International Journal of Agricultural Science and Food Technology
Nutritional characteristics and phenolic compounds of ripe fruit pulp from six accessions of Mammea americana L.
Armelle Péroumal1, Audrey Vingadassalon1, Genica Lawrence1, Sandra Adenet2, Katia Rochefortd2, Louis Fahrasmane3 and Guylène S Aurore1*
2Pôle Agroalimentaire Régional de Martinique, Habitation Petit Morne n°375, F-97 232 Le Lamentin, France
3INRA, UMR 1270 QUALITROP, Domaine Duclos Prise d’eau, F-97 170 Petit-Bourg, France
Cite this as
Péroumal A, Vingadassalon A, Lawrence G, Adenet S, Rochefortd K, et al. (2022) Nutritional characteristics and phenolic compounds of ripe fruit pulp from six accessions of Mammea americana L. J Agric Sc Food Technol 8(1): 033-037. DOI: 10.17352/2455-815X.000142Copyright License
© 2022 Péroumal A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Mamey apple (Mammea americana) is a tropical fruit largely consumed and more and more coveted as it is a rich source of antioxidants. There is considerable interest in fruits phenolic compounds due to their different characteristics and particularly to their role in protection against cardiovascular diseases and cancers through their antioxidant activity.
Nutritional characteristics were determined in six mamey apple fruit accessions: Sonson, Pavé 11, Lézarde, Ti Jacques, Escouët and Antonio. The pulps of the ripe fruits were analyzed for moisture, ash, total lipid, total protein, and carbohydrate contents.
This study represents a detailed report of phenolic compounds in mamey apple pulp. The metabolite profiling associated with the antioxidant potential represents an important step to the bioactive molecules characterization. The results showed that the accessions studied contained three main classes of phenolic compounds: flavan-3-ols, phenolic acids and flavonols. Those phenolic compounds characterization was then carried out first using liquid chromatography (HPLC) coupled to Diode Array Detector (DAD). Finally, spectral characteristics and fragmentations were acquired by Mass Spectrometry (MS) using the Electrospray Ionization Interface (ESI). All the identified compounds are interesting metabolites known for their biological properties and nutritional benefits
Introduction
Mamey apple (Mammea americana) is a fruit with a reddish yellow color, aromatic, and edible pulp, popularly known as “abricot-pays” in the West Indies. This fruit has an important agro-industrial potential and this pulp can be used in salad dressing, processed into jam or beverages. Few researches dealing with the pulp composition of Mammea americana fruits has been reported in the literature. The phytochemical study of the fruits focused on the composition of primary metabolites such as proteins, fats and carbohydrates. It also showed the presence, mainly of carotenoids, vitamin C, coumarins and xanthones, known for their biological properties, in particular their antioxidant property [1-5].
Mammea americanaL. (Clusiaceae family) adult trees seasonally bear a few hundred large pomes. Mamey apple fruits were generally elliptical, presenting a ratio of equatorial circumference to longitudinal circumference of close to 1 with a diameter of 7-20cm, or sometimes egg-shaped [6]. The average weights of the fruits ranged from 560g to 1100g (± 43%). The fruit is protected by a thick skin with a leathery appearance, making up 20%-27% of the whole fruit weight. The fruits of this species contain one to four rough seeds, bitter to resinous, representing 7%-21% of the whole fruit weight. The fruit pulp is adhesive to the seeds, according to the accession. The fruit texture in the hand was rather firm, ranging from 45 to 64 N, comparable to the apple (57N).
The whole fruit weight contained around 50% to 60% of pulp. The color of the mamey fruit pulp is dominated by a yellow to orange hue. Apart from these phenotypic characteristics, few publications described the content of bioactive compounds in Mammea americana such as total phenolic compounds and total flavonoids [6] and coumarins [7].
Ripe mamey fruits are highly perishable, as it is a climacteric fruit. At 27°C, its respiration rate and ethylene production are respectively 14 to 20μL CO2 /kg/h and 408μL/kg/h [8]. Fruits harvested ripe green can ripen naturally in three to four days after harvesting. Storage of fruit harvested at the mature green stage at 15 ±2°C under a controlled atmosphere (5.1% CO2, 5.6% O2, 89.3% N2) for two weeks delays the ripening process 9. The extension of shelf life is an interesting factor for the sale or export of good quality fruit.
The fruit pulp is an excellent source of carotenoids and some essential vitamins and minerals [5,10,11]. Many secondary metabolites such as flavonoids, triterpenoids and coumarins have been identified in different parts of the tree [7] and fifty-one constituents of mamey apple pulp, free and glycosidically bound volatiles, were also describes [12]. Many parts of this tree are used in traditional medicine in Central America and the West Indies for the prevention and treatment of a wide number of health disorders such as rheumatism, tissue inflammation, indigestion, coughs and for blood pressure reduction [13-15]. The main traditional non-food use known is as a crop insecticide [13,16-18].
In this study we described the nutritional value and phenolic composition of the pulp of six mamey apple (M. americana) accessions using liquid chromatography coupled to Diode Array Detector (DAD). Then, spectral characteristics and fragmentations were acquired by Mass Spectrometry (MS) using the electrospray Ionization Interface (ESI) analysis. This analytical method has been described for the profiling, identification and simultaneous quantification of chemical constituents in plants [4,19] and has been used in different previous studies [7,20]. This study represents a detailed report of phenolic compounds in mamey apple pulp.
Materials and methods
Plant material collection and preparation
Six mamey apple (Mammea americana L.) accessions : Sonson, Pavé 11, Lézarde, Ti Jacques, Escouët, Antonio, bearing fruits deemed to be of superior pomological and organoleptic quality, collected in Martinique by CIRAD botanists, were the source of our samples for all the experiments in this study [2]. Each accession comes from vegetative multiplication by grafting. Ripe fruits were obtained from five-year-old trees. All the trees of the studied accessions were grown on the same site, the experimental station Rivière Lézarde in Martinique. In addition, grafted accessions have the advantage of being small trees with fruit within reach, which facilitated their harvesting. For each mamey apple accession 30 fruits were randomly collected during February - July. The peels, pulp and seeds were manually separated from every fruit obtained for each mamey apple accession. The pulp was cut into pieces, immediately frozen in liquid nitrogen and stored at -80°C until chemical analyses. The samples were kept at 4°C overnight after removing them from -80°C before analysis. For the characterization of phenolic compounds, frozen pulp (-80°C) was lyophilized (72h, -80°C, 1 mbar) and reduced to powder. All the experiments were performed in triplicate.
Reagents and chemicals
Chemical analyses were carried out on the pulp of the 30 ripe fruits collected for each accession studied. The reagents used for our nutritional experiments were acquired from Sigma-Aldrich Co. (Germany). All solvents used for extraction and chromatography were of analytical purity and were purchased from Fisher Scientific (Illkirch, France). The toluene-α-thiol came from Merck (Germany). The water used to prepare the mobile phase of specific polyphenol analyses was high purity water (18MΩ) obtained using a milli-Q water system (United States). Chemical standards used to identify and quantify polyphenolic compounds were acquired from Sigma-Aldrich Co. (Germany) and Extrasynthese SA (France).
The distilled water used for the other analyses came from a bi-distillatory system (Autostill, D-Ionstill).
Moisture content
Fresh pulp samples (2g of crushed, homogenized pulp) were collected from each mamey fruit for moisture determination in triplicate, using a ventilated oven at 70°C for 5h at reduced pressure (−1 bar). The moisture content was evaluated by the direct drying method as per 21 with some modifications. The homogenized pulp sample (2g) was dried overnight in an air-oven set at 70°C until constant sample weight was obtained. The moisture content was defined by the difference between initial weight and constant weight after drying.
Ash content
Sample ash content was calculated from a crushed fresh pulp sample (2g) following heating to 525±25°C for 5h as per the AOAC official method 923.03 (1996).
Lipid content
Lipids were extracted from the sample (2g) after acid hydrolysis with 50ml of 8N HCl, at 80°C. The mixture was filtered and rinsed with boiling water until neutral pH. The filters were dried. The residues were placed in glass cartridges with 140ml petroleum benzene and some pumice stones to extract the lipids in a Soxtherm extractor (Gerhardt Laboratory Systems, Königswinter, Germany). After extraction, the cartridges were oven dried at 101°C. The amounts of lipids were determined by different weighing.
Protein content
The protein content was determined through the quantification of total nitrogen using Kjeldahl’s method. After mineralization of the sample (2g) in 25ml of 95% H2SO4 in the presence of a catalyst in a Turbotherm mineralizator (Gerhardt Laboratory Systems, Königswinter, Germany). The mineralizate was distilled, in the presence of soda, in a Vapodest distiller (Gerhardt Laboratory Systems, Königswinter, Germany). The distillate was collected in 40ml of boric acid 40g l−1 in the presence of a few drops of Tashiro’s indicator. The distillate was titrated by hydrochloric acid HCl (0.1N).
Carbohydrate content
This was obtained by subtraction of the amount of other constituents from the dry extract (dry extract − (ash + lipids + proteins)). Total Carbohydrate (TC) represents the total fiber, starch and sugars content.
Energy Value (EV)
This is the amount of energy provided by all the Total Protein (TP), Total Lipid (TL) and Total Carbohydrate (TC) values previously defined, expressed in kilocalories or kilojoules per 100g of fresh material. It was obtained by the following formulas: EV (kcal/100g) = 9 TL+4 TC+4 PT or EV (kJ/100g) = 37TL+17TC+17PT.
Phenolic compounds of mamey apple pulp characterization
No information was available on the characterization of phenolic compounds of mamey apple pulp. Our study focused on identification and quantification of these compounds present as monomers and polymers. A methodology based on combination of chromatography method (HPLC) and mass spectroscopy (HPLC-MS-ESI) was applied.
Monomers characterization
Method of extraction: To 400mg of lyophilized pulp powder sample, weighed in an Eppendorf tube, were added 1200μL methanol: glacial acetic acid (99: 1; v/v). The assembly was vortexed and subjected to ultrasound for 15 minutes at room temperature (30°C). The mixture was centrifuged at 10 000rpm for 15 minutes. After filtration (PTFE 0.45μm), the sample was injected (20μL) into the UFLC chain. The analyses were performed twice.
Liquid chromatographic conditions: Qualitative and quantitative analyses of lyophilized mamey apple pulp extracts were carried out using an Ultra-Fast Liquid Chromatography (UFLC) Shimadzu Prominence (Kyoto, Japan) analytical chain comprising the following elements: - two LC-20AD Prominence pumps, - an analytical column Lichrocart Lichrospher PR-18 (250mmx4mm, 5μm) (Merck, Darmstadt, Germany) equipped with a pre-column Lichrospher PR-18 (5μm) (Merck, Darmstadt, Germany), a DGU-20A5 Prominence degasser, - SIL-20ACHT Prominence automatic sample changer, a SPD-M20A Prominence Diode Array Detector (DAD), and a LC Solution software acquisition system (Shimadzu, Kyoto, Japan).
The compounds to be separated were eluted by a concentration gradient consisting of:
- Solvent A: water: acetic acid (97.5: 2.5; v/v)
- Solvent B: Acetonitrile.
The gradient elution program used for the LC analysis was 60 minutes long, during which the percentage of solvent A (%) and solvent B (%) evolved according to a timing (m minutes) in accordance with the following triplets (%A, %B, m): (97, 3, 0); (91, 9, 5); (84, 16, 15); (50, 50, 45); (10, 90, 48); (10, 90, 52); (97, 3, 55); (97, 3, 60).
The flow rate run was 1 ml/min, the injection volume was 20μL, the column temperature was 30°C and the detection range was between 200 and 800nm. The samples and standard solutions were maintained at 4°C before injection. The analyses were carried out at three different wavelengths: 280nm for flavan-3-ols monomers and thioether derivatives, 320nm for hydroxycinnamic acids and 350nm for flavonols. Standard 4g/l solutions were prepared and injected into the UFLC chain under the conditions described above. The chromatogram peaks obtained for the different extracts were identified by comparing their retention time and their UV-visible spectrum with those of the analytical standards. The compounds were quantified, according to an external calibration, by integration of the peaks of the chromatogram obtained.
To obtain additional information and to ensure that there was no co-elution phenomenon concerning the identified phenolic compounds, we used an Acquity Ultra Performance LC™ (UPLCTM) device from Waters, coupled with a Brucker Daltonics mass spectrometer (Bremen, Germany) with an electrospray ionization source (ESI) used in negative mode (UPLC-MS-ESI). The characteristics of the analytical column were the same as those mentioned above. The mobile phase consisted of water: formic acid (99.95: 0.05; v/v) (solvent A) and acetonitrile (solvent B). The gradient elution program was like the one used for UFLC-DAD analysis. The flow rate run was 1ml/min and the sample injection volume was 2μL. The analyses were performed at 280nm, 320nm and 350nm. Mass spectra were generated in ultra-scan mode with m/z ratios of between 100 and 900. Nitrogen was used as a nebulizer gas. The data was collected and processed by a Bruker Compass Data Analysis software acquisition system. Compounds identification was based on the analysis of retention times and fragmentations acquired by mass spectrometry.
Polymer characterization by thiolysis (or thioacidolysis) reaction
400mg of lyophilized pulp were weighed in an Eppendorf tube and 400μL of anhydrous methanol acidified with hydrochloric acid (3N) and 800μL of a 5% (v/v) toluene-α-thiol solution prepared in anhydrous methanol added. The mixture was vortexed and then raised to 40°C for 30 minutes stirring every 5 minutes. The thiolysis reaction was stopped by lowering the temperature in crushed ice for at least 5 minutes.
After filtration (PTFE 0.45μm), the reaction mixture (20μL) was extemporaneously injected into the UFLC chain. The analyses were repeated twice. Polymeric phenolic compounds were identified and quantified after thioacidolysis by UFLC-DAD then by UPLC-MS-ESI under the same conditions described above.
Statistical analysis
The statistical analysis was computed using Statgraphics centurion 15.2.06 (Statpoint technologies Inc., Virginia, USA). The data were analyzed for significant differences (P≤0.05) by means of analysis of variance (ANOVA). To identify samples that differ from each other, Duncan’s multiple comparison tests was conducted.
Results
Nutritional characteristics of mamey apple accessions
Macronutrients: Water was the main component of mamey apple pulp (Table 1). The average moisture content was 85.95%. Among the accessions we studied, we found that Sonson produced the fruit with the highest water content (88%) while Ti Jacques produced the fruit with the lowest water content (84%). The ash content was on average of 0.18 g/100g, but highly variable, because Escouët pulp contained the highest ash content (0.22g100g), and was therefore the richest in mineral compounds, while Sonson pulp with half this amount (0.11g/100g) had the lowest ash content.
The six accessions studied had low contents of protein (about 0.40g/100g) and lipid (<0.20g/100g). There were no statistically significant differences between the levels for these compounds in the accessions studied. The measured protein contents were lower than those established for other tropical fruits and vegetables such as papaya (0.83g/100g), carambola (0.9g/100g), dessert banana (1.1g/100g), breadfruit (1.12g/100g) or cinnamon apple (1.22g/100g) [22-25]. Indeed, edible fruits generally have low protein (less than 5g/100g) and lipid (less than 2g/100g) contents.
Total carbohydrates were quantitatively the second component of mamey apple pulp. The accessions we studied exhibited on average a variation range of total carbohydrates of between 12 and 16g/100g. Mamey apple pulp is consumed as a dessert fruit because of its sweet taste.
The nutritional data we obtained are like the few data found in the literature [26,27], except for the total carbohydrate content and therefore the energy value which are higher. The Pavé 11, Lézarde and Ti Jacques accessions bore fruits with their pulp richer in total carbohydrates (between 13 and 16g/100g) and were the most energy-rich (between 56 and 65 kcal/100g). This finding leads us to suppose that these differences with the data in the literature may be due to several factors: the singularity of the accessions studied the pedoclimatic effects, post-harvest changes and/or the more or less advanced stage of maturity of the fruits [27].
Phenolic nutrients: The significant differences between accessions in amounts of total phenolic compounds observed were probably due to their genotype because they developed under the same ecological conditions and according to the same cultivation method (Table 1).
Total phenolic compounds and total flavonoids contents in mamey apple pulp have already been established [6]. The results of a main component analysis and a hierarchical ascending classification of the six accessions studied, had shown that Pavé 11 and Lézarde accessions were in the same homogeneous group. They would therefore be grouped in the same set (Pavé 11-Lézarde) for the characterization of phenolic compounds.
Preliminary considerations: The composition of the extraction solvent is a parameter that can strongly influence the efficiency of extraction, as well as the resolution of chromatographic peaks. Methanolic, acetonic, ethanolic or waterless solvents are the most used [28]. We therefore set up preliminary analyses to determine the most suitable solvent for chromatographic analysis of our samples. Two solvents of different natures were tested:
- A methanol: acetic acid solvent (99: 1; v/v) (A)
- An acetone: water: acetic acid solvent (70: 29: 1; v/v/v) (B).
We observed that the chromatographic peaks of methanol extracts (A) were well resolved at the different wavelengths analyzed. However, the elution profile of the hydroacetone extracts (B) at 280nm, 320nm and 350nm showed poor chromatographic peak separation, making qualitative and quantitative analysis relatively difficult. Indeed, the chromatograms were characterized by an increase in the baseline over most of the profile, particularly at 280nm. The total phenolic compounds content was determined according to the Folin-Ciocalteu method, on both extracts according to the protocol described previously [6]. The assay was repeated three times and the results were expressed in mg gallic acid equivalent per 100g of fresh material. We noted that the total phenolics content of the hydroacetone extract (118.26mg/100gF.W.) was at least three times higher than that of the methanol extract (34.72mg/100g F.W.). From a quantitative point of view, phenolics compounds are therefore better extracted in hydroacetone solvent. The objectives of our study being not only quantitative but also qualitative, we retained the methanol solvent giving a better resolution of the chromatographic peaks for HPLC analysis of the extracts.
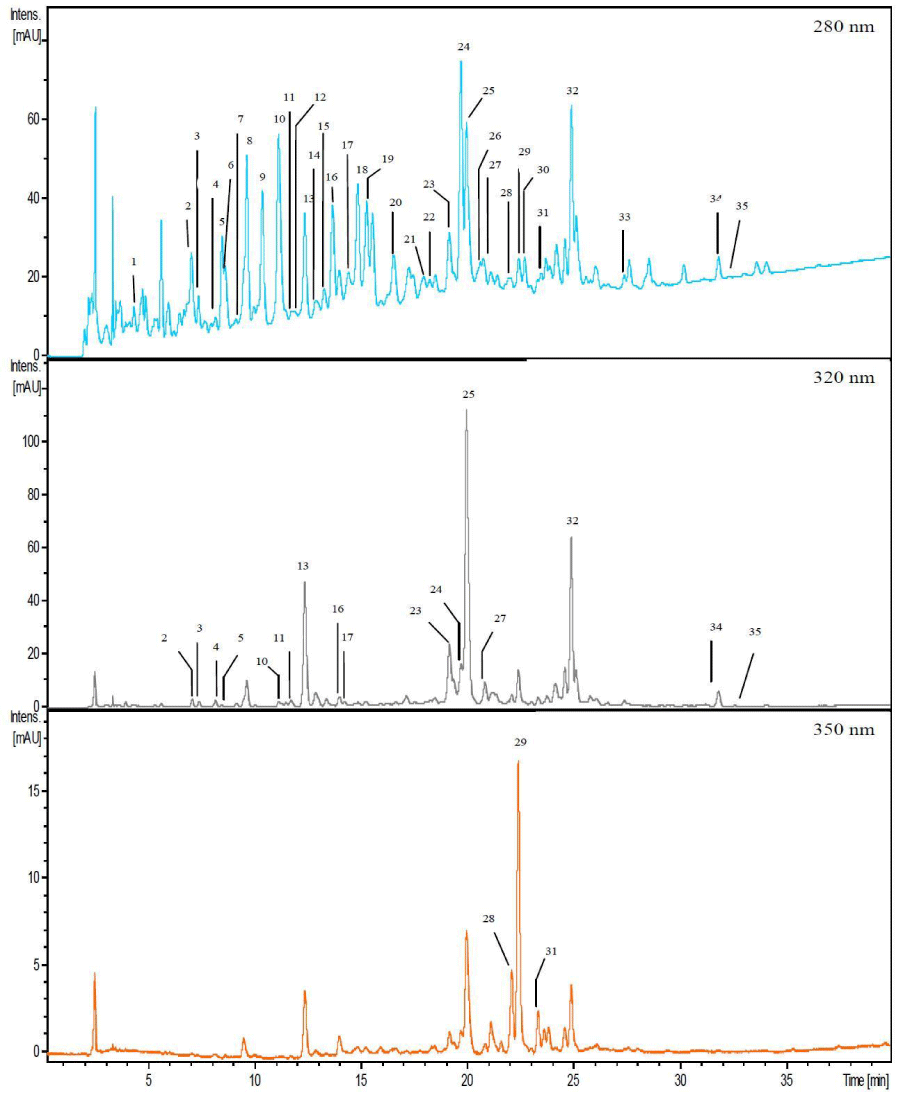
Qualitative analyses of crude phenolic compounds:LC analysis: the identification of the compounds present in the pulp extracts of the different accessions is based on the comparison of their absorption spectrum and their retention time with those of the standards analyzed under the same conditions. There was a great similarity in phenolics composition of the different accessions. Given this great similarity, only the elution profile of the Pavé 11-Lézarde accession is presented (Figure 1). The chromatographic profile of the compounds from the extracts was monitored at 280nm, the characteristic wavelength of phenolic compounds. Indeed, the phenolic cycle is characterized by a maximum absorption peak at 280nm. The B cycle of flavones and flavonols is generally characterized by an absorption peak in the range 300nm to 380nm. The hydroxycinnamic acids have a maximum absorption peak at 320nm. This is the reason why chromatograms at characteristic wavelengths of 320nm and 350nm were also analyzed. There were seven characterized compounds grouped into three classes of monomeric phenolic compounds according to the profiles and absorption maxima of the UFLC-DAD spectra:
- First class: Flavan-3-ol (Characterization wavelength, 280nm)
- (-)-epicatechin a polar compound (1) Retention time: 15.2 min. - Second class: Hydroxycinnamic acids (Characterization wavelength, 320nm)
- Hexoside derivative of sinapic acid (2) Retention time: 19.9 min.
- Hydroxycinnamic acid, unidentified (3) Retention time: 24.8 min.
- Hydroxycinnamic acid, unidentified 4) Retention time: 31.5 min.
- Hydroxycinnamic acid, unidentified (5) Retent (ion time: 32.28 min. - Third class: Flavonols (quantification wavelength, 350nm)
- Quercetin-3-O-galactoside (6) Retention time: 22.0 min.
- Quercetin-3-O-glucoside (7) Retention time: 22.4 min.
The same compounds are observed for the other samples (Sonson, Escouët, Antonio and Ti Jacques) with relative differences in peak intensity. Hydroxycinnamic acids, found in our samples, are a majority class of phenolic compounds. They are widespread in the plant kingdom and known for their significant biological effects [29,30]. However, three other compounds such as (-)-epicatechin, quercetin-3-O-galactoside and quercetin-3-O-glucoside were clearly identified in our extracts. They are also found in apples, red grapes, apricots and mangos [31-35]. As for anthocyanins, the analysis of chromatograms obtained at 520nm revealed that these compounds were absent from our samples.
In order to confirm the identified compounds (1, 2, 6 and 7), and try to identify molecules 3, 4 and 5 and to characterize the other chromatographic peaks, we decided to associate UFLC-DAD method and UPLC coupled with mass spectrometry (UPLC-MS-ESI).
UPLC-MS-ESI analyses:After analysis of retention times, spectral characteristics and fragmentations acquired by UPLC-MS-ESI, we identified or attempted to identify 33 compounds in mamey apple pulp extracts.
The analytical data recorded by MS for each chromatographic peak are presented in Table 2.
The compounds identified in mamey pulp extract by UPLC-MS analysis are reported in the Figure 2.
Compounds 1 to 33 presented in Table 2.
The phenolic classes found in our samples are cited below:
- First class: flavan-3-ols including one monomeric compound and 6 dimers or trimers subform (peaks n°8, 14, 15, 18, 20, 21, 33),
- Second class: 10 glycosylated hydroxycinnamic acids and some derivatives (peaks n° 4, 10, 11, 12, 13, 16, 23, 25, 26, 32),
- 6 glycosylated hydroxybenzoic acids and some derivatives (peaks n° 2, 3, 5, 9, 17, 24), - Third class: 3 glycosylated flavonols (peaks n° 28, 29, 31).
Phenolic acids represent one third of dietary phenols. They are generally found in almost all plants in free form or bound to a sugar molecule or by an ether, ester or acetal type bond to another molecule. These compounds are of growing interest and are increasingly studied for their antioxidant activity [34]. We observed that the phenolic acids characterizing our extracts were generally glycosylated. On the other hand, caffeic acid is the major representative compound of hydroxycinnamic acids. It is found in foods mainly as chlorogenic acid (or 5-caffeoylquinic acid) [29]. In the case of our samples, it was present only in glycosylated form. In addition, the analyses performed by coupling UFLC with MS made it possible to:
‒ Confirm the presence of a hexoside derivative of sinapic acid (peak n° .25) which would be bound to a compound characterized by an m/z ratio of 216,
‒ To highlight the presence of sinapic acid hexoside (peak n° 32).
The other two hydroxycinnamic acids detected by UFLC-DAD previously, at retention times of 31.5 and 32.28 minutes (peak n° 34 and 35 in Figure 2), could not be identified by UFLC-MS. On the other hand, we noticed that for retention times between 19 and 20.5 min, we obtained many phenolic acids bound to a compound with an m/z ratio of 216.
Concerning the flavan-3-ols class, we clearly identified (-)-epicatechin, very often present in fruits such as grapes and in tea [20,36].
Flavonols are generally represented in fruits by myricetin, quercetin and kaempferol. Quercetin, present in many fruits, vegetables and drinks, is the majority flavonol in our diet [29]. Two of the detected flavonols (peaks n° 28 and 29) were clearly characterized: quercetin-3-O-galactoside and the quercetin-3-O-glucoside. We also detected a quercetin linked to a pentose (peak n° 31).
Based on the fragmentation patterns and spectral characteristics reported in the literature, we have attempted to propose structures for compounds 1 and 3 (Table 2). According to Liu and collaborators [19], hydrated meranzin, belonging to the coumarin family, has the same fragmentation pattern as compound no.1 contained in our samples (m/z[m-H]-= 277 and m/z MS2= 233). Based on the fragmentation scheme obtained by Perestrelo and coworkers [20], compound no.3 would be a protocatechuic acid bound to a glucose molecule (m/z[m-H]-= 315 and m/z MS2= 153).
Quantitative analyses of crude phenolic compounds: The results of quantitative analyses of monomeric phenolic compounds are given in Table 3. Not all compounds were quantified because there are no commercial standards.
Among the 7 quantified monomeric phenolic compounds, we observed that (-)-epicatechin (peak n° 18), followed by the sinapic acid derivative hexoside (peak n° 25) were the majority compounds in the pulp extracts. Antonio and Ti Jacques gave the extracts containing the highest concentrations of (-)-epicatechin (2.47mg/100g F.W.) and sinapic acid hexoside derivative (2.25mg/100g F.W.) respectively. (-)-epicatechin is found in greater amounts in apples (golden) at 5.9mg/100g F.W [31]. Among the 2 quantified flavonols, isoquercitrin (peak n° 29) is the majority compound with values three times higher than those of hyperoside (peak n° 28). Only the pulp from Antonio and Sonson stood out with very low concentrations of isoquercitrin compared to other accessions.
The results of the quantitative analyses of dimeric and trimeric procyanidins identified in our samples are presented in Table 4.
Among the procyanidins detected, trimer C1 was the majority compound in pulp extracts. Only pulp from Pavé11-Lézarde had content about seven times lower (0.50mg/100g F.W.) than the other accessions. Concerning the dimers identified, dimer B1 was the most abundant. We observed that the pulp from Pavé 11-Lézarde was the richest in dimers B1, B2 and B5. Apple (golden) contains much higher levels of procyanidins B2 (7.2mg/100g F.W.) than our samples [31].
The chromatograms obtained by UFLC-DAD at 280nm and 360nm after thiolysis showed great similarity in the composition of the different samples. Regarding the compounds identified and quantified after the thiolysis reaction, we observed that the procyanidins contained in our samples corresponded to catechin and epicatechin polymers without galloylation. (+)-catechin was found only as a terminal unit whereas (-)-epicatechin could constitute both an extension unit and a terminal unit. Guyot and coworkers [31] showed that is also the case for apples (Golden). The results of the analyses revealed that Pavé11-Lézarde followed by Ti Jacques had the highest concentrations of (-)-epicatechin benzylthioether. In addition, we observed that according to the accessions, the DPn (average polymerization degree) varied from 18 to 33. Pavé 11-Lézarde stood out with the highest DPn (33). The DPn for the other accessions were between 18 and 25.
The procyanidins contained in the mamey apple pulp therefore consisted primarily of (-)-epicatechin, with an average DP of about 24. This value is four times higher than that of the apple (Golden), which indicates that our samples contain a higher concentration of oligomeric procyanidins.
Literature review on the mamey apple showed that no information was available on the characterization of phenolic compounds in the fruit pulp. Our study therefore focused on the identification and quantification of these compounds present in monomer and polymer form. Our results showed that our 5 extracts contain three main classes of phenolic compounds:
- Flavan-3-ols in the form of monomers, dimers and trimers,
- Phenolic acids (hydroxycinnamic acids and hydroxybenzoic acids),
- Flavonols.
These are compounds naturally present in many fruits and known for their important biological effects. Of the accessions studied, the pulp from Pavé11-Lézarde stood out with the highest content in dimers B1, B2 and B5 and the highest degree of polymerization (33.0). Antonio and Ti Jacques also stood out with extracts characterized by higher concentrations of (-)-epicatechin and hexoside derivatives of sinapic acid, respectively.
The complexity of the extracts and the absence of information in the literature on phenolic compounds in the fruit pulp prevented us from identifying all the compounds detected. Thus, subsequent studies are to be carried out using other analysis tools (LC/MS, RMN) to:
‒ Confirm the results obtained,
‒ Undertake more in-depth structural elucidations,
‒ Allow the localization and determination of the sugars present.
Conclusion
The present study focused on the identification and quantification of phenolic compounds present in monomer and polymer form in the fruit pulp. We applied a methodology essentially based on the combination of chromatography liquid methods (UFLC-DAD and UPLC-MS-ESI).
We focused on identifying bioactive compounds and quantifying phenolic compounds present in monomer and polymer form, using UFLC-DAD and UPLC-MS analyses. The results showed that the accessions studied contained three main classes of phenolic compounds: flavan-3-ols in the form of monomers ((-)-epicatechin), and of polymers procyanidins (dimers (B1, B2, and B5) and trimer (C1)), phenolic acids (hydroxycinnamic acids and hydroxybenzoic acids) and flavonols, all known for beneficial effects on health. The procyanidins in our samples were catechin and epicatechin polymers. (+)-catechin was found only as a terminal unit whereas (-)-epicatechin could constitute both an extension unit and a terminal unit. Also, procyanidins in mamey apple pulp had an average DPn of about 24.
Among the accessions studied, Pavé 11-Lézarde fruit stood out with pulps containing higher concentrations of dimers B1, B2 and B5 and with the highest degree of polymerization (DP= 33). Antonio and Ti Jacques also stood out with higher levels of (-)-epicatechin and hexoside derivatives of sinapic acid respectively.
The authors would like to thank the Territorial Community of Martinique, as well as the European Funds (FEADER) for the funding granted to this research work, and the Agricultural Research Center of Development (CIRAD) for providing fruit samples.
- Lim YY, Lim TT, Tee JJ (2007) Antioxidant Properties of Several Tropical Fruits: A Comparative Study. Food Chem 103: 1003-1008. Link: https://bit.ly/3fEqjJe
- Gervais L, Lavigne C (2007) Mamey ( Mammea Americana L.) in Martinique Island: An Inheritance to Be Developed. Fruits 62: 237–246. Link: https://bit.ly/3rvtGYz
- Du L, Mahdi F, Jekabsons MB, Nagle DG, Zhou YD (2011) Natural and Semisynthetic Mammea-Type Isoprenylated Dihydroxycoumarins Uncouple Cellular Respiration. J Nat Prod 74: 240–248. Link: https://bit.ly/3tFvy3J
- Lima LGB, Montenegro J, Abreu JP, de Santos MCB, Nascimento TP, et al. (2050) Metabolite Profiling by UPLC-MSE, NMR, and Antioxidant Properties of Amazonian Fruits: Mamey Apple (Mammea Americana), Camapu (Physalis Angulata), and Uxi (Endopleura Uchi). Molecules 25: 342. Link: https://bit.ly/3qFD9xn
- Lemus C, Smith-Ravin J, Marcelin O (2021) Mammea Americana: A Review of Traditional Uses, Phytochemistry and Biological Activities. J Herb Med 29: 100466. Link: https://bit.ly/3GGzNiZ
- Péroumal A, Adenet S, Rochefort K, Fahrasmane L, Aurore G (2017) Variability of Traits and Bioactive Compounds in the Fruit and Pulp of Six Mamey Apple (Mammea Americana L.) Accessions. Food Chem 234: 269–275. Link: https://bit.ly/3FTcIc1
- Yang H, Jiang B, Reynertson KA, Basile MJ, Kennelly EJ (2006) Comparative Analyses of Bioactive Mammea Coumarins from Seven Parts of Mammea Americana by HPLC-PDA with LC-MS. J Agric Food Chem 54: 4114–4120. Link: https://bit.ly/3FGiYUh
- Yahia EM, Guttierrez-Orozco F (2011) Mamey Apple (Mammea Americana L.). In Postharvest Biology and Technology of Tropical and Subtropical Fruits; Yahia, E. M., Ed.; Woodhead Publishing 474–482e. Link: https://bit.ly/3nFLPlg
- Manzano-Mendez JE, Dris R (2001) Effect of Storage Atmosphere and Temperature on Soluble Solid Mamey Amarillo (Mammea Americana L.) Fruits. Acta Hortic 675–676. Link: https://bit.ly/3FBgWVf
- de Rosso VV, Mercadante AZ (2007) Identification and Quantification of Carotenoids, by HPLC-PDA-MS/MS, from Amazonian Fruits. J Agric Food Chem 55: 5062–5072. Link: https://bit.ly/3Kohdyz
- ordoñez-santos L, Martínez-Álvarez G, Riascos A (2014) Effect of Processing on the Physicochemical and Sensory Properties of Mammee Apple (Mammea Americana L.) Fruit. Agrociencia 48: 377–385. Link: https://bit.ly/3Ko40G1
- Morales AL, Duque C (2002) Free and Glycosidically Bound Volatiles in the Mammee Apple (Mammea Americana) Fruit. Eur Food Res Technol 215: 221–226. Link: https://bit.ly/33SPMMI
- Longuefosse JL (2008) Plantes Médicinales Caribéennes: 2; Orphie. 2.
- Meléndez PA, Capriles VA (2002) Molluscicidal Activity of Plants from Puerto Rico. Ann Trop Med Parasitol 96: 209–218. Link: https://bit.ly/3qHLT6e
- Toma W, Hiruma-Lima CA, Guerrero RO, Brito AR (2005) Preliminary Studies of Mammea Americana L. (Guttiferae) Bark/Latex Extract Point to an Effective Antiulcer Effect on Gastric Ulcer Models in Mice. Phytomedicine 12: 345–350. Link: https://bit.ly/32fgYVo
- Reyes-Chilpa R, Estrada-Muñiz E, Vega-Avila E, Abe F, Kinjo J, et al. (2008) Trypanocidal Constituents in Plants: 7. Mammea-Type Coumarins. Mem Inst Oswaldo Cruz 103: 431–436. Link: https://bit.ly/3fECHcu
- Boulogne I, Germosen-Robineau L, Ozier-Lafontaine H, Jacoby-Koaly C, Aurela L, et al. (2012) Acromyrmex Octospinosus (Hymenoptera: Formicidae) Management. Part 1: Effects of TRAMIL’s Insecticidal Plant Extracts. Pest Manag Sci 68: 313–320. Link: https://bit.ly/3qGqjii
- Robineau L (2014) Pharmacopée Végétale Caribéenne; Réseau Canopé.
- Liu WY, Zhou C, Yan CM, Xie SL, Feng F, et al. (2012) Characterization and Simultaneous Quantification of Multiple Constituents in Aurantii Fructus Immaturus Extracts by HPLC-DAD-ESI-MS/MS. Chin J Nat Med 10: 456–463. Link: https://bit.ly/3nFKtqW
- Perestrelo R, Lu Y, Santos SAO, Silvestre AJD, Neto CP, et al. (2012) Phenolic Profile of Sercial and Tinta Negra Vitis Vinifera L. Grape Skins by HPLC–DAD–ESI-MSn: Novel Phenolic Compounds in Vitis Vinifera L. Grape. Food Chem 135: 94–104. Link: https://bit.ly/3qDkiD6
- Chew LY, Prasad KN, Amin I, Azrina A, Lau CY (2011) Nutritional Composition and Antioxidant Properties of Canarium Odontophyllum Miq. (Dabai) Fruits. J Food Compost Anal 24: 670–677. Link: https://bit.ly/3nC98wg
- Tripathi S, Suzuki JY, Carr JB, McQuate GT, Ferreira SA, et al. (2011) Nutritional Composition of Rainbow Papaya, the First Commercialized Transgenic Fruit Crop. J Food Compost Anal 24: 140–147. Link: https://bit.ly/3AkLOZm
- Clerici MTPS, Carvalho-Silva LB (2011) Nutritional Bioactive Compounds and Technological Aspects of Minor Fruits Grown in Brazil. Food Res Int 44: 1658–1670. Link: https://bit.ly/3KkGV74
- Aurore G, Parfait B, Fahrasmane L (2009) Bananas, Raw Materials for Making Processed Food Products. Trends Food Sci Technol 20: 78–91. Link: https://bit.ly/33JmtvQ
- Nacitas J (2012) Composition Du Fruit à Pain Récolté Sur Un Territoire Contrasté : Structure, Propriétés et Aptitudes Technologiques de Son Amidon. Link: https://bit.ly/3tBbI9K
- Souci SW, Fachmann W, Kraut H (2000) Food Composition and Nutrition Tables. Food composition and nutrition tables.
- Macías-Ganchozo E, Bello-Moreira I, Trueba-Macías S, Anchundia-Muentes X, Anchundia-Muentes M, et al. (2018) Design, Development and Performance of Solar Dryer for Pineapple (Ananas Comosus (L.) Merr.), Mamey (Mammea Americana L.) and Banana (Musa Paradisiaca L.) Fruit Drying. Acta Agronómica 67. Link: https://bit.ly/3GGCoJQ
- Sun T, Ho CT (2005) Antioxidant Activities of Buckwheat Extracts. Food Chem 90: 743–749. Link: https://bit.ly/3rvHjXE
- Tapiero H, Tew KD, Ba GN, Mathé G (2002) Polyphenols: Do They Play a Role in the Prevention of Human Pathologies? Biomed Pharmacother 56: 200–207. Link: https://bit.ly/3GGvLr8
- Quideau S, Deffieux D, Douat-Casassus C, Pouységu L (2011) Plant Polyphenols: Chemical Properties, Biological Activities, and Synthesis. Angewandte Chemie - International Edition 50: 586–621. Link: https://bit.ly/34UyuPz
- Guyot S, Le Bourvellec C, Marnet N, Drilleau JF (2002) Procyanidins Are the Most Abundant Polyphenols in Dessert Apples at Maturity. LWT - Food Sci Technol 35: 289–291. Link: https://bit.ly/3rM1CAz
- Dragovic-Uzelac V, Levaj B, Mrkic V, Bursac D, Boras M (2007) The Content of Polyphenols and Carotenoids in Three Apricot Cultivars Depending on Stage of Maturity and Geographical Region. Food Chem 102: 966–975. Link: https://bit.ly/33lOObT
- Alonso Borbalán AM, Zorro L, Guillén DA, Barroso CG (2003) Study of the Polyphenol Content of Red and White Grape Varieties by Liquid hromatography-Mass Spectrometry and Its Relationship to Antioxidant Power. J Chromatogr A 1012: 31–38. Link: https://bit.ly/3qM9pz3
- Ferreira Gomes CC, de Siqueira Oliveira L, Rodrigues DC, Ribeiro PRV, Canuto KM, et al. (2021) Evidence for Antioxidant and Anti-Inflammatory Potential of Mango (Mangifera Indica L.) in Naproxen-Induced Gastric Lesions in Rat. J Food Biochem e13880. Link: https://bit.ly/3tRZBFF
- Quintana SE, Salas S, García-Zapateiro LA (2021) Bioactive Compounds of Mango (Mangifera Indica): A Review of Extraction Technologies and Chemical Constituents. J Sci Food Agric 101: 6186–6192. Link: https://bit.ly/33rVSne
- Heim KE, Tagliaferro AR, Bobilya DJ (2002) Flavonoid Antioxidants: Chemistry, Metabolism and Structure-Activity Relationships. J Nutr Biochem 13: 572–584. Link: https://bit.ly/33KWKmy
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
