International Journal of Pharmaceutical Sciences and Developmental Research
In-silico analysis of tenidap and its derivative as a novel 5-lipoxygenase inhibitor
Asma Noor*, Ardas Masood, Jawaria Khan, Tanveer Abbas and Muhammad Imran Qadir
Cite this as
Noor A, Masood A, Khan J, Abbas T, Qadir MI (2017) In-silico analysis of tenidap and its derivative as a novel 5-lipoxygenase inhibitor. Int J Pharm Sci Dev Res 3(1): 036-038. DOI: 10.17352/ijpsdr.000015Tenidap is a derivative of Flavonoids which are actually plant derivatives, it shows inhibition activity of 5-lipoxygenase. Molecular models were directed to discover molecular docking mode, also to help explain molecular tool behind its inhibitory action. Molecular relations of tenidap with the catalytic trio (His523, His518, Ile875) inside active or dynamic position of 5-lipoxygenase through hydrogen bonding, appears to the major reason elaborate in its substantial 5-lipoxygenase activity of inhibition.
Introduction
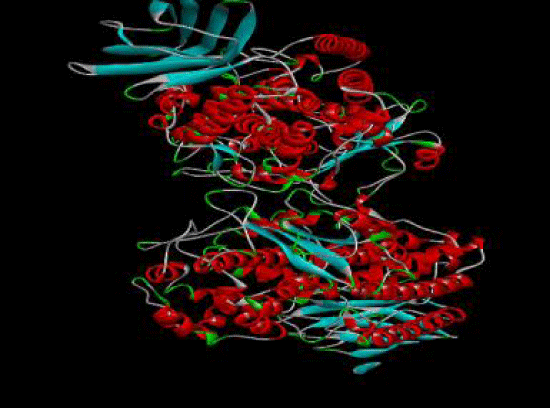
Tenidap is a Flavonoids compound. It is derived from plants of different species e.g. Dodonaea viscose etc. It shows significant inhibitory effect against 5-lipoxygenase. 5- Lipoxygenase or 5-LOX is an enzyme shown in figure 1, responsible for the assembly of inflammatory leukotrienes. 5-lipoxygenase is a lipoxygenase enzyme present in mammals. Lipoxygenases are present mostly in plants or fungi and animals. The desireable substrates comprises polyunsaturated fatty acids carring a serial from double bonds cis. These are important fatty acids in humans also because lipoxygenase belongs to family containing iron having the ability of catilizing polyunsaturated fatty acids into lipids. However, they are not found in bacteria and yeast.
D.viscose plant is investigated experimentally for many pharmacological activities due to its crude extracts which show important activities likes as a local anesthetic, relaxant of smooth muscles, antiseptic, anti-inflammatory, antifungal, anti-ulcerogenic, antiascariasis, vasoconstrictive activity in diverse experimental models (Venkatesh et al., 2008). In this present study a Flavonoids (tenidap) was used with auspicious lipoxygenase activity of inhibition alongside with the computational visions founded on the molecular docking [1]. The main objective of study is to find out new derivatives of Tenidap for the inhibition of 5-lipoxygenase which is involve in inflamatory leukotrienes [2].
Objective
The main objective of current study is an In-silico analysis of tenidap and its derivative as a novel 5-lipoxygenase inhibitor.
Target molecule: 5-lipoxygenase.
Ligand: Tenidap and its derivatives.
Softwares used: During molecular docking of our materials we used following softwares.
1) M.g.l tools software.
2) Autodock vina.
3) Discovery studio.
4) Pymol.
5) Python.
Materials and methods
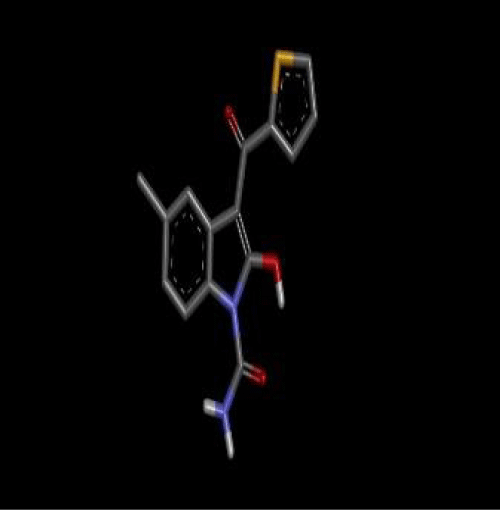
Tenidap pubchem CID (54683953).
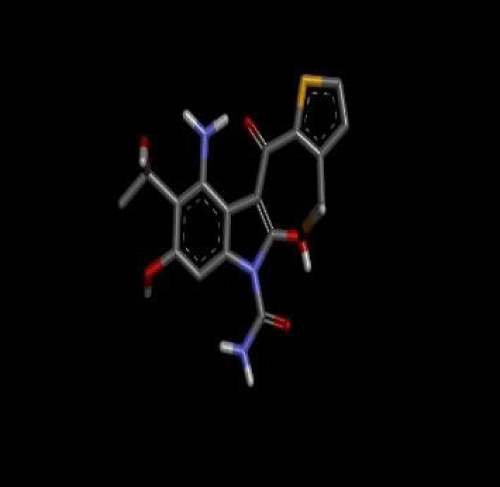
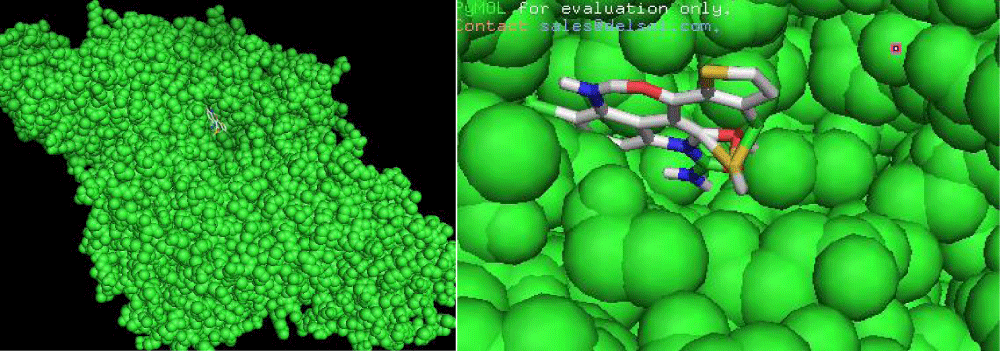
First of all the ligand tenidap shown in figure 2 was downloaded from pubchem.ncbi.org as sdf file. Then it is opened in discovery studio which was already installed. Hydrogen is added to the ligand to fulfill its valency, then further modification was done to the original ligand molecule and it was saved as PDB file. After that this PDB ligand file is opened in Autodock to convert it to PDBQT file to use it in autodock vina [3]. Then target molecule 5-lipoxygenase was downloaded from the RCSB as a text file. This file was opened in discovery studio and ligand which was already present in the protein was selected and deleted to proceed our docking as the already present ligand disturbs the docking with a new ligand 3 shown in figure 3. The file was saved as PDB in discovery studio. Then the protein PDB file was opened in autodock and water was deleted and hydrogen was added to the protein molecule. Then the ligand attachment site was selected by making grid box which covered the protein molecule and the values of dimensions and centers were noted. This protein then saved as PDBQT file. Then ligand and protein (Figure 4) files was copied to the vina folder and the values of grid was put and target and inhibitor names was changed as the names of the files copied. Then the conf file was saved and auto docking was started through opening the command prompt [4]. The docking started after command and the program completed docking itself.
Results and Discussion
The studies show that viscosine was already docked Khan et al (2013). Original result of viscosine which was already used as an inhibitor of lipoxygenase was -8.1kcal/mol shown in table 1. Then I changed the lipoxygenase inhibitor and used many Flavonoids derivatives e.g. Pinocembrin, Curcumin, Dalbergichromene, Mecliphenamat Mecliphenamate and Tenidap.
All these compounds were docked and their ADMET Properties were shown in table 2, but their results were less than the Viscosine even after many times modifications. Among all these compounds Tenidap a Flavonoids derivative it shows -6.6 kcal/mol binding affinity with lipoxygenase. These results were not good because the ligand does not show much binding ability as compared to original inhibitor of lipoxygenase so I modified all the Flavonoids derivatives from which a modified tenidap shows grater binding affinity as compared to its original form and viscosine that is its original inhibitor. The modifications were made through discovery studio. First of all the tenidap was labeled then I changed and replaced atoms randomly.
The changes were H6 to O6, H9 to O9, H10 to C10, H7 to N7, H14 to S14 and H4 to C4.These changes in the structure of ligands were made to study the effect of better binding ability of ligand as compared to original inhibitor, because the main objective of current study is to find out new compounds that can efficiently inhibit 5-lipoxygenase activity. The result of these change are shown in table 3. Each time the hydrogen was added and again all the atoms were labeled. After docking the drug scoring was done online through DSX. The ADMET properties were checked through Medchem Designer software. ADMET properties of both were checked.
- Khan I, Nisar M, Ebad F, Nadeem S, Saeed M, et al. (2009) Anti-inflammatory activities of Sieboldogenin from Smilax china Linn.: experimental and computational studies. Journal of ethnopharmacology 121: 175-177. Link: https://goo.gl/tm1Ey5
- Muhammad A, Anis I, Khan A, Marasini BP, Choudhary MI, et al. (2012) Biologically active C-alkylated flavonoids from Dodonaea viscosa. Archives of pharmacal research 35: 431-436. Link: https://goo.gl/aGfifj
- Coleman RG, Sterling T, Weiss DR (2014) SAMPL4 & DOCK3. 7: lessons for automated docking procedures. Journal of computer-aided molecular design 28: 201-209. Link: https://goo.gl/SX3Fma
- Sandeep G, Nagasree KP, Hanisha M, Kumar MMK (2011) AUDocker LE: A GUI for virtual screening with AUTODOCK Vina. BMC research notes 4: 445. Link: https://goo.gl/Lg25bt
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
