Global Journal of Infectious Diseases and Clinical Research
Risk factors of adult Dengue Fever in urban community of Nepal
Harishchandra Shah1* and Basu Dev Pandey2
2MD, Ph.D, Professor, Department of Molecular Epidemiology, Institute of Tropical Medicine, Nagasaki, University, Japan
Cite this as
Shah H, Pandey BD (2021) Risk factors of adult Dengue Fever in urban community of Nepal. Glob J Infect Dis Clin Res 7(1): 064-069. DOI: 10.17352/2455-5363.000049Copyright
© 2021 Shah H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.With the objectives of determining the association between risk factors and Dengue Fever (DF), a case-control study with a random sample size of 102:102 in each was conducted at Hetauda and Kamalamai Municipalities, Nepal. The hypothesis of risk factors for DF like low level of knowledge about DF, discarded receptacles, old tires, containers, etc. nearby house, and use of stagnant AC/Coolers was used to conduct the study. Traveling to DF affected areas nearly 2 weeks before the onset of disease was significantly associated with dengue fever (OR= 6.10, 95% cl: 1.31-28.34, p<0.021). Waste disposal of old containers, receptacles, tires during the rainy season were significantly associated with the incidence of DF (AOR= 6.308, 96% cl: 2-751-14.462, p<0.000). The frequency of DF was associated with the middle social class level (p<0.05, d. f. 2). Uncovered water tanks of the household were significantly associated with DF (AOR= 3.78, 95% cl: 1.51-9.45, p<0.0043). As the number of families increases in the household, the number of cases increases with a positive correlation (r = +0.62). Crowded households with more than 2 occupants in one room were at risk of dengue infection. The study concluded that DF was associated with the risk factors of traveling to endemic areas, discarded waste containers, receptacles, tires, and uncovered water tanks, middle social class, and crowded households. Public health managers should prioritize these risk factors while planning for DF control and prevention.
Introduction
DF is an acute febrile disease transmitted by the bite of an Aides mosquito infected with any one of the four serotypes of dengue viruses (DEN-1, 2, 3, and 4). Dengue virus (DENV) belongs to the family Flaviviridae, the genus Flavivirus. Aedes aegypti and Aedes albopictus are the main vectors of DENV [1]. It is a significant public health problem in tropical and sub-tropical countries of the world [2]. There is an estimated 100-400 million infections each year. DVI is mostly found in tropical and sub-tropical climates worldwide, mostly in urban and semi-urban areas. Approximately two-thirds of the world’s population lives in areas infested by a mosquito [3,4]. DF, with its severe manifestations, Dengue Hemorrhagic Fever (DHF) and Dengue Shock Syndrome (DSS), has emerged as a great public health concern, spreading to all tropical and sub-tropical countries in the world [5].
The first dengue case was detected in 2004. The caseload has increased over time and in 2019 a total of 17 992 dengue cases were reported from 68 districts (from all seven provinces). Compared to the case incidence in 2016, the incidence was over 140 times higher in 2019 (IRR: 141.6; 95% CI 45.8–438.4) [6]. Outbreaks of DF have been reported nearly every year in the country during monsoon season mainly in the Terai region. Major outbreak years were: 2007,2009, 2010, 2013, 2016,2019, 2020 with serotypes DENV-1 and DENV1-2 in several districts of Nepal [7]. Currently, over 300 clinical cases are present in the country.
Literatures reviews have shown that waste disposal receptacles, old tires, pots, tins, etc. holding water inside the house or immediate surroundings, keeping flower pots inside the house or immediate surroundings, using cooler at home, non-use of aerosol mosquito repellants (coils/mats/liquid), use of impregnated mosquito nets, uncovered water tanks are associated with a high incidence of DF [4,5,8]. However, such socioeconomic, behavioral, environmental factors showing association with the incidence of DVI have neither been identified nor their strength of association been determined in the context of Nepal [1].
People’s behavior of disposing of discarded receptacles, containers, old tires, and tins is frequent. In Nepal, due to scarcity of water, water is stored in an uncovered water tank which usually becomes a source of dengue mosquito habitat at their houses. Habits of day-sleeping of the people in Nepal are very rare. However, some people particularly other than laborers take a small nap after lunch at mid-day during the summer and rainy seasons. Most people are day-workers at study areas.
The present study was intended to determine risk factors for DF associated with the incidence of disease so that the generated evidence may be utilized for preventing future epidemics, to find estimates (OR) of association between exposures and outcome DF and its strength of association.
Materials and methods
(Date: The study was conducted from Tuesday, June 15, 2021-Wednesday, September 15, 2021 AD).
An unmatched case-control study was conducted [9]. A mid-sized case-control study should nearly include 100 in each group [10]. Cases were randomly selected from the record of dengue patients of the Epidemiology Disease Control Division (EDCD), Department of Health Services (DHS). Kamalamai Municipality, of Sindhuli district, and Hetauda, of Makwanpur district, were selected conveniently. Both cases and controls were selected using the same criteria. The source of cases was the general population and the source of controls was neighborhoods from the same locality as cases. Controls were selected as far as possible of the similar age group of the cases; and the same sex as of cases. The number of cases was 102, and the number of controls was 102. The total number of subjects was 204. Children if selected for case or control might or might not be school attending. The methods for data collection were a personal interview with survey questionnaires and observation of the respondent’s households with a checklist. The software used for data analysis was Epi Info 7.2. Definition of the case: A case was defined as a person who had suffered from a DF/DHF/DSS diagnosed with a dengue test kit (RDT-Combo NS1, IgM, IgG) during 2020 and 2021 at the study areas without taking into consideration of rash and disease severity of cases. Definition of control: A control was a person who did not suffer from DF or dengue-like illness during the same period.
Results
The mean age of cases(n=102) was 40.94 years and that of controls(n=102) was 41.06 years. Their mean ages were not significantly different (P-value > 0.94956) as shown by the t-test. Cases and controls were homogenous in regards to age and sex. Male for cases and controls were 57 in each, a total of 114 (55.88%); and females for cases and controls were 45 in each, a total of 90 (44.12%). Sex Ratio for Male/Female = 1.26.
Analysis of important risk variables for cases and controls has been given in Table 1 showing exposure rates in cases and controls, odds ratios, 95% c. l., and their p-values. Since exposure rates are multiple on a single outcome, logistic regression was also performed to Adjust the estimates (AOR).
Social class: In our study, stratification was broadly classified into three major divisions of social class based on socioeconomic factors like wealth, income, race, education, ethnicity, gender, and occupation: upper class, middle class, and lower class. By considering the social classes, the cases and controls belonged to 3 different groups, high level, middle level, and lower level of social classes. The study found that the frequency of DF was associated with the middle social class level (p<0.05, d. f. 2).
Travel: Out of 102 cases, 12 (11.76%) cases of DF had traveled to DF affected areas before becoming diseased nearly 2 weeks before the symptoms started. Traveling to an epidemic area of dengue fever was significantly associated with the importation of DVI into the host community. The epidemic areas were in Nepal as well as in the towns of India at that time. The transmission of infection was six times likely to be greater than from a nonexposed. (OR= 6.10, 95% cl: 1.31-28.34, p<0.021), Table 2. After the endemic transmission had been established in the host community, epidemics in the community flared up.
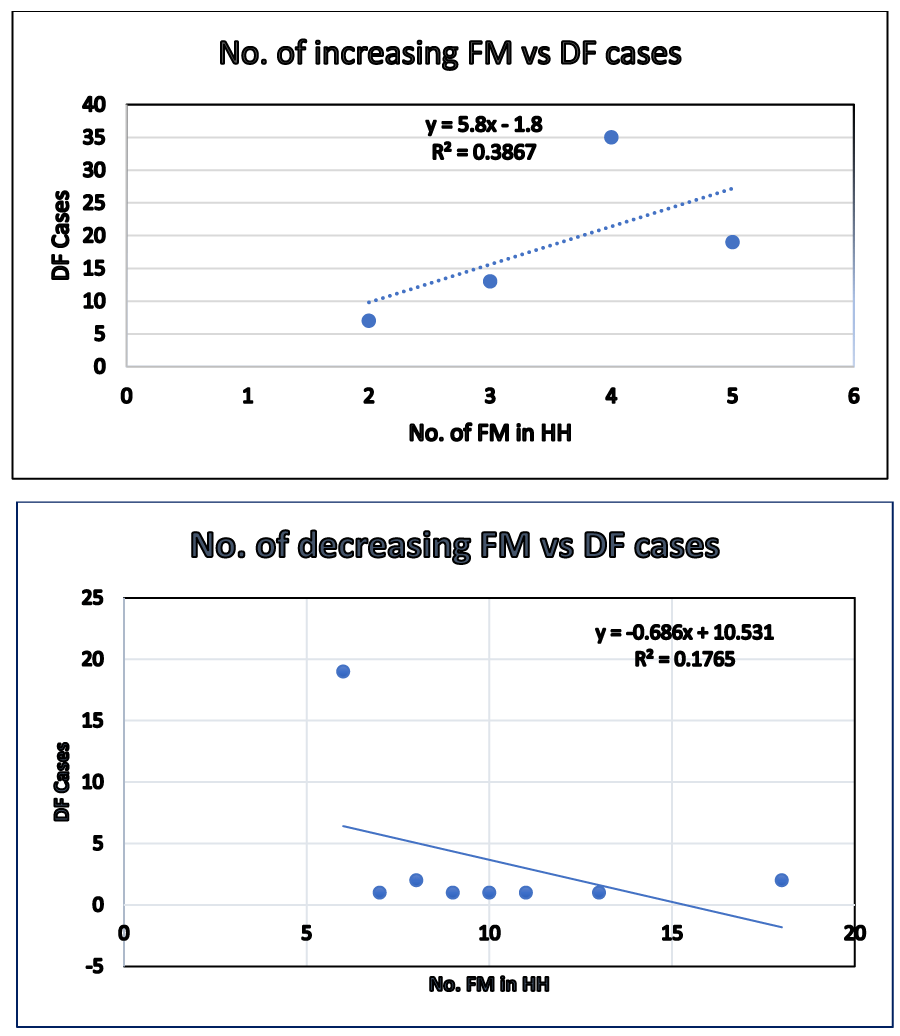
Table 3 showing No. of DF increases as FM increase.
Knowledge: Poor knowledge of dengue fever was a risk factor for DV. The low level of knowledge of DF was significantly associated with a high incidence of DVI (AOR=2.81, Cl: 1.30-6.11, p <0.0087), the risk of infection was nearly three times greater than nonexposed Table 2.
Mosquito repellents: The electrical mosquito repellents, mosquito liquid vaporizers and mosquito mat or coil repellents are significantly associated with protective effects against transmission of DVI, electrical mosquito killing devices (OR=0.10, 95% cl: 0.04-0.20, 0.000), liquid vaporizer (OR= 0.05, 95% cl: 0.01-0.16, p<0.000), mat/coils (OR= 0.19, 95% cl: 0.09-0.38, p<0.000). Besides, window/doors screening were also protective for DVI (OR= 0.50,95% cl: 0.28-0.88, p<0.015). However, on applying unconditional logistic regression the mosquito electrical repellents had significant results against DVI. (AOR=0.136, 95% cl: 0.058-0.318, p<0.000).
Tires/Receptacles: During the rainy season, water collected in the waste containers, tires, and receptacles attract mosquitoes for breading. This social behavior of keeping empty tires around houses was very widespread in urban or peri-urban areas. The alternative hypothesis is that discarded containers, waste receptacles, tires were significantly associated with transmission of infection of dengue virus. The risk of transmission was six times stronger than a nonexposed area (AOR= 6.308, 96% cl: 2-75-14.462, p<0.000). Tables 1,2.
AC/Coolers: The study has found that in an endemic area and the hot and rainy seasons, using stagnant cooler/AC at the house was associated with the high incidence of DVI (AOR= 8.28, 95% cl: 4.13-16.59, p = 0.000). Table 2.
Dirty water collection nearby houses: Our study result showed that during the rainy season, collection of dirty water around houses, favored breeding sites for mosquitoes (OR: 1.95, 95% cl:1.03-3.68, p=0.036). Table 1.
Open tank: Our present study has found that water storage in an open tank in the household was associated with transmission of DVI (AOR= 3.78, 95% cl: 1.51-9.45, p<0.0043). Table 2. The risk for transmission was 3.78 times likely of being stronger in an epidemic area than in a non-endemic area.
Mosquito nets: Our study has found a significant relationship between the use of mosquito nets, not impregnated with insecticide, and the likelihood of dengue virus infection (AOR=2.05, 95% cl: 1.07-3.93, p<0.031) as shown in the Table 2.
No. cases and family members (FM) of Households (HH): The culture of Nepal has the system of joint family from ancient time. As the number of FM increases in the household, the number of cases increases with positive correlation (r= +0.62) and R2 = 0.38 up to the FM of 5. Then, with continuing increase of FM, the number of dengue cases decreases with negative correlation (r = -0.42) and R2 =0 .17. Table 3, and Chart 1 and 2.
Categories of residents, either temporary or permanent, either for cases or for controls, were not statistically significant (P= 081). Houses made of bricks or stones, tins, and mud with thatches were not significant for cases and controls.
Indoor residual insecticidal spray (IRS) or fogging activities are usual government programs with the killing effects on adult mosquitoes. Due to environmental pollution threats, the government policy is discouraging the practice of IRS or fogging. These activities were not well conducted with insignificant effect (p= 0.31) during the epidemic period. Table 1.
Blocked drain nearby house: Water collected due to blocked drains is considered to be a potential source for producing larva of dengue mosquitos. The municipalities had implemented repairment activities for the blocked drainage system. The problem was not significant for cases and controls for contracting DF (OR= 1.00, 95% cl:0.47-2.08, p=1.000). Table 1.
Domestic garbage: In our study, it was not found as a risk factor for DF (OR=0.63, 95% cl:0.26-1.47, p=0.28). This might be due to regular garbage disposal at the municipalities.
Presence of under-construction building site nearby household: This is a usual problem in unorganized, peri-urban, or semi-urban areas. This variable may be associated with the likelihood of transmission of dengue virus infection during the rainy season within a distance of 100 meters. The site may be a favorable place for mosquitoes for larval breeding. However, this exposure has not been revealed as a risk factor for DF in our study areas (or= 0.84, 95% cl: 0.47-1.49, p =0.56). Table 1.
Discussion
Our study showed that the frequency of DF was significantly associated with the middle social class level (p<0.05, d. f. 2). In a study of Indonesia, the data suggested that dengue infections were linked to socio-economic factors, and the burden of dengue was, directly and indirectly, related to economic factors [11,12]. The severity of the disease varied according to the socioeconomic level of the patient [13,14]. Another similar study of China found that socioeconomic factors had a stronger influence on DF epidemics than environmental factors in the study area [15,16].
Our study found that traveling to an epidemic area of dengue fever was significantly associated with the importation of DVI into the host community; and six times likely to be greater than from a nonexposed area (OR= 6.10, 95% cl: 1.31-28.34, p<0.021). A similar study showed dengue epidemics were explosive with major economic, social, and political impacts [17-19]. Related studies have also shown that urbanization affected the propagation of DF throughout the world [20,21]. The risk of dengue importation was greatest in August, September, and October. A secondary vector of the virus, Aedes albopictus, is an Asian mosquito that can tolerate lower temperatures than A aegypti and has spread to North America and Europe due to increased international trade in used tires.
Our study found the low level of knowledge of DF was significantly associated with a high incidence of DVI (AOR=2.81, Cl: 1.30-6.11, p <0.0087) with 3 times greater risk of transmitting infection in the exposed group than non-exposed. The residents of the epidemic areas didn’t have much knowledge specifically on health education, information, and Communication (IEC) regarding transmission and prevention of DF. A similar study reported that knowledge had significant associations with dengue (p = 0.004) [13]. Another study found that there was a significant positive correlation between knowledge and attitudes, knowledge and practice, and attitudes and practice [22]. A study report showed that attitudes toward DF were significantly associated with the level of education and employment status (p<0.05) [23]. The study also demonstrated attitudes of the community towards home gardening, composting, waste management, and maintenance of a clean and dengue-free environment [24]. Understanding local knowledge, attitudes and practices was therefore essential to designing suitable strategies to fit the local context [25-28].
Our study found that the mosquito electrical repellents had significant results against DVI. (AOR=0.136, 95% cl: 0.058-0.318, p<0.000) showing protective effect. In a study of China, it was found that climate factors and mosquito density were the key drivers of DF incidence in different high-risk areas of China [29]. A Relevant study from China has shown an association between the monthly average temperature and with dengue occurrence, with a threshold of 23°C and the optimal range was 22–25 °C (F2.0, 9.7 = 195.4, p < 0.01). [30,31].
Discarded containers, waste receptacles, tires were significantly associated with transmission of infection of dengue virus with six times stronger risk in an exposed area than a nonexposed area (AOR= 6.308, 96% cl: 2-75-14.462, p<0.000). A study conducted in India found that availability of water-holding containers (p=0.005), and availability of stagnant water around houses (p=0.009) were important risk factors for dengue [32]. Breeding of Ae. aegypti was observed only in containers with non-potable water. Cement cisterns and tanks, stone tubs, and clay pots were the major breeding habitats of Ae. aegypti [12,33]. Vector surveys in the draining area revealed Aedes breeding with a high house index [34-36].
Our study has found that, in an endemic area and in hot and rainy seasons, using stagnant cooler/AC at the house was associated with the high incidence of DVI (AOR= 8.28, 95% cl: 4.13-16.59, p = 0.000). The households which had the coolers were at risk of eight folds greater than those that hadn’t. Previous studies have shown that users who never or rarely changed the water of coolers, or never used disinfectant to destroy larva of the stagnant water of AC/Cooler, were associated with the risk of transmission of DVI [5,7,8]. Because 85.71% of users of AC/Coolers did not change the water regularly at least once a week in our study areas.
Our study result showed that during the rainy season, collection of dirty water around houses had a significant association with DF with greater risk in the case group than the control group (OR: 1.95, 95% cl:1.03-3.68, p=0.036). Table 1. The problem was more common in unorganized peri-urban and slums areas. A study found that rapid urbanization in Southeast Asia brought a higher population density of infected mosquitoes to transmit the dengue virus [20]. Vector surveys in the draining area revealed Aedes breeding with a high house index [34]. Many under-construction houses, garbage dumps, and water pools, swamp areas, non-availability of proper water drainage system in the area, uncovered water storage in houses and open water tanks on roofs were sites for Aedes breeding [37].
Our present study has found that water storage in the open tank in households was associated with transmission of DVI (AOR= 3.78, 95% cl: 1.51-9.45, p<0.0043) with a 3.78 times stronger risk of transmitting infection in epidemic areas than a non-endemic areas. Similar relevant studies have shown significant associations of transmission of dengue virus infection with uncovered water storage in houses and open water tanks on roofs [33,37].
Our study has found a significant relationship between the use of mosquito nets, not impregnated with insecticide, and the likelihood of dengue virus infection (AOR=2.05, 95% cl: 1.07-3.93, p<0.031). This situation was found during acute out-break of DF in the municipalities. The result showed that mosquito nets did not prevent dengue fever since they were used at night yet dengue vectors bite early morning and late afternoon.
As the number of family members increased from 2 to 5 in the household, the number of DF increased showing a positive correlation (r = +0.62), then with continuing increase of FM from 6 to18, the number of dengue cases decreased showing -ve correlation (r= -0.32). In general, high population density is a risk factor for dengue transmission. The study found that crowded households with more than 2 occupants in one room were at risk of dengue infection. Parents who share rooms with their children pay additional attention to the use of anti-mosquito measures and the maintenance of good environmental sanitation to protect their children from mosquito bites. Moreover, high numbers of occupants in one room are associated with the decreased probability of mosquito bites when the number of mosquitoes was fixed [4].
Conclusion
Traveling to the epidemic area of dengue fever was significantly associated with infection of dengue virus (OR= 6.10, 95% cl: 1.31-28.34, p<0.021). Our study showed that the frequency of DF was significantly associated with the middle social class level (p<0.05, d. f. 2). During the rainy season, the risk factors of disposing of old containers, waste receptacles, tires were significantly associated with transmission of DVI. The risk of transmission was 6 times stronger than a non-epidemic area (AOR= 6.308, 96% cl: 2-75-14.46, p<0.000). The mosquito electrical repellents had significant protective effects against DVI. (AOR=0.136, 95% cl: 0.058-0.318, p<0.000). Our present study has found that water storage in the open tank in the household was associated with transmission of DVI (AOR= 3.78, 95% cl: 1.51-9.45, p<0.0043). As the number of FM increased from 2 to 5 in the household, the number of cases increased showing a positive correlation (r = +0.62). The study found that crowded households with more than 2 occupants in one room were at risk of dengue infection.
Recommendation
To conduct other case-control studies, preferably in different endemic geographic areas, to educate the public for promoting behaviors to destroy or manage mosquito vector larval habitats, to conduct an entomological study to identify the most productive larval habitats for their elimination, to promote personal protective against day-biting mosquitoes through repellents; screening of houses, improving housing conditions, frequent change of water of AC/Cooler at least once in a week or use of larvicides.
My cordial thanks expressed to my research supervisor, Dr. Basu Dev Pandey, former Director of Epidemiology and Disease Control Division of Department (EDCD) of Department of Health Services, Teku, Kathmandu, Nepal, and his subordinate officer Mr. Utam Raj Pyakurel, Senior Vector Control Officer. I am highly indebted to Mr. Pranay Upadhya, Senior Public health Administrator, Chief of District Public Health Office (DPHO), Hetauda, Makawanpur, and his subordinate officers, Mr. Suresh Pasman, Kamalamai Municipal, and his colleagues who supported me for data collection.
Ethical consideration
Every study participant was explained about the aims and objectives of the study and informed consent was also obtained from the participants for the publication of information. During the interview, cases and controls were assured anonymity and confidentiality about their data. Since the study did not use any manipulation or intervention to the subjects, however, the ethical approval was taken from Nepal Health Research Council (NHRC).
Limitation of the study: Though the study was conducted for the supplementary requirement of the Ph.D. course at Singhania University, Rajasthan, India, the author’s entire intention was to offer full accuracy and quality.
Funding: Entirely self-funding.
- Sah OP, Subedi S, Morita K, Inone S, Kurane I, et al. (2009) Serological study of dengue virus infection in Terai region, Nepal. Nepal Med Coll J 11: 104–106. Link: https://bit.ly/3Dzwo3c
- Savargaonkar D, Sinha S, Srivastava B, Nagpal BN, Sinha A, et al. (2018) An epidemiological study of dengue and its coinfections in Delhi. Int J Infect Dis 74: 41-46. Link: https://bit.ly/32WMzek
- Prasad Gupta B, Haselbeck A, Kim JH, Marks F, Saluja T (2018) The Dengue virus in Nepal: gaps in diagnosis and surveillance. Ann Clin Microbiol Antimicrob. 17: 32. Link: https://bit.ly/31AsAS4
- Liu J, Tian X, Deng Y, Du Z, Liang T, et al. (2019) Risk factors associated with dengue virus infection in Guangdong province: A community-based case-control study. Int J Environ Res Public Health 16: 617. Link: https://bit.ly/3y11fVi
- Chaudhry M, Ahmad S, Nisar M, Bin Rashid H, Hassan Mushtaq M, et al. (2015) A Matched Case-Control Study To Identify Potential Risk Factors Of Dengue Fever Among Residents Of A Local University, Lahore. 21. Link: https://bit.ly/3dpxtQz
- Rijal KR, Adhikari B, Ghimire B, Dhungel B, Pyakurel UR, et al. (2021) Epidemiology of dengue virus infections in Nepal, 2006–2019. Infect Dis Poverty 10: 1-10. Link: https://bit.ly/3pvhxC5
- Khetan RP, Stein DA, Chaudhary SK, Rauniyar R, Upadhyay BP, et al. (2018) Profile of the 2016 dengue outbreak in Nepal. BMC Res Notes 11: 1-6. Link: https://bit.ly/3lHxIed
- Maramraj KK (2018) Concurrent Chikungunya and Dengue Outbreak NSB Enclave, Kondapur, Rangareddy District, Telangana, July 2018. New Delhi, India.
- Gordis L (2009) Epidemiology. 4th editi. Merritt James, editor. Philadelphia, PA: Saunders Elsevier.
- Indrayan A (2008) Basic Methods of Medical Research (2nd edition: 2008). Indian J Community Med 33: 208. Link: https://bit.ly/3GolqQa
- Wijayanti SPM, Porphyre T, Chase-Topping M, Rainey SM, Mcfarlane M, et al. (2016) The Importance of Socio-Economic Versus Environmental Risk Factors for Reported Dengue Cases in Java, Indonesia. PLOS Neglected Trop Dis 10: e0004964. Link: https://bit.ly/3GjCp5P
- NadjibID M, Setiawan E, Putri S, Nealon J, Beucher S, et al. (2019) Economic burden of dengue in Indonesia. Econ Burd dengue Indones Mardiati 13: 1-14. Link: https://bit.ly/3DCEAju
- Malhotra V, Kaur P (2014) The community knowledge, attitude and practices regarding dengue fever in field practice area of urban training health centre of Patiala. Int J Res Dev Heal 2: 19–26. Link: https://bit.ly/3DpLSqA
- Nalongsack S, Yoshida Y, Morita S, Sosouphanh K, Sakamoto J (2009) Knowledge, attitude and practice regarding dengue among people in Pakse, Laos. Nagoya J Med Sci 71: 29–37. Link: https://bit.ly/338jexJ
- Liu K, Sun J, Liu X, Li R, Wang Y, et al. (2018) Spatiotemporal patterns and determinants of dengue at county level in China from 2005–2017. Int J Infect Dis 77: 96–104. Link: https://bit.ly/3EyE6vM
- Eldigail MH, Adam GK, Babiker RA, Khalid F, Adam IA, et al. (2018) Prevalence of dengue fever virus antibodies and associated risk factors among residents of El-Gadarif state, Sudan. BMC Public Health 18: 921. Link: https://bit.ly/3rHikSV
- Katzelnick LC, Coloma J, Harris E (2017) Dengue: knowledge gaps, unmet needs, and research priorities. The Lancet Infectious Diseases. Lancet Publishing Group 17: e88–100. Link: https://bit.ly/3EwxG0p
- Aparecida M, Figueiredo A, Rodrigues LC, Barreto ML, Wellington J, et al. (2010) Allergies and Diabetes as Risk Factors for Dengue Hemorrhagic Fever: Results of a Case Control Study. PLoS Negl Trop Dis 4: e699. Link: https://bit.ly/3lGLoGv
- Barrera R, Amador M, Mackay AJ (2011) Population Dynamics of Aedes aegypti and Dengue as Influenced by Weather and Human Behavior. PLoS Negl Trop Dis 5: e1378. Link: https://bit.ly/3lERPcY
- Je S, Bae W, Kim J, Seok SH, Hwang ES (2016) Epidemiological characteristics and risk factors of dengue infection in korean travelers. J Korean Med Sci 31: 1863-1873. Link: https://bit.ly/3Dr41nL
- Ali A, Nassar H, Abdullah M, Amad A (2018) Risk Factors Associated With Dengue FeverOutbreak in Taiz Governorate, Yemen, 2018: Case control Study. Res Square 1-14. Link: https://bit.ly/3IuIrT2
- Harapan H, Rajamoorthy Y, Anwar S, Bustamam A, Radiansyah A, et al. (2018) Knowledge, attitude, and practice regarding dengue virus infection among inhabitants of Aceh, Indonesia: A cross-sectional study. BMC Infect Dis 18: 96. Link: https://bit.ly/31rtf8R
- Mohamad M, Selamat MI, Ismail Z (2014) Factors Associated with Larval Control Practices in a Dengue Outbreak Prone Area. J Environ Public Health 2014: 459173. Link: https://bit.ly/304mqJC
- Udayanga L, Gunathilaka N, Iqbal MCM, Pahalagedara K, Amarasinghe US, et al. (2018) Socio-economic, Knowledge Attitude Practices (KAP), household related and demographic based appearance of non-dengue infected individuals in high dengue risk areas of Kandy District, Sri Lanka. BMC Infect Dis 18: 188. Link: https://bit.ly/3Dxbxhd
- Kumaran E, Doum D, Keo V, Sokha L, Sam BL, et al. (2018) Dengue knowledge, attitudes and practices and their impact on community-based vector control in rural Cambodia. PLoS Negl Trop Dis 12: e0006268. Link: https://bit.ly/3y03mJ5
- Mutheneni SR, Ganesh Kakarla S, Caminade C, Morse AP, Upadhyayula SM, et al. (2019) Epidemiology and Infection Lag effect of climatic variables on dengue burden in India. Epidemiol Infect 147: e170. Link: https://bit.ly/3ovd3ft
- Kumar Acharya B, Cao C, Xu M, Khanal L, Naeem S, et al. (2018) Present and Future of Dengue Fever in Nepal: Mapping Climatic Suitability by Ecological Niche Model. Int J Environ Res Public Heal 15: 187. Link: https://bit.ly/31zjSnt
- Eldigail MH, Abubaker HA, Khalid FA, Abdallah TM, Adam IA, et al. (2020) Recent transmission of dengue virus and associated risk Facors among residents of Kassala state, eastern Sudan. BMC Public Health 20: 530. Link: https://bit.ly/3IrfmIj
- Charette M, Berrang-Ford L, Coomes O, Llanos-Cuentas EA, Cárcamo C, et al. (2020) Dengue incidence and sociodemographic conditions in Pucallpa, Peruvian Amazon: What role for modification of the dengue-temperature relationship? Am J Trop Med Hyg 102: 180–190. Link: https://bit.ly/3pR7mbh
- Liu K, Hou X, Wang Y, Sun J, Xiao J, et al. (2019) The driver of dengue fever incidence in two high-risk areas of china: A comparative study. Sci Rep 9: 19510. Link: https://go.nature.com/3EAaWN4
- Thi Nguyen L, Xuan Le H, Thanh Nguyen D, Quang Ho H, Chuang TW (2020) Impact of Climate Variability and Abundance of Mosquitoes on Dengue Transmission in Central Vietnam. Int J Environ Res Public Heal 17: 2453. Link: https://bit.ly/33bnzQV
- Degife LH, Worku Y, Belay D, Bekele A, Hailemariam Z (2019) Factors associated with dengue fever outbreak in Dire Dawa administration city, October, 2015, Ethiopia - Case control study. BMC Public Health 19: 650. Link: https://bit.ly/336A4x0
- Arunachalam N, Murty US, Kabilan L, Balasubramanian A, Thenmozhi V, et al. (2004) Studies on dengue in rural areas of Kurnool District, Andhra Pradesh, India. J Am Mosq Control Assoc 20: 87–90. Link: https://bit.ly/3Ey52vQ
- Savargaonkar D, Sinha S, Srivastava B, Nagpal BN, Sinha A, et al. (2018) An epidemiological study of dengue and its coinfections in Delhi. Int J Infect Dis 74: 41-46. Link: https://bit.ly/31F2v4s
- Suwannapong N, Tipayamongkholgul M, Bhumiratana A, Poolthin S (2014) Effect of community participation on household environment to mitigate dengue transmission in Thailand. Trop Biomed 31: 149-158. Link: https://bit.ly/3Irp5hP
- Kumar Paul K, Dhar-Chowdhury P, Emdad Haque C, Mohammad Al-Amin H, Rani Goswami D, et al. (2018) Risk factors for the presence of dengue vector mosquitoes, and determinants of their prevalence and larval site selection in Dhaka, Bangladesh. PloS ONE 13: e0199457. Link: https://bit.ly/3GhWwBu
- Saeed S, Chaudhry A, Baig MA (2019) Outbreak investigation and a case control study of Dengue fever at Rawalpindi, Pakistan during July to September 2019. Int J Infect Dis 101: 249. Link: https://bit.ly/3rM1TVw
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
