Global Journal of Medical and Clinical Case Reports
A case with rare manifestation of Cronkhite-Canada Syndrome with remission after treatment and literature review
Patel Dharmik and Tripathi Kaushalendra Mani*
Cite this as
Dharmik P, Mani TM (2018) A case with rare manifestation of Cronkhite-Canada Syndrome with remission after treatment and literature review. Glob J Medical Clin Case Rep 5(4): 025-029. DOI: 10.17352/2455-5282.000065Cronkhite-Canada syndrome is a rare disorder which is sporadic in nature, its pathology and cause is not fully understood, usually characterized by the presence of polyps in the stomach and large intestine and less frequently in the oesophagus with associated, the aim of this report is to discuss the pathogenesis of this syndrome and possible treatment options, in this case there was a non-conventional approach applied to this with a good prognosis, the disease itself is usually present among the Japanese population with a male to female ratio 2:1 but was of irrelevance as our patient was Indian, this report will also shed some light on the use of herbal medicine and its application in the treatment of this disease along with conventional medicine.
Introduction
Cronkhite-Canada Syndrome is a sporadic condition which involves tissues arising from ectodermal germ cell layer [1]. It is characterized by gastrointestinal hamartomatous polyposis, diarrhoea, hypoproteinaemia and cutaneous manifestations including alopecia, onychodystrophy, hyperpigmentation with other symptoms such as dysguisea, hypoguisea and xerostomia due to zinc and cobalt deficiency have been reported in some patients. [2,3]. Its Incidence is low, but mortality is high. Only 500 cases of CCS have been reported around the world with an approximate 60% overall mortality rate [4,5], with a mean age of presentation being 59 with a male predominance in the ratio of 3:2 [6], with a majority of cases happening in ages older than 50 .Although the first case of Cronkhite-Canada syndrome was reported in 1955, the pathogenesis of this disease has never been fully understood [7], although two theories support an immunological and an infectious basis to the pathogenesis of the disease with the latter being considered because of the histopathological findings from gastrointestinal polyps showing inflammatory cells infiltration with mononuclear cells and eosinophils [8]. The CCS polyps are infiltrated with IgG4 plasma cells; elevated antinuclear antibody and a decreased manifestation of disease occurs when treated with corticosteroids, and association with other autoimmune disease such hypothyroidism, systemic lupus erythematosus, rheumatoid arthritis and scleroderma strongly supports autoimmune pathophysiology of this disease [9]. The only postulated risk factors for CCS includes mental and physical stress [10], we are going to discuss a case report of a 44-year-old male patient with a systemic manifestation of the disease and its remission to further understand the disease and possible treatment options for this syndrome.
Case Report
Presenting history
A 44-year-old Indian male residing in Canada for the past 16 years with a history of gout in the last 7 years presented with complaints of chronic diarrhoea, intermittent bloody stools, nausea, abdominal pain, weight loss for 2 months, with occasional alcohol intake for the past 10 years. He was on proton pump inhibitors for chronic gastritis, Allopurinol for gout, ibuprofen for symptomatic relief of gouty pain and after a week of Ibuprofen he spontaneously developed diarrhoea which did not resolve with medication with a frequency of up to 10 times a day, with his hair greying initially from his pubis advancing to his axillary hair, to his moustache and beard with the scalp being the last affected site. He initially had intermittent abdominal cramps which turned into several bouts of abdominal pain. He began to shed all his hairs including nasal hairs causing continuous nasal discharge and dried nasal mucosa. And developed brittle nails which also started to fall off. During this period, he also suffered from severe malnutrition and lost almost 4 kg weight in 3 weeks. On his first hospital visit, a git pathology was assumed, pantoprazole with antibiotics were prescribed after X-ray and blood works. On further investigation, he underwent an Esophagogastroduodenoscopy with biopsy which revealed small sessile polyps in the entire digestive tract except in the oesophagus, he was soon started on corticosteroids, with a possible immune response leading to the formation of polyps, kept in mind. In the coming 5 weeks, he further lost 6 kg of weight with a loss of taste sensation. He then underwent video colonoscopy during which biopsy from colonic and gastric polyps was taken for immuno-histological findings.
Physical exam
On physical examination, he was fairly built, poorly nourished, weighing 68 kg, presenting without eyebrows, baldness. onychodystrophy with loss of finger and toenail. The gouty swelling was present on the right toe. There were hyperpigmented oval spots measuring 5mm to 15mm in diameter all over his palms and sole. His palms were sweaty with some scattered hyperpigmentation noted on different parts of the body. Physical exam also showed bilateral symmetric oedema in lower extremities. No other significant findings were present.
Laboratory
His eosinophil count was 7.4% (normal range: 0.0 – 6) with an absolute count of 0.58 x 10^3 / μl (normal range: 0-0.5), his haemoglobin was 12.5 g/dl (normal range 13 – 17), he had a Mean Corpuscular Haemoglobin of 26 pq (normal range 27-32), Mean Corpuscular Haemoconcentration of 26.5 g/dl (normal range 31.5 – 34.5). Red Cell distribution Width – Sd of 53.6 fL (normal range 39-46). Red Cell distribution Width – CV of 14.6 % (normal range of 11.6-14), total 25-OH vitamin D of 6.5 ng/ml, Calcium levels of 8.09 mg/dl (normal 8.8 – 10.6), uric acids levels of 9.4 (normal range 3.7 – 9.2), Estimate Glomerular filtration rate of 81 (between 60 – 89 indicates a mild decrease).
Endoscopy with biopsy
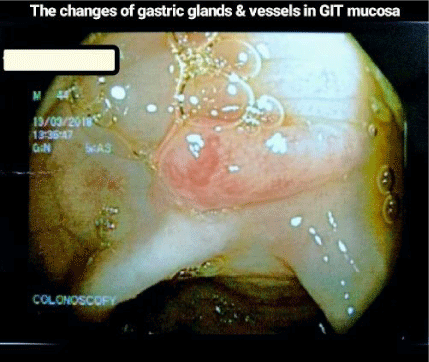
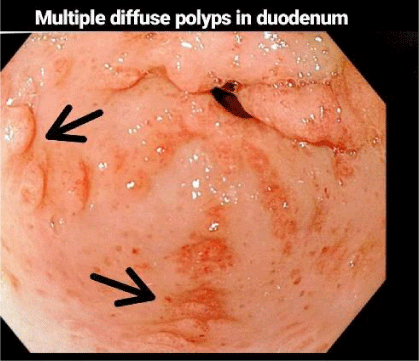
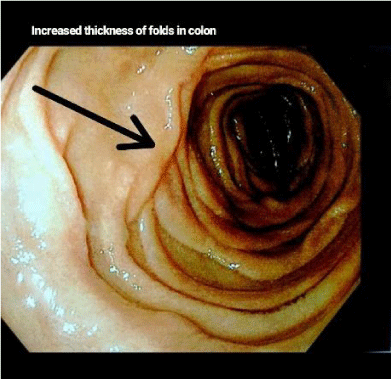
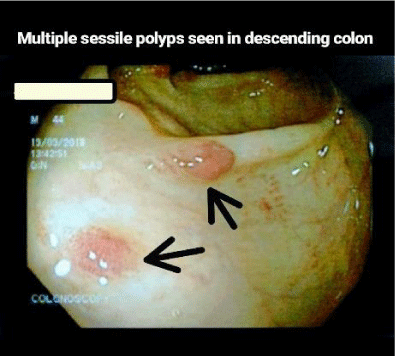
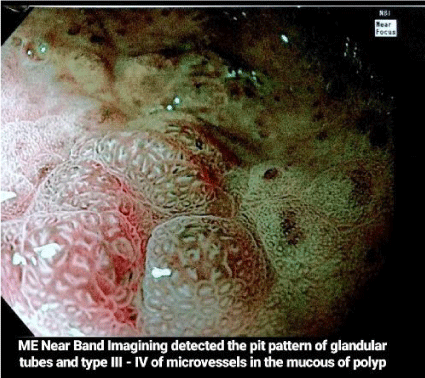
Esophagogastroduodenoscopy revealed polyps with stomach villous atrophy and a normal oesophagus with changes seen in gastric glands and mucosa (Figure 1). A few haemorrhagic foci were present on in the inner surface of the stomach. In the duodenum, rugal folds were thickened and had brownish discoloration. Multiple polyps were seen in the body and antrum of the stomach; the first, second and third parts of the duodenum (Figure 2). Colonoscopy revealed multiple polyps and inflammation throughout the colon with increased thickened folds (Figure 3). The intensity of inflammation and density of polyps were more in the ascending colon, transverse colon (Figure 4) and descending colon (Figure 5) with a reduced number seen in sigmoid colon and rectum while terminal ileum noted normal. Piles were also present. Magnification Endoscopy-Narrow Band imaging detected the pit pattern of glandular tubes Type 3-4 of micro vessels in the mucous of the polyps (Figure 6). On the basis of these abnormal features, the duodenum, entire stomach, and polyps were extensively biopsied for histopathological imaging.
Histopathology
Duodenal biopsy revealed subtotal to total villous atrophy, moderately expanded oedematous lamina propria, mononuclear inflammatory infiltrate with prominent eosinophils, scattered foci of crypt and glandular dilation, mildly increased intraepithelial lymphocytes not reaching diagnostic threshold for marsh lesion without any dysplasia or malignancy while Antral and Oxyntic gastric mucosa exhibited prominent expansion of lamina propria by oedema, mononuclear inflammatory infiltrate with prominent eosinophils, architecture abnormalities including foveolar hyperplasia and dilatation with focal intestinal metaplasia. Foci of epithelial attenuation, denuded surface gastric epithelium without surface erosion were present. Scattered hemosiderin-laden macrophages indicated prior haemorrhage. The biopsy of polyps showed benign polypoid colonic tissue with a few dilated, abscessed crypts and benign polypoid gastric mucosa having chronic inflammation with intestinal metaplasia without dysplasia.
Immunostaining
CD34, CD117 and CD68, highlighted prominent vascularity especially on the surfaces, patchy prominent number of mast cells, and large number of macrophages, respectively. Actin and Desmin immunostains demonstrate splaying of smooth muscle fibres in between glands and foveolar pits. Immunostaining with CAM5.2, CK AE1/AE3, and EMA gained negative result for any infiltrating malignant epithelial cell. Also, screening with IgG4 yielded negative result.
Computed tomography
10 weeks post treatment CT showed mild bilateral pelviectasis indicating a distended urinary bladder otherwise showed no other abnormalities.
Treatment
Symptomatic treatment initially included corticosteroids, analgesics, broad-spectrum antibiotics. Nutritional support was provided with multivitamin tablets, calcium and iron. PPIs were stopped. Protein rich diet was recommended. It reduced symptoms but not effectively. Then in India, he stopped all medications and started Indian herbal medicine with a diet-based treatment. Herbal powder was mixture of dried fruit of Aegle marmelos (Indian bael), Zingiber officinale (Dried ginger), Gymnema sylvestre leaf extract in ratio of 1:1:2 bowls respectively. Daily Diet consisted of turmeric milk, cracked wheat and puffed rice only. Calcium and Iron folic acid tablets were supplemented. This diet was followed for six weeks. By the end of sixth week there was only 2 episodes of bloody diarrhea, stool frequency decreased to 5-6 times from 10 times per day. After six-week, khichri, an Indian dish made from cracked wheat and lentils was added into his diet. Herbal powder was replaced by herbal tablets consisting of bark extracts from Holarrhena antidysenterica, root extracts from Plumbago zeylanica, root extracts of Piper longum, leaf extracts of Bryophyllum pinnatumand bark extracts of Cinnamomum camphora. Six weeks later he started growing white hair on his eyebrow, moustache and pubic area. Frequency of stool decreased to 3-4 times a day. Hair started blackening, weight increased by 2 kg and lastly new nails started growing, at the end of the 12 weeks from starting diet-based treatment, an evaluatory CT Scan supported the improvement in pathology. Currently he is off all herbal medication and takes Mesalazine 1200 mg, four times a day.
Case Discussion
CCS is typically diagnosed with a multidisciplinary approach with characteristic clinical, endoscopic, radiologic, and histologic findings, supporting the resultant malabsorption features as well [11]. Most reported cases have a mean age of 59 years. But the age of our patient was 45 years which is relatively infrequent in literature. Clinical features such as weight loss, diarrhoea and triad of ectodermal changes [12] are suggestive of CCS. The most common initial endoscopic findings are gastric infection, antral malignancy [13] and polyps which are found in the entire alimentary tract, except the notable exception of the oesophagus [14]. According to analysis of Watanabe et al. [15], CCS polyps have a typical histopathology associated with focal dilated cystic glands, some filled with proteinaceous fluid or inspissated mucus, the polyp and interpolyp area are oedematous, with congestion and chronic inflammation of the lamina propria and submucosa. These findings are consistent with our case.
Differential diagnosis include rare hamartomatous GI polyposis (juvenile polyposis, peutz- jeghers syndrome, cowden syndrome), Menetrier disease, multiple hyperplastic polyposis and eosinophilic gastroduodenitis. The hemartamous GI polyposis are considered unlikely in view of the inflamed stomach on endoscopy, the diffuse nature of the disease, and the presence of marked villous atrophy in the duodenum. Menetrier disease is also unlikely as it typically spares the antrum and does not involve the small bowel. Hyperplastic polyposis is frequently multiple and most often involve the antrum, but they are seldom so numerous as to diffusely involve the entire stomach. Eosinophilic gastroduodenitis is considered due to the prominent number of eosinophils; however, it does not usually exhibit this degree of lamina propria oedema as well as the described architectural abnormalities. While diffuse polyposis in the upper gastrointestinal tract, mostly sessile, a second less common scenario is diffusely thickened or atrophic rather than polypoid mucosa, often raising the differential diagnosis an infiltrative neoplastic process (especially lymphoma or linitis plastica with carcinomatosis), which are ruled out by immuno-stains such as CAM5.2, CK AE1/AE3, and EMA.
There is association between autoimmune disease and CCS, but in this patient, it is associated with gout, an inflammatory disease. Sweetser et al. found that 52% of CCS polyps were positive (>5 cells/HPF) for IgG4 immunostaining, which provided support for autoimmune aetiology of the disease. But, in our patient IgG4 and ANA antibodies both yielded negative results, rejecting the autoimmune hypothesis. While other inflammatory markers were positive in this patient’s polyp further providing support for inflammatory hypothesis.
The optimal therapy for CCS is not known but several treatment options have been described with various approaches having different degrees of success [16].
Aegle marmelos (Indian bael) fruit constitutes phenolics, flavonoids, carotenoids and vitamin C [17]. Phytochemicals isolated from fruits are marmelosin, luvangetin, aurapten, psoralen, marmelide, and tannin [18], which provides antidysenteric, antiulcer, antipyretic, chemopreventive, antineoplastic, antigenotoxic and anti-inflammatory properties. Antidysenteric property is due to marmelosin which act as antibacterial and anthelmintic [19] even Mazumdar et al., found that the extract was comparable to that of ciprofloxacin [20]. Luvangetin (pyranocumarin) and quercetin (flavonoid) protects against ulceration. Polyphenolic constituents possess chemopreventive effects via protecting DNA from reactive oxygen species and S9 dependent mutagens [21]. Lupeol, another compound present in A. marmelos, possesses antineoplastic effects on various human neoplastic cell lines [22], preventing risk of malignancy associated with CCS [23]. The rhizomes of Zingiber officinale, commonly known as ginger contains pungent vanillyl ketones, including gingerol, shogaol and poradol, provides anti-inflammatory property via anti-lipoxygenase activity which is more significant than that of diclofenac [24]. It is of immense value in treating various gastric ailments like dyspepsia, belching, bloating, gastritis, epigastric discomfort, gastric ulcerations, indigestion, nausea and vomiting and scientific studies have validated the ethnomedicinal uses. It is also effective in preventing gastric ulcers induced by nonsteroidal anti-inflammatory drugs, reserpine, ethanol, stress [25]. It is important to note that patient was on NSAIDs. Also, Calva et al., postuated stress as important risk factor for CCS. Gymnema sylvestre, known for sweet suppression activity is due to the phytoconstituents triterpene saponins including gymnemic acids, gymnemasaponins, and a polypeptide, gurmarin. The herb exhibits a broad range of therapeutic effects as an effective natural remedy for diabetes, besides being used for arthritis, anemia, osteoporosis, indigestion, and anti-inflammatory [26]. Gymnemagenol, another isolated chemical exhibited anticancer potential on HeLa cancer cell lines [27]. Thus, initial aggressive treatment was targeted towards inflammation control, mutation correction, symptomatic relief and improving digestion, hence absorption and nourishment.
The aim of next regimen was to maintain remission, prevent relapse, eliminate probability of malignancy via antioxidant and antineoplastic remedies. Conessine, an alkaloid extracted from bark of Holarrhena antidysenterica gives protection against multiple stages of diarrhea [28]. Pal et al. observed that the bark powder administered to patients with bleeding piles at a dose of 4 g twice a day for 2 weeks each showed significant efficacy [29], Plumbago zeylanica possess bactericidal activity [30] and also helps in normalizing gut flora [31]. Piperlongumine and piperine, amide-alkaloids derived from Piper longum, have longer lasting preventive potentials against chronic mild stress triggered gastric ulcers [32]. Bryophyllum pinnatumleaf extracts exhibits in vivo gastroprotective activity by markedly decreasing the incidence of ulcers in ethanol induced ulcer rats [33]. The beneficial health effects of bark extracts from Cinnamomum zeylanicum includes antioxidant and free-radical scavenging properties; anti-secretagogue, hepatoprotective and anti-gastric ulcer effects; anti-microbial, anti-parasitic, anti-nociceptive and anti-inflammatory activities [34].
Conclusion
In summary, there is still much to be understood about this syndrome. Although this is a nonfamilial disorder of unknown aetiology, some studies suggest an immune-mediated disorder based on IgG4 positive staining. Contrary to this, our patient had a negative IgG4 result, resistant to steroids; supporting inflammatory mechanism which is a rare report. An unusual feature of our case is its association with an inflammatory condition: gout, and NSAIDs. This report may help further understand the disease and develop new drugs from the isolation of herbal extracts along with conventional medicine as its treatment is based mostly on trials . We recommend post treatment monitoring and appropriate measure to prevent relapse.
- Bandyopadhyay D, Hajra A, Ganesan V, Kar SS, Bhar D, Layek M, et al. (2016) Cronkhite- Canada Syndrome; a Case Report and Review of the Literature. Case Rep Med 4210397. Link: https://goo.gl/vk3fqr
- Seth S, Boardman LA. Cronkhite-Canada Syndrome an Acquired Condition of Gastrointestinal Polyposis and Dermatologic Abnormalities. Gastroenterol Hepatol (N Y) 8: 201–203. Link: https://goo.gl/pNnUa6
- Berzin TM, Greenberger NJ, Levy BD, Loscalzo J (2012) Worth a second look. New Eng J Med 366: 463–468. Link: https://goo.gl/jvM19v
- Rubio CA, Bjork J (2016) Cronkhite-Canada syndrome - A Case report. Anticancer Res 36: 4215–4217. Link: https://goo.gl/HDKQDg
- Daniel ES, Ludwig SL, Lewin KJ, Ruprecht RM, Rajacich GM, Schwabe AD (1992) The Cronkhite-Canada Syndrome. An analysis of clinical and pathologic features and therapy in 55 patients. Medicine (Baltimore) 61: 293-309. Link: https://goo.gl/YhC56E
- Kao KT, Patel JK, Pampati V (2009) Cronkhite-Canada syndrome: a case report and review of literature. Gastroenterology Research and Practice 2009: 4. Link: https://goo.gl/FRmmPz
- Cronkhite LW, Canada WJ (1955) Generalised gastrointestinal polyposis—an unusual syndrome of polyposis, pigmentation, alopecia and onychotrophia. The New England Journal of Medicine 252: 1011–1015. Link: https://goo.gl/WMiT8C
- Ward EM, Wolfsen HC (2002) Review article: the non-inherited gastrointestinal polyposis syndromes. Alimentary Pharmacology and Therapeutics 16: 333–342. Link: https://goo.gl/k92D2o
- Riegert-Johnson DL, Osborn N, Smyrk T, Boardman LA (2007) Cronkhite-Canada syndrome hamartomatous polyps are infiltrated with IgG4 plasma cells. Digestion 75: 96–97. Link: https://goo.gl/k43GCm
- Calva D, Howe JR (2008) Hamartomatous polyposis syndromes. Surgical Clinics of North America 88: 779–817. Link: https://goo.gl/3knvp8
- Chakrabarti S (2015) Cronkhite-Canada Syndrome (CCS)—A Rare Case Report. J Clin Diagn Res 9: OD08–OD09. Link: https://goo.gl/zeh7fr
- Seshadri D, Karagiorgos N, Hyser MJ (2012) A case of cronkhite-Canada syndrome and a review of gastrointestinal polyposis syndromes. Gastroenterol Hepatol (N Y) 8: 197-201. Link: https://goo.gl/w29efS
- Bettington M, Brown IS, Kumarasinghe MP, de Boer B, Bettington A, et al. (2014) The Challenging Diagnosis of Cronkhite-Canada Syndrome in the Upper Gastrointestinal Tract. Am J Surg Pathol 38: 215–223. Link: https://goo.gl/sCc4Z1
- She Q, Jiang JX, Si XM, Tian XY, Shi RH, et al. (2013) A Severe Course of Cronkhite-Canada Syndrome and the Review of Clinical Features and Therapy in 49 Chinese Patients. Turk J Gastroenterol 24: 277–285. Link: https://goo.gl/xw5i7T
- Watanabe C, Komoto S, Tomita K, Hokari R, Tanaka M, et al. (2016) Endoscopic and Clinical Evaluation of Treatment and Prognosis of Cronkhite–Canada Syndrome: a Japanese Nationwide Survey. J Gastroenterol 51: 327–336. Link: https://goo.gl/BES3sn
- Ward EM, Wolfsen HC (2003) Pharmacological Management of Cronkhite–Canada Syndrome. Expert Opin Pharmacother 4: 385–389. Link: https://goo.gl/sPumRV
- Charoensiddhi S; Anprung P (2010) Characterization Of Bael Fruit (Aegle Marmelos [L.] Correa) Hydrolysate As Affected By Enzyme Treatment. Journal of Food Biochemistry 34: 1249–1267. Link: https://goo.gl/qZKYyf
- Maity P1, Hansda D, Bandyopadhyay U, Mishra DK (2009) Biological Activities of Crude Extracts and Chemical Constituents of Bael, Aegle Marmelos (L.) Corr. Indian J Exp Biol 47: 849–861. Link: https://goo.gl/qK5yDL
- Ghosh S, Playford RJ (2003) Bioactive Natural Compounds for the Treatment of Gastrointestinal Disorders. Clin Sci (Lond) 104: 547–556. Link: https://goo.gl/bLXjnL
- Mazumder R, Bhattacharya S, Mazumder A, Pattnaik AK, Tiwary PM, et al. (2006) Antidiarrhoeal Evaluation Of Aegle Marmelos (Correa) Linn. Root Extract. Phytother Res 20: 82–84. Link: https://goo.gl/uz4Ucw
- Kaur P, Walia A, Kumar S, Kaur S (2009) Antigenotoxic activity of polyphenolic rich extracts from Aegle marmelos (L.) Correa in human blood lymphocytes and E. coli. Nat Prod Rec 3: 68–75. Link: https://goo.gl/vUXUfp
- Baliga MS, Thilakchand KR, Rai MP, Rao S, Venkatesh P (2013) Aegle Marmelos (L.) Correa (Bael) and Its Phytochemicals in the Treatment and Prevention of Cancer. Integr Cancer Ther 12: 187–196. Link: https://goo.gl/nqDCLR
- Zong Y, Zhao H, Yu L, Ji M, Wu Y, et al. (2017) Case Report—Malignant Transformation in Cronkhite–Canada Syndrome Polyp. Medicine (Baltimore) 96: e6051. Link: https://goo.gl/JScGK4
- Ezzat SM, Ezzat MI, Okba MM, Menze ET, Abdel-Naim AB (2018) The Hidden Mechanism beyond Ginger ( Zingiber Officinale Rosc.) Potent in Vivo and in Vitro Anti-Inflammatory Activity. J Ethnopharmacol 214: 113–123. Link: https://goo.gl/u4nt1R
- Haniadka R, Saldanha E, Sunita V, Palatty PL, Fayad R, et al. (2013) A Review of the Gastroprotective Effects of Ginger (Zingiber Officinale Roscoe). Food Funct 6: 845–855. Link: https://goo.gl/LKze6s
- Tiwari P, Mishra BN, Sangwan NS (2014) Phytochemical and Pharmacological Properties Of Gymnema Sylvestre: An Important Medicinal Plant. BioMed Research International, 2014: 1–18. Link: https://goo.gl/mVhhRF
- Jain KS, Kathiravan MK, Somani RS, Shishoo CJ (2007) The Biology and Chemistry of Hyperlipidemia. Bioorg Med Chem 15: 4674–4699. Link: https://goo.gl/e1zqqJ
- Jamadagni PS, Pawar SD, Jamadagni SB, Chougule S, Gaidhani SN, et al. (2017) Review of Holarrhena Antidysenterica (L.) Wall. Ex A. DC.: Pharmacognostic, Pharmacological, and Toxicological Perspective. Pharmacogn Rev 11: 141–144. Link: https://goo.gl/YEGRkH
- Pal A, Sharma PP, Mukherjee PK (2009) A clinical study of Kutaja (Holarrhena antidysenterica wall) on Shonitarsha. AYU 30: 369–372. Link: https://goo.gl/JYnpJD
- Wang YC1, Huang TL (2005) Anti-Helicobacter Pylori activity Of Plumbago Zeylanica L. FEMS Immunol Med Microbiol 43: 407–412. Link: https://goo.gl/yGkM2m
- Iyengar MA, Pendse GS (1996) Plumbago Zeylanica L. (Chitrak) A Gastrointestinal Flora Normaliser. Planta Med 14: 337–351. Link: https://goo.gl/TbFd19
- Yadav V, Chatterjee SS, Majeed M, Kumar V (2015) Long Lasting Preventive Effects of Piperlongumine and a Piper Longum Extract against Stress Triggered Pathologies in Mice. J Intercult Ethnopharmacol 4: 277–283. Link: https://goo.gl/QG2xVS
- Sharma AL, Bhot MA, Chandra N (2014) Gastroprotective Effect of Aqueous Extract and Mucilage from Bryophyllum Pinnatum (Lam.) Kurz. Ancient Science of Life 33: 252–258. Link: https://goo.gl/YKNcV6
- Ranasinghe P, Pigera S, Premakumara GA, Galappaththy P, Constantine GR, et al. (2013) Medicinal Properties of ‘True’ Cinnamon (Cinnamomum Zeylanicum): a Systematic Review. BMC Complement Altern Med 13: 275. Link: https://goo.gl/YiRXp2

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
