Med Chem 5: 57-71. Link: http://bit.ly/3en3uKL
- Fiorucci S, Antonelli E, Burgaud JL, Morelli A (2001) Nitric oxide-releasing NSAIDs: a review of their current status. Drug Saf 24: 801-811. Link: http://bit.ly/3bqtQJT
- Abdellatif KRA, Abdelall EK, Bakr RB (2017) Nitric Oxide-NASIDS Donor Prodrugs as Hybrid Safe Anti-inflammatory Agents. Curr Top Med Chem 17: 941-955. Link: http://bit.ly/3bnTtej
- Wallace JL (2019) Nitric oxide in the gastrointestinal tract: Opportunities for drug development. Br J Pharmacol 176: 147-154. Link: http://bit.ly/3rm4faF
- Moore PK, Marshall M (2003) Nitric oxide releasing acetaminophen (nitroacetaminophen). Digestive and Liver Disease 35: S49-S60. Link: https://bit.ly/3bozKuZ
- Profire L, Cojocariu A, Oprea AM, Lupusoru CE, Ghiciuc CM, et al. (2010) Synthesis and Characterization of New Nitric Oxide Donor Compounds Based on Theophylline and Paracetamol. Revista de Chimie 61: 1150-1154. Link: https://bit.ly/2OavHKa
- Carvalho PS, Maróstica M, Gambero A, Pedrazzoli JJ (2010) Synthesis and pharmacological characterization of a novel nitric oxide-releasing diclofenac derivative containing a benzofuroxan moiety. Eur J Med Chem 45: 2489-2493. Link: http://bit.ly/3t2vOpJ
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
