Global Journal of Rare Diseases
Gottron’s acrogeria and peliosis hepatis
Gloria Cobo Ramírez*, Mónica López Rodríguez, Andrés González García, Marta Rosas Cancio-Suárez, Luis Adrián Viteri and Luis Manzano Espinosa
Cite this as
Ramírez GC, Rodríguez ML, García AG, Cancio-Suárez MR, Viteri LA, et al. (2019) Gottron’s acrogeria and peliosis hepatis. Glob J Rare Dis 4(1): 017-021. DOI: 10.17352/2640-7876.000017We report the case of a 62-year-old woman with Gottron’s acrogeria and peliosis hepatis. Gottron’s acrogeria is a rare congenital disease characterized by atrophic acral skin with mottled hyperpigmentation and an aged appearance. It is mostly caused by a mutation in COL3A1 gene that leads to defective production of type III collagen. These abnormalities in collagen produce fragility in different organs such as blood vessels, causing multiple vascular disorders, which would include peliosis hepatis. Peliosis hepatis is a pathological condition with multiple blood-filled lacunar lesions within the hepatic parenchyma. It is induced by several mechanisms, from chronic debilitating conditions to infections or immunocompromised status, standing out long-term oral contraceptive therapies, which our patient had taken for fifteen years. Peliosis hepatis lesions tend to regress after cessation of estrogen treatment, but our patient developed the vascular disorder eighteen years after ending the therapy, with no evidence of peliosis in previous radiological tests. We suggest that peliosis is related to collagen defects caused by Gottron’s acrogeria. No previously known associations between these entities were found despite exhaustive investigations.
Case Report
We present the case of a 62-year-old woman with Gottron’s acrogeria that associated peliosis hepatis. Gottron’s acrogeria was diagnosed at the age of 28 years. The diagnose was clinical and no genetic tests have been performed. Her mother suffered from the same disease, dying at the age of 56 years due to pericarditis.
Development in early childhood was normal. Her past medical history included multiple episodes of periodontitis that required a complete dental prosthesis at the age of 17 years, associating lower lip and mandibular atrophy that underwent surgery. She had one trouble-free pregnancy and her son did not develop the condition. She started treatment with oral contraceptives (ciproterone/ethinylestradiol) after having her son and took them for 15 years. In her adult life, she was diagnosed with arterial hypertension, iron deficiency anemia, subclinical hypothyroidism, and atrophic gastritis. At the age of 51 years, she presented one episode of pericarditis with severe pericardial effusion, performing at the time a complete body computed tomography (CT) that showed no abnormalities (Figure 1). In the last 15 years, the patient developed multiple cutaneous ulcers in both legs that caused intense pain and episodes of cellulitis, osteomyelitis and septic arthritis, requiring several hospitalizations. Skin graft surgery was finally undergone, with an eventual good control of symptoms. Our patient worked as a cook in schools. She retired at the age of 54 years due to severe functional limitation in lower limbs caused by the absence of subcutaneous tissue.
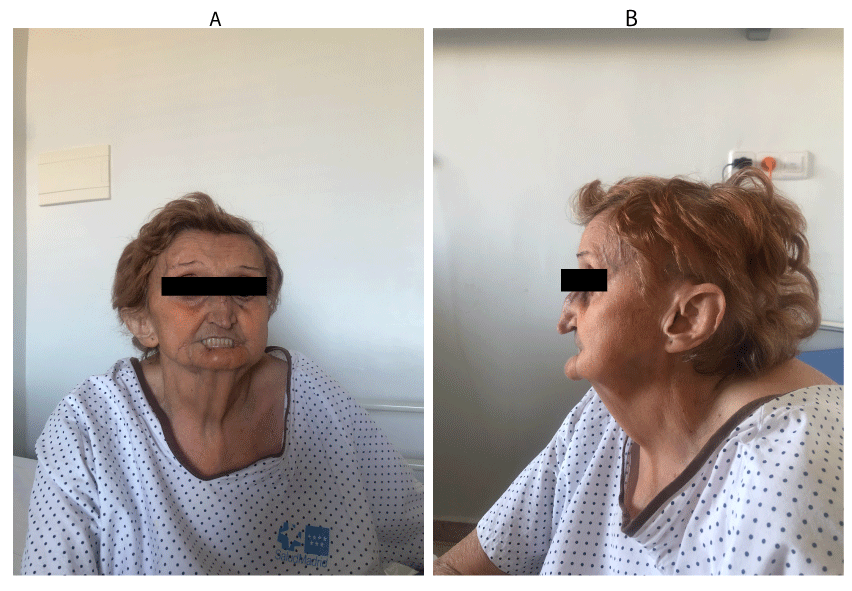
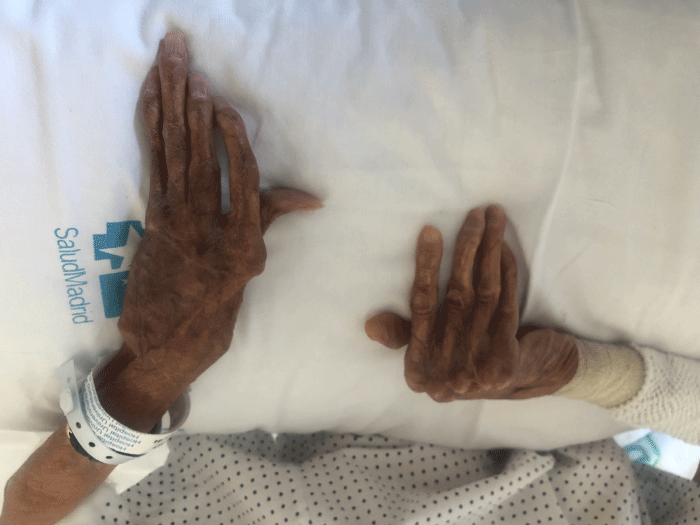
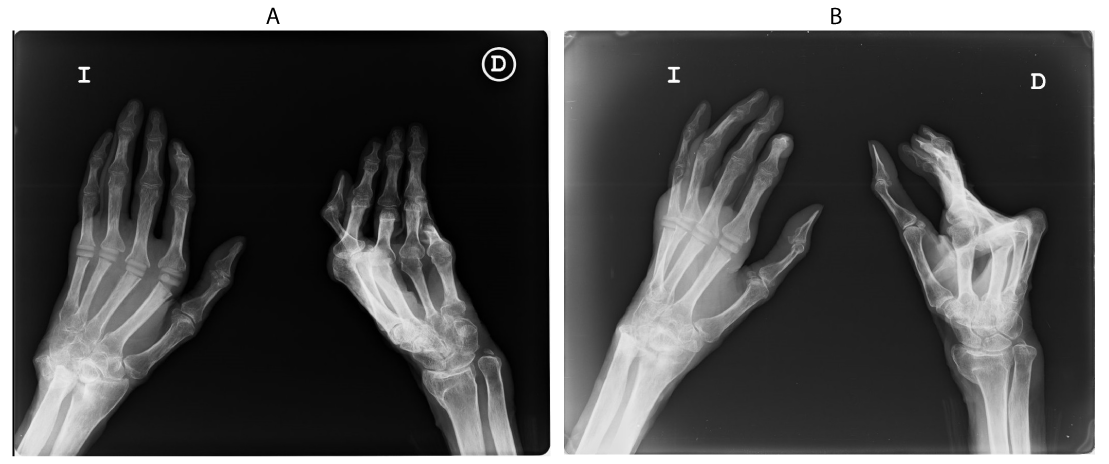
Our patient measured 151 cm and had a body weight of 38.8kg. The physical examination showed atrophic skin changes on her face and extremities. She presented the typical slim and sharp nose and the ears were atrophic with the typical absence of well-formed earlobes. Hypoplasia of the mandible and the maxillae could be seen too, along with complete atrophy of the lower lip (Figure 2). Her hands had wrinkled skin and hyperpigmentation on the dorsal aspects and fingers. The nails were dystrophic. Bone deformation could be seen, especially affecting the distal part of the limbs (Figure 3). There were no sclerotic changes. The abdomen presented some bruises and many scars from skin grafts, with no hepatomegaly. The skin on the legs was thin, associating multiple skin grafts from recent surgery. The underlying structures such as vessels and tendons could be seen due to the lack of subcutaneous tissue. Intense scoliosis was observed too. The radiographs showed osteoporosis and acro-osteolysis on both hands, the left hand showing hypoplastic terminal phalangeal, as well as prosthesis in metacarpophalangeal joints from second to the fifth finger (Figure 4).
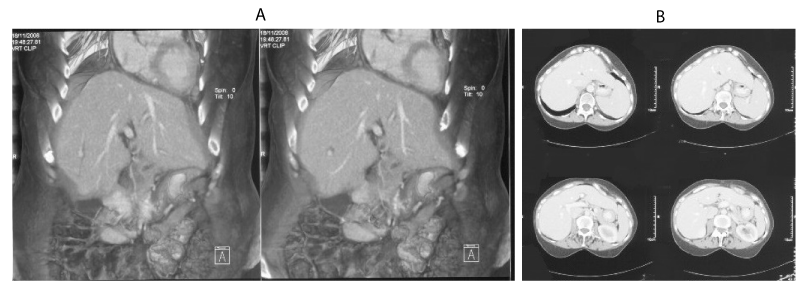
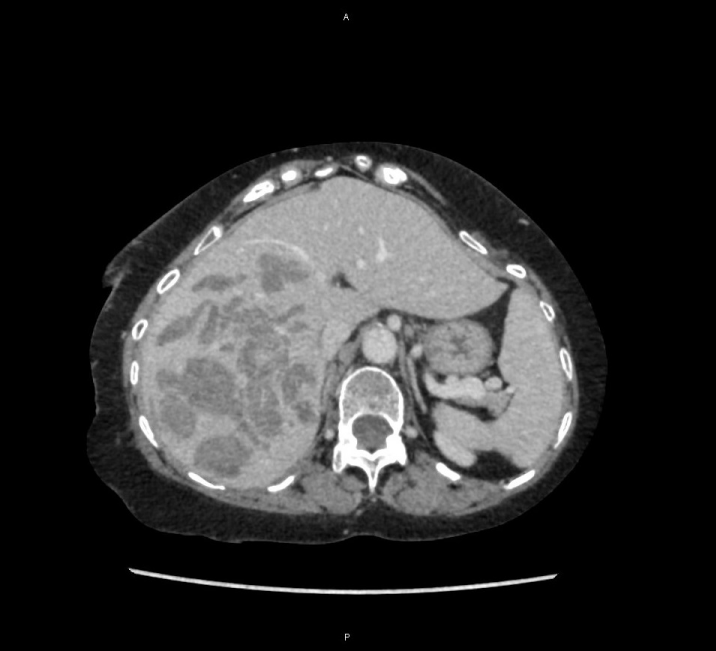
The patient came to our hospital due to low back pain that longed for 2 days. Blood tests evidenced anemia with hemoglobin of 7mg/dl and she was transfused with 2 units of red blood cells. A CT was performed, discovering a hepatic lesion formed by multiple hypodense blood-filled cysts within the right lobule and medial segments of the left lobule, having a mass effect that respected portal vessels. These images were informed as peliosis hepatis versus abscesses (Figure 5). The patient was hospitalized for a week, performing different tests. Her liver function tests were altered, with aspartate transaminase (AST) of 388U/L, alanine aminotransferase (ALT) of 516U/L, gamma-glutamyltransferase (GGT) 166U/L, alkaline phosphatase (ALP) 264U/L and lactate dehydrogenase (LDH) 442U/L, except for total bilirubin (TB) of 0.37mg/dL which was normal. C-reactive protein persisted with high levels during the stay, going from 276mg/L to 106mg/L. Tumor markers such as alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), human chorionic gonadotropin (hCG), CA-125 and CA 19-9 were normal, as well as hepatitis virus antigens, proteinogram and immunoglobulin levels, antinuclear antibodies, extractable nuclear antigens, and complement. An MRI was performed, showing hyperintense lesions in T2-weighted-sequences and T1-weighted-sequences, with no enhancement at the time of contrast medium administration and no signs of over infection. The radiological diagnosis was peliosis hepatis with associated bleeding (Figure 6). We decided not to perform a percutaneous biopsy because of the significant vascularity of the lesion and the consequent high risk of bleeding. The patient was discharged 10 days after admission in good physical condition with normalized liver function, Hb of 9.8mg/dL and no signs of active bleeding.
Methods
We encountered a patient with Gottron’s acrogeria and newly formed hypervascular hepatic masses corresponding to peliosis hepatis and investigated the clinicopathological features underlying this condition.
Informed written consent forms were obtained from the patient.
Discussion
Acrogeria is a term created by Heinrich Gottron in 1941, referring to a rare congenital disease characterized by atrophic acral skin with mottled hyperpigmentation, dystrophic nails, micrognathia and an atrophic tip of the nose, resulting in an aged appearance. Its pathogenesis is very heterogeneous, including several subtypes. Gottron-type acrogeria is mostly caused by abnormal biosynthesis of type III collagen due to mutations on the COL3A1 gene located in the region of chromosome 2q31. These mutations have also been described in type IV Ehlers-Danlos syndrome, also known as vascular type, considering both entities part of the same phenotypic spectrum whose manifestations are determined by the site, position and type of mutation within the COL3A1 gene [1]. The second type of acrogeria also affects the acral skin but keeps a normal production of type III collagen [2]. The third group is overlapped with metageria [3]. Even though acrogeria of the Gottron type is considered a variant of Ehlers-Danlos syndrome type IV, recent studies have shown some clinical and genetic features that overlap with other entities such as Hutchinson-Gilford progeria syndrome, mandibuloacral dysplasia type A or mandibuloacral dysplasia type B [4]. Therefore, Gottron’s acrogeria might be related to various biochemical defects of connective tissue resulting in either exclusively dermatological manifestations or multisystem disorders overlapping with other connective tissue diseases [2]. The mode of inheritance is not fully established since the first reported cases were siblings offspring of non-affected parents but in the last years, several familial cases have been described [5]. However, most reported cases are sporadic [4]. In only a few cases was consanguinity known, not considering this as a risk factor. The genetic options are heterogeneity with dominant or recessive types, dominance with reduced penetrance o dominance with high fetal mortality [6]. Further studies should be undergone.
As said before, Gottron’s acrogeria is a subtype of acrogeria where collagen production is altered. Type III collagen is a structural protein present in multiple organs such as skin, blood vessels, ligaments or organ lining. The protein is coded by COL3A1 gene, which codes a triple helix of glycine and two different amino acids that change [7]. Diseases resulting from COL3A1 mutations are rare but potentially fatal. Since they affect vessels structures, they are characterized by vascular fragility with aneurysms and arteriovenous fistulae that can lead to premature sudden death. Studies undergone with electron microscopy in patients affected by COL3A1 mutations showed enlarged fibrils in the skin and arterial walls compared with normal controls. Due to these defects, the pathologically fragile tissues cannot resist the normal mechanical pressures caused by bloodstream and they burst more easily, leading to bleeding [7]. Some other associations include bone abnormalities and disorders of dermal elastic tissue, as characteristically seen in Ehlers-Danlos syndrome. Histologically, the illness shows replacement of subcutaneous fat by defective connective tissue [6].
Peliosis hepatis (PH) is a very rare disease characterized by multiple, small, blood-filled cysts within the liver [8]. PH was first described in 1861 by E. Wagner; and Shoenlack named it peliosis in 1916 from Greek pelios, meaning purple discolored by extravasated blood [9,10]. PH pathogenesis remains uncertain, but it can be induced by several diseases such as wasting disease, hematologic malignancy, hepatocellular carcinoma, liver and renal transplantation, autoimmune mechanisms, human immunodeficiency virus (HIV) infection, hepatic sarcoidosis and tuberculosis, Bartonella infection and many different drugs [8,11,12]. Among the drugs that have been associated with PH onset, steroids and oral contraceptives are the most reported. Some other biological drugs and chemotherapies such as tamoxifen, methotrexate, thiopurine, azathioprine, and iron chelating agents are also described [8,12-15].
The liver is the most frequently affected organ, but the same process may occur in the bone marrow, lymph nodes or the spleen, where peliosis related to increased VEGF has been reported [16,17]. In hepatic localizations, the differential diagnoses of PH include adenomas, hemangioma, focal nodular hyperplasia, Caroli’s disease, multiple abscesses and metastatic adenocarcinomas [8].
CT images of peliosis may show small lesions that go from a few millimeters up to various centimeters in diameter. The lesions generally appear as multiple hypodense areas at unenhanced CT, although their aspect depends on the degree of thrombosis or bleeding inside the cysts and the presence of concomitant liver conditions such as steatosis, fibrosis or cirrhosis [8,10,18]. At medium-enhanced mode, in the arterial phase the lesions appear initially hypodense, progressively becoming hyperdense. In the portal venous phase, the enhancement may be either complete or early globular vessel-like enhancement, progressing centripetally o centrifugally. In the delayed phase, diffuse increased attenuation could be seen [19,20]. However, MRI examination represents the gold standard in the radiologic diagnosis of this disease [21]. The imaging varies depending on the blood component of the lesion. In T2-weighted-sequences, the most common images are hyperintense lesions compared to the liver parenchyma with high-signal multiple spots that represent hemorrhagic necrosis. In T1-weighted-sequences, hypo, iso or hyperintense lesions can be detected, and they generally show centrifugal enhancement at the time of contrast medium administration. The acquisition of the contrast medium during the late phase suggest a hemangioma [8]. Therefore, the differential diagnosis may include hemangiomas, but also different entities as hypervascular metastases or abscesses. Hypervascular metastases are typically hypo or isodense in the portal venous phase, while hemangiomas show discontinuous globular enhancement in the arterial phase. Abscesses generally present cluster-of-grapes appearance with septs and absence of enhancement of the collection [20]. As the differential diagnosis of liver masses is complex, and benign and malignant lesions share overlapping imaging characteristics, peliosis hepatis is very difficult to diagnose, so a careful history-taking is necessary [11,20].
PH does not show a well-defined evolution. It can remain asymptomatic and present as an accidental finding. In other cases, the disease shows important effects of compression with stenosis of sinusoids or the vena cava. The possibility of hepatic rupture varies in percentage considering the different locations of the disease: on the right liver, it bursts in 75% of the cases, on the left liver in 11% of the cases and when it affects both lobules in 14% of the cases [22]. This risk is the reason why some studies have described that the incidental finding of a lesion, potentially compatible with PH, requires elective surgery with diagnostic and therapeutic intents [8]. However, other studies report that surgery should be reserved for cases where hemorrhage is life-threatening [12]. In cases caused by some specific drug intake, a total regression was noticed after the interruption of the treatment, which reinforces the indication of conservative management [12-15,23].
Since the seventies, most studies have shown an association between the use of oral contraceptive pills (OCPs) and a variety of liver tumors. The evidence includes a large number of case reports of tumors in women taking OCPs, reports of regression after cessation of OCPs and recurrence of tumors after resection in patients that persisted with hormonal therapy [24]. The most common entities are benign hepatic neoplasms such as adenomas, hemangiomas or peliosis [14,24]. This is likely caused by the presence of estrogen receptors in liver cytosol and nuclei that stimulate hepatic activity and facilitate the development of defective tissue [24]. The first report of generalized peliosis hepatis as a complication of long-term use of oral contraceptives was made by Van Erpecum et al. in 1988 [25]. Chronic use of OCP has been associated with sinusoidal dilatation, although they do not cause sinusoidal endothelial cell injury as it happens with other agents. In most cases, the lesions show regression of the severity of peliosis after interruption of usage [9,21,25]. Generalized PH leading to cirrhosis has also been reported after long-term use of contraceptives [25].
According to this, we have a patient with peliosis hepatis and two conditions that favor its development: a defect on collagen production due to Gottron’s acrogeria and long-term use of oral contraception. Most studies show a relationship between the use of contraception and hepatic vascular disorders such as peliosis, while there is no description in reviewed literature of association between peliosis and acrogeria. However, relation between splenic peliosis and Ehlers-Danlos syndrome, which is part of the collagen alteration spectrum, has been reported [16]. Hence, the main hypothesis would be that our patient’s peliosis is caused by long-term estrogen exposure. However, those studies also state that peliosis tends to regress a few months after the withdrawal of contraception. Our patient ended her contraception treatment at the age of 46 years and a complete body CT was performed at the age of 51 with no references to blood-cysts in the liver. Therefore, the lesions observed in the current hospitalization must have developed later.
In conclusion, we describe a patient with a history of Gottron’s acrogeria and long-term use of contraceptives that develops peliosis hepatis. However, no liver masses had been described in studies performed before this episode and years after cessation of estrogen therapy. We suggest that Gottron’s acrogeria (that associates defects on type III collagen of blood vessels) could play a facilitating role to develop peliosis hepatis, with a significant weakness in hepatic tissue.
- Jansen T, de Paepe A, Luytinck N, Plewig G (2000) COL3A1 mutation leading to acrogeria (Gottron Type). Br J Dermatol 142: 178-180. Link: http://bit.ly/2rxizmJ
- Bruckner-Tuderman L, Vogel A, Schnyder UW (1987) Fibroblasts of an acrogeria patient produce normal amounts of type I and III collagen. Dermatologica 174: 157-165. Link: http://bit.ly/36OqJHn
- Gilkes JJ, Sharvill DE, Wells RS (1974) The premature ageing syndromes. Report of eight cases and description of a new entity named metageria. Br J Dermatol 91: 243-262. Link: http://bit.ly/33CKu2y
- Maroofian R, Murdocca M, Rezaei-Delui H, Nekooei A, Mojarad M, et al. (2018) A novel in-frame deletion in ZMPSTE24 is associated with autosomal recessive acrogeria (Gottron type) in an extended consanguineous family. Clin Dysmorphol 27: 88-90. Link: http://bit.ly/2Q5CBir
- Blaszczyk M, Depaepe A, Nuytinck L, Glinska-Ferenz M, Jablonska S (2000) Acrogeria of the Gottron type in a mother and son. Eur J Dermatol 10: 36-40. Link: http://bit.ly/2pQL0f1
- de Groot WP, Tafelkruyer J, Woerdeman MJ (1980) Familial acrogeria (Gottron). Br J Dermatol 103: 213-223. Link: http://bit.ly/3722sOm
- Pope FM, Narcisi P, Nicholls AC, Germaine D, Pals G, et al. (1996) COL3A1 mutations cause variable clinical phenotypes including acrogeria and vascular rupture. Br J Dermatol 135: 163-181. Link: http://bit.ly/2O2A3ib
- Crocetti D, Palmieri A, Pedulla G, Pasta V, D'Orazi V, et al. (2015) Peliosis hepatis: Personal experience and literature review. World J Gastroenterol 21: 13188-13194. Link: http://bit.ly/32Cq7kK
- Samyn M, Hadzic N, Davenport M, Verma A, Karani J, et al. (2004) Peliosis hepatis in childhood: case report and review of the literature. J Pediatr Gastroenterol Nutr 39: 431-434. Link: http://bit.ly/2O4ncfs
- Iwata T, Adachi K, Takahashi M (2017) Peliosis Hepatis Mimicking Malignant Hypervascular Tumors. J Gastrointest Surg 21: 1095-1098. Link: http://bit.ly/32BJDxX
- Kim SB, Kim DK, Byun SJ, Park JH, Choi JY, et al. (2015) Peliosis hepatis presenting with massive hepatomegaly in a patient with idiopathic thrombocytopenic purpura. Clin Mol Hepatol 21: 387-392. Link: http://bit.ly/36Mt4ml
- Cimbanassi S, Aseni P, Mariani A, Sammartano F, Bonacina E, et al. (2015) Spontaneous hepatic rupture during pregnancy in a patient with peliosis hepatis. Ann Hepatol 14: 553-558. Link: http://bit.ly/2CxjsOw
- Hui CL, Lee ZJ (2019) Hepatic disorders associated with exogenous sex steroids: MR imaging findings. Abdom Radiol (NY) 44: 2436-2447. Link: http://bit.ly/34PD504
- Ponnatapura J, Kielar A, Burke LMB, Lockhart ME, Abualruz AR, et al. (2019) Hepatic complications of oral contraceptive pills and estrogen on MRI: Controversies and update - Adenoma and beyond. Magn Reson Imaging 60: 110-121. Link: http://bit.ly/2X6w1tA
- Staub PG, Leibowitz CB (1996) Peliosis hepatis associated with oral contraceptive use. Australas Radiol 40: 172-174. Link: http://bit.ly/2NDO2MN
- van Bon AC, Kristinsson JO, van Krieken JH, Wanten GJ (2009) Concurrent splenic peliosis and vascular Ehlers-Danlos syndrome. Ann Vasc Surg 23: 256. e1-4. Link: http://bit.ly/2CtiTVQ
- Joseph F, Younis N, Haydon G, Adams DH, Wynne S, et al. (2004) Peliosis of the spleen with massive recurrent haemorrhagic ascites, despite splenectomy, and associated with elevated levels of vascular endothelial growth factor. Eur J Gastroenterol Hepatol 16: 1401-1406. Link: http://bit.ly/2NCBe92
- Iannaccone R, Federle MP, Brancatelli G, Matsui O, Fishman EK, et al. (2006) Peliosis hepatis: spectrum of imaging findings. AJR Am J Roentgenol 187: W43-W52. Link: http://bit.ly/2q2nXO9
- Gouya H, Vignaux O, Legmann P, de Pigneux G, Bonnin A (2001) Peliosis hepatis: Triphasic helical CT and dynamic MRI findings. Abdom Imaging 26: 507-509. Link: http://bit.ly/32A5Tbp
- Torabi M, Hosseinzadeh K, Federle MP (2008) CT of nonneoplastic hepatic vascular and perfusion disorders. Radiographics 28: 1967-1982. Link: http://bit.ly/36UV6Mf
- Ferrozzi F, Tognini G, Zuccoli G, Cademartiri F, Pavone P (2001) Peliosis hepatis with pseudotumoral and hemorrhagic evolution: CT and MR findings. Abdom Imaging 26: 197-199. Link: http://bit.ly/33EBkTe
- Sommacale D, Palladino E, Tamby EL, Diebold MD, Kianmanesh AR (2013) Spontaneous hepatic rupture in a patient with peliosis hepatis: A report of one case. Int J Surg Case Rep 4: 508-510. Link: http://bit.ly/2Q34sQh
- Ikeda N, Oka K, Yonekawa N, Takaku H, Suzuki T, et al. (2011) Pelioid-type well-differentiated hepatocellular carcinoma in a patient with a history of taking oral contraceptives: report of a case. Surg Today 41: 1270-1274. Link: http://bit.ly/2O3aKNj
- Kern F, ErflingW, Simon FR, Dahl R, Mallory A, et al. (1978) Effect of estrogens on the liver. Gastroenterology 75: 512-522. Link: http://bit.ly/2WZ04mS
- van Erpecum KJ, Janssens AR, Kreuning J, Ruiter DJ, Kroon HM, et al. (1988) Generalized peliosis hepatis and cirrhosis after long-term use of oral contraceptives. Am J Gastroenterol 83: 572-575. Link: http://bit.ly/36SY1Fq

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Save to Mendeley
Save to Mendeley
