Global Journal of Infectious Diseases and Clinical Research
Leishmania sis during the increased Syrian refugee traffic
Nurittin Ardic1*, Alper Fatih Ardic2 and Zeynep Gunel3
2Gazi University, School of Medicine, Ankara, Turkey
3Koc University, School of Medicine, Istanbul, Turkey
Cite this as
Ardic N, Ardic AF, Gunel Z (2018) Leishmaniasis during the increased Syrian refugee traffic. Glob J Infect Dis Clin Res 4(1): 013-019. DOI: 10.17352/2455-5363.000020Especially in recent years, important population mobility occurs worldwide, including refugee crisis affecting especially Middle East and Europe. Consequently, like other infectious diseases have significance for public health, Leishmaniasis is spreading globally. 350 million people in 88 countries, mostly in developing areas, are at risk of Leishmaniasis. The diseases may be seen in three clinical forms as cutaneous, mucocutaneous, or visceral caused by about 20 different species of Leishmania parasite. The parasitological diagnosis is made by microscopic examination, cultivation, PCR and serological methods. There is currently no available vaccine for this infection. Pentavalent antimonials are the first line therapeutic choice, whereas amphotericin B, pentamidine, miltefosine, and paromomycin are alternative drugs. Other side, increasing resistance and toxicity of the current therapeutics are significant problems. The combination therapy, immunotherapy, anti-Leishmanial synthetic and natural products, and local hyperthermic applications are promising for future treatment of Leishmaniasis. The preventive efforts have been applying based on individuals and community.In CHB patients, habits of drinking alcohol and cigarette smoking, elevated serum levels of TBil and serum AST/ALT ratio, increased duration of hepatitis B, a family of hepatitis B, male gender and older age can increase the risk of LC development.
Introduction
In last several years, an important population relocation occurs worldwide for various reasons, including immigrants, refugees, and international students [1,2]. In the US National Intelligence Council study (2000), it is reported that nearly two million people each day travel across borders, and it is estimated that ∼2% of the world’s population resides in a nation different than the one in which they were born [3]. This movement of people led to the spread of global infectious disease [4].
During the on-going conflict in Syria, which is one of the southern border neighbor of Turkey, about 6.5 million people are displaced within the country, as well as more than 4.2 million Syrians have crossed borders. Turkey hosts more than 2.7 million, Lebanon 1 million, Jordan 630,000 and Iraq 235,000 refugees, and approximately 682,000 asylum applications happened to Europe countries [5-7]. Cutaneous Leishmaniasis (CL) is highly endemic in Syria. In addition to some other infectious diseases, the number of Leishmaniasis, especially cutaneous form, cases increased in Turkey and the other countries depend on crowded and bad environments. The increased mobility due to the conflict can be considered an emerging new threat for unaffected regions [6]. Since these kinds of risk factors led to an increase in the variability of species and a rise in the frequency of infectious agents, it is required to reevaluate the treatment policy [8,9]. We focused on Leishmaniasis rather than other diseases because CL is a major health problem in Turkey. Therefore, we aimed to summarize the current knowledge on Leishmaniasis in this review.
Protozoan parasites of the genus Leishmania and the causative agent of Leishmaniasis diseases, are spread via the bite of female sand flies belonging to either Phlebotomus spp (Old World) or Lutzomyia spp (New World). Also, the parasite has about 20 different species; and the disease caused by the parasite could emerge as cutaneous Leishmaniasis (CL), mucocutaneous Leishmaniasis (MCL), or visceral Leishmaniasis (VL) (Table 1) depending upon the infecting species and the host’s immune status [10]. The visceral form could be fatal, while the skin lesions in cutaneous form tend to heal spontaneously, or scarification affecting life quality [11,12].
Epidemiology and etiology
The Leishmaniasis is widely seen across the tropical, subtropical, and temperate regions in 88 countries, mostly in developing areas. 350 million people are at risk of the diseases. Over 12 million people currently suffer from Leishmaniasis, with 500,000 new cases of VL per year and 1 to 1.5 million new cases of CL [13].
Cutaneous for of the disease is the most common in general. Different Leishmania species cause Old World (the Eastern Hemisphere) versus New World (American, the Western Hemisphere) CL [14]. In the Old World, the most common species that cause CL are L. tropica, L. aethiopica, and L. major; otherwise, in the New World, they are L. mexicana, L. amazonensis, L. braziliensis, L. panamensis, and L. guyanensis[12,15]. The causative agents of VL are commonly L. donovani in Indian subcontinent and Africa, L. infantum in Mediterranean, L. chagasi in Central and South America [13].
CL cases mostly occur in Afghanistan, Algeria, Brazil, Colombia, Iran, Pakistan, Peru, Saudi Arabia and Syria. The CL epidemiology in the Region of the Americas is complex depend on variation in transmission cycles, reservoir hosts, sandfly vectors [16].
The most endemic city in Turkey, which cutaneous form more common, is Sanliurfa with over 50% of 50381 cases between 1988-2010 years. Although Sanliurfa is in subtropical region of Turkey, some sporadic cases have been observed in the non-endemic west and north regions due to the population movements in recent years [1]. However Lebanon had no cases of CL before 2008, 96.6% of 1033 confirmed cases were among Syrian refugees by 2013, and some sporadic cases were seen in the following years [17]. Similarly, it was detected different Leishmania species between travelers, observed Leishmaniasis cases, to the Mediterranean and New World regions. This implicate that the species distribution is related to exposure region [18]. The refugees take huge supports for health by the government in Turkey, maybe in some other countries. Even if so, some factors such as health accessibility, reservoir hosts, sandfly vectors become important issues for Leishmaniasis cases in any big population movement.
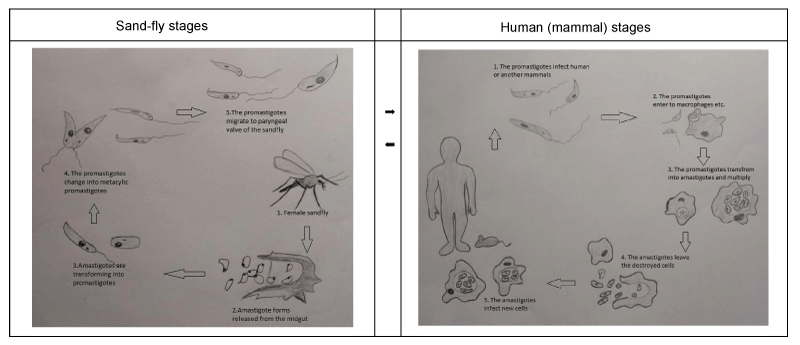
Upon the sand-fly injecting the promastigote form (1.5 to 3.5 by 15 to 200 µm) into the skin of the host, the parasites are taken up by macrophages. Then the promastigote forms differentiate into amastigote forms (3 to 5 µm in length) (Figure 1). Interestingly, the parasites replicate in the immune defense cells following the differentiation [13].
Immunologic response in Leishmaniasis
The host immunological response varies widely between species; especially Th1-type immune response has a crucial role in the infection control [13]. Parasitic clearance is happened by nitric oxide (NO) which is produced by macrophages. Some cytokines such as IFN-γ and TNF-α activate macrophages to produce nitric oxide, while cytokines like IL-4, IL-10, IL-13, and TGF-β dampen NO production, thereby decreasing the leishmanicidal ability of the cells [19,20]. Besides being an important effector cell, macrophage have a role in antigen-presenting to T cells [21]. On the other hand, macrophage can serve as a host for the replication and spread of the parasite by in inhibition of phagosome–endosome fusion, thereafter referring as Trojan horse [22]. In condition aparasite-driven Th2 response, it is seen a progression of the disease mediated by the expression of non-protectiveTh2-related cytokines such as IL-4, IL-10 and IL-13 [20]. Like this, Th1 suppressing cytokines such as TGF-β and IL-10 increase susceptibility to VL [23].
Clinical Manifestations of Leishmaniasis Visceral Leishmaniasis
Visceral form of the illness is a systemic infection, which affecting the liver, spleen, and bone marrow. The spectrum of VL varies from asymptomatic to life-threatening degree in severity. VL is caused by both New and Old World species [24,25].The incubation period ranges from as short as 10 days or longer than a year(usually 3-6 months)depending on the species of Leishmania, as well as the host’s immune status, age and the other factors [13].
The disease is known as kala-azar due to the characteristic darkening of the skin. The most common sign and symptoms are hepatosplenomegaly, hypergammaglobulinemia, pancytopenia, fever, and weight loss. The untreated VL may result in death frequently within two years because of hemorrhage, serious anemia, immunosuppression, and secondary infections. Sometimes, multiple, hypopigmented, erythematous macular lesions occur in skin following VL therapy. This is called as post-kala-azar dermal Leishmaniasis (PKDL) [20].Lesions in PKDL may resemble leprosy clinically and pathologically [13].
Cutaneous Leishmaniasis (CL)
Depending on species of the parasite and host factors, CL represents a broad spectrum in clinical manifestation. The hallmark for CL is skin lesions. The lesions are usually painless and can spontaneously heal in 2-10 months [23]. The incubation period of CL typically ranges from 2 weeks to several months, and sometime years. There is no evident systemic symptom. A species-specific immunity occurs following recovery or successful treatment of CL. Generally, L. tropica tends to cause “dry,” crusted, slowly enlarging lesions, while the lesions in L. major and L. braziliensis infections tend to be “moist,” exudative lesions and larger (Figure 2) [13].
The classic form of Old World CL is also known as “oriental sore” by a variety of colorful local expressions. The clinical spectrum in CL may vary from single or multiple, localized, cutaneous ulcers to diffuse cutaneous Leishmaniasis (DCL) [13]. Both New World and Old World CL may represent localized cutaneous involvement. Old World CL usually presents multiple lesions, while New World CL is usually associated with a solitary nodule [24].
DCL is usually associated with a defect in the cell-mediated immune response and anergy to Leishmania antigen. The initial lesion does not ulcerate, and then satellite lesions develop around the initial papule, and organisms gradually spread in the skin, resulting in dissemination. DCL is an uncommon syndrome mainly associated with L. aethiopica, L. mexicana, and L. amazonensis infection [24,25].
Leishmaniasis recidivans (LR) is an infrequent syndrome that may be seen years after resolution of a localized cutaneous lesion caused by L. tropica [24]. LR lesion is generally seen on the face. LR may infer from reactivation of dormant parasites or new infection with a different species [24,25]. LR has been demonstrated in North Africa, the Middle East, Southwest Asia, as well as Turkey and Iran [13].
Mucocutaneous Leishmaniasis (ML)
Mucocutaneous form of the disease, which has a significant rate of morbidity and mortality, is rarely seen in the Old World [26]. ML is mainly a problem of the New World. ML occurs in 2% to 5% of persons infected with L. braziliensis, and more rarely in persons infected with L. panamensis, L. guyanensis, or L. amazonensis [13]. ML is characterized by mucosal destruction and also known as “espundia” in South America. It usually develops by metastasis from disseminated protozoa rather than by local spread [23]. In ML, mucous membrane involvements usually occur in the nose, oral cavity, pharynx, or larynx months to years after their skin lesions recovered (Table 1) [13].
Diagnosis
The parasitological diagnosis of Leishmaniasis is made by microscopic examination, cultivation, PCR and serological methods. The samples obtained by biopsy, scrapings, and/or needle aspirates in the CL, or the specimens from peripheral blood, bone marrow, liver, or splenic aspirates in the VL are examined for parasitological investigation. The smears stained by Leishman, Giemsa, or Wright stains are examined under oil immersion microscope [24]. Specimens are inoculated into one of several media (Schneider’s modified media, Novy-MacNeal-Nicolle [NNN] media, and others) for culture of the parasites. A definite diagnosis can be made by amplifying Leishmania-specific DNA or RNA in a PCR assay. The serologic tests are usually used for VL. The serological methods using recombinant k39 (rk39) have revealed excellent sensitivity and specificity for the diagnosis [13].
Treatment
Currently, there is no available vaccine for the prevention Leishmaniasis. Furthermore, the variability in species of the parasite affects standard treatment recommendations negatively [13]. The first line therapeutic choice is pentavalent antimonials, sodium stibogluconate (SSG) and methylglucamine antimoniate (MA), for all forms of Leishmaniasis around the world. The drugs are given intramuscularly (IM), intravenously (IV), or intralesional (IL). Recommended IM and IV regimen is 15-20 mg SbV/kg of body weight daily for 21-28 days [27]. Painful injection, high toxicity and resistance problems are significant disadvantages of the drugs [28,29].
Amphotericin B with different formulations, pentamidine, miltefosine,and paromomycin with intramuscular and topical formulations are commonly used as alternative drugs in the treatment of Leishmaniasis [28,29].
Prevention
The preventive approaches to Leishmaniasis are handled based on individuals and community. Unfortunately, there is no individual immunoprophylactic prevention (vaccine or immunoglobulin) method for travelers to endemic areas. Standard personal protective measures are recommended applying permethrin or other insecticides to clothing and insecticide-impregnated fine-mesh bed nets. In endemic areas, due to zoonotic features of the disease, the vector control and reservoir control applications are effective for community-based preventive efforts. Residual insecticides can be applied in houses and other buildings. The early diagnosis and complete treatment, integrated vector management and effective vector and disease surveillance establish basis of elimination of the infection [13].
Conclusion
Although Leishmaniasis is an old disease, it remains as a significant public health. The disease is spreading and globalizing due to the heavy population movement. To prevent and treat the disease effectively, an important task awaits the scientists of the future, including us.
- Harman M (2015) Cutaneous Leishmaniasis. Turk J Dermatol 9:168-176. Link: https://goo.gl/YtjgKy
- Park JJH, Adamiak P, Jenkins D, Myhre D (2016) The medical students’ perspective of faculty and informal mentors: a questionnaire study. BMC Med Educ 16: 4. Link: https://goo.gl/7C39H2
- Gushulak BD, MacPherson DW (2004) Globalization of infectious diseases: the impact of migration. Clinical Infectious Diseases 38: 1742–1748. Link: https://goo.gl/Npbos6
- EASAC (2007) Impact of migration on infectious diseases in Europe. Royal Soc 1-8,www.easac.eu.
- Refugee crisis Situation Update 1 (2018) WHO Rgeional office for Europe. Link: https://goo.gl/RNfZnZ
- Leblebicioglu H, Ozaras R (2015) Syrian refugees and infectious disease challenges. Travel Med Infect Dis 13: 443-444. Link: https://goo.gl/5grhed
- Syria Regional Refugee Response Inter-agency Information Sharing Portal (2016) Link: https://goo.gl/WTtdMV
- Saroufim M, Charafeddine K, Issa G (2014) Ongoing epidemic of cutaneous leishmaniasis among Syrian refugees, Lebanon. Emerg Infect Dis 20: 1712-1715. Link: https://goo.gl/oNYNLg
- Turan E, Yeşilova Y, Sürücü HA (2015) A Comparison of Demographic and Clinical Characteristics of Syrian and Turkish Patients with Cutaneous Leishmaniasis.Am J Trop Med Hyg 93: 559-563. Link: https://goo.gl/uQ1ike
- Salam N, Al-Shaqha WM, Azzi A (2014) Leishmaniasis in the middle East: incidence and epidemiology. PLoS Negl Trop Dis 8: e3208. Link: https://goo.gl/uCUkTt
- Alawieh A, Musharrafieh U, Jaber A (2014) Revisiting leishmaniasis in the time of war: the Syrian conflict and the Lebanese outbreak. Int J Infect Dis 29: 115-119. Link: https://goo.gl/ss4Dkt
- Mears ER, Modabber F, Don R, Johnson GE (2015) A Review: The Current In Vivo Models for the Discovery and Utility of New Anti-leishmanial Drugs Targeting Cutaneous Leishmaniasis. PLoS Negl Trop Dis 9: e0003889. Link: https://goo.gl/YCDXvs
- Magill AJ (2015) Leishmania Species: Visceral (Kala-Azar), Cutaneous, and Mucosal Leishmaniasis. In: Bennett JE. Dolin R, Blaser MJ (eds). Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 8th ed., Saunders/Elsevier, Philadelphia 3091-3107.
- Link: https://goo.gl/UoTfz4
- Harhay MO, Olliaro PL, Costa DL, Costa CH (2011) Urban parasitology: visceral leishmaniasis in Brazil. Trends Parasitol 27: 403-409. Link: https://goo.gl/GaTYz4
- Link: https://goo.gl/k216Qr
- Sharara SL, Kanj SS (2014) War and Infectious Diseases: Challenges of the Syrian Civil War. PLoS Pathog 10: e1004438. Link: https://goo.gl/mnxU4T
- Wall EC, Watson J, Armstrong M, Chiodini PL, Lockwood DN (2012) Short Report: Epidemiology of Imported Cutaneous Leishmaniasis at the Hospital for Tropical Diseases, London, United Kingdom: Use of Polymerase Chain Reaction to Identify the Species. Am. J Trop Med 86: 115–118. Link: https://goo.gl/6b1Kc7
- Cummings HE, Tuladhar R, Satoskar AR (2010) Cytokines and Their STATs in Cutaneous and Visceral Leishmaniasis. J Biomed Biotechnol 294389. Link: https://goo.gl/jQ5BPd
- Natarajan G, Oghumu S, Varikuti S, Thomas A, Satoskar AR (2014) Mechanisms of Immunopathology of Leishmaniasis. In: Pathogenesis of Leishmaniasis: New Developments in Research. Springer Science+Business Media New York.
- Nagill R, Kaur S (2011) Vaccine candidates for leishmaniasis: a review. Int Immunopharmacol 11: 1464-1488. Link: https://goo.gl/RukoCp
- Ritter U, Frischknecht F, van Zandbergen G (2009) Are neutrophils important host cells for Leishmania parasites? Trends Parasitol 25: 505-510. Link: https://goo.gl/WmuCn1
- Murphy ML, Wille U, Villegas EN, Hunter CA, Farrell JP (2001) IL-10 mediates susceptibility to Leishmania donovani infection. Eur J Immunol 31: 2848–2856. Link: https://goo.gl/R2eMJX
- Stark CG, Vidyashankar C, Chandrasekar PH (2016) Leishmaniasis. Link: https://goo.gl/ud2Kea
- Aronson N, Weller PF, Baron EL (2018) Clinical manifestations and diagnosis of cutaneous leishmaniasis. Link: https://goo.gl/Scbs1d
- Yeşilova Y, Aksoy M, Sürücü HA, Uluat A, Ardic N et al. (2015) Yesilova A.Lip leishmaniasis: Clinical characteristics of 621 patients. Int J CritI lln Inj Sci 5: 265-266. Link: https://goo.gl/Y7hiCH
- Lindoso JA, Cota GF, QueirozIA, Goto H (2012) Review of the current treatments for leishmaniasis. Research and Reports in Tropical Medicine 3: 69–77.
- de Menezes JP, Guedes CE, Petersen AL, Fraga DB, Veras PS (2015) Advances in Development of New Treatment for Leishmaniasis. Biomed Res Int 815023. Link: https://goo.gl/9M3DQJ
- Monzote L (2009) Current Treatment of Leishmaniasis: A Review. The Open Antimicrobial Agents Journal 1: 9-19. Link: https://goo.gl/CDbbnE
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
