Forensic Sci Today
Postmortem Neurochemistry Shows High Dopamine Levels in Brain of Cocaine Consumers
Virginia M Carvalho1*, André R Fukushima2, Luiz R Fontes3, Jorge C Florio2 and Alice AM Chasin4
2Universidade de São Paulo, Faculty of Veterinary Medicine and Animal Science, Department of Pathology, Av. Dr. Orlando Paiva, 87, 05508-270, São Paulo, Brazil
3Legal Medical Institute of São Paulo, Av. Dr. Enéas de Carvalho Aguiar, 600, 05403-000, São Paulo, Brazil
4Universidade de São Paulo, Faculdade de Ciências Farmacêuticas, Department of Clinical and Toxicological Analysis, Av. Lineu Prestes, 580, 05508-900, Sao Paulo, Brazil
Cite this as
Carvalho VM, Fukushima AR, Fontes LR, Florio JC, Chasin AAM (2016) Postmortem Neurochemistry Shows High Dopamine Levels in Brain of Cocaine Consumers. Forensic Sci Today 2(1): 013-019. DOI: 10.17352/fst.000007Cocaine abuse represents a serious problem to many countries and is frequently related to deaths. Differences in response, drug tolerance, drug sensitization and postmortem redistribution difficult clarifying cocaine related deaths. Thus, additional parameters associated to cocaine exposition could assist the pathologist in interpreting the deaths cases. In order to verify whether the cocaine action mechanism indicates a postmortem biochemical profile, dopamine and serotonin levels were quantified in brain samples. Two groups were established, one composed by cocaine consumers and other composed by non-cocaine consumers. Levels of dopamine and its metabolites were higher in basal ganglia of cocaine consumers group, but the concentrations were weakly correlated with cocaine concentrations in brain and in whole blood. Although catecholamine at autopsy may show considerable variations due to various factors, the high dopamine and metabolites levels can be associated with the cocaine action mechanism which is based on increased of dopamine levels in dopaminergic pathways in the central nervous system.
Introduction
Cocaine abuse represents a serious concern to many countries. While the United Nations Office on Drug and Crimes (UNODC) estimates that 17 million people used cocaine at least once in the past year (0.37% of the global population aged 15-64) [1], the last survey conducted in Brazil indicates a cocaine consumption rate of 2.2% (3.7% in men and 0.7% in women) and prevalence of addiction of 0,6 % and 41,4 % in total population and cocaine consumers, respectively [2].
Cocaine (COC) is a strong central nervous system (CNS) stimulant and enhances dopamine (DA) neurotransmission by interacting with DA transporter and inhibiting the clearance of extracellular DA. DA is metabolized into 3,4-dihydroxyphenylacetic acid (DOPAC) and homovanilic acid (HVA) by the catechol-O-methyltransferase and monoamine oxidase. Furthermore, cocaine blocks serotonin (5-HT) transporter in the CNS [3] that is involved in appetite control, sleep regulation, memory and learning establishment, temperature regulation, depression, etc. 5-HT is metabolized into 5-hydroxyindolacetic acid (5-HIAA) by aldehyde dehydrogenase and monoamine oxidase.
Cocaine is associated with cardiovascular complications, violent behavior and is frequently related to deaths examined in medical legal departments. Cocaine can be implicated in the direct cause of death or a significant condition contributing to death. The diagnostic criteria for COC overdose commonly include a review of police investigation, autopsy, identification and quantification of COC and its metabolites in the blood [4,5]. COC concentrations of 1 µg/mL or more were referred in fatal cases (6), but when blood is not available, brain is an interesting alternative sample to determine the cocaine use, considering brain cocaine/benzoylecgonine ratio can give an idea if the exposure was recent or not and is the psychoactive drugs target organ, it is inside an isolated compartment, and postmortem drug concentrations are close or equal to perimortem concentrations [7-9]. Moreover, COC concentrations in body fluids or tissues are influenced by various chemical changes, postmortem redistribution, chronic use and individual susceptibility to the drug.
Interpreting deaths related indirectly to COC is a challenging task because concentrations do not explain the cause of death, especially in chronic use with cardiovascular complications or bizarre behaviors like jumping of buildings or running in a hard traffic road stimulated by COC psychoactive effect. Thus other biological parameters associated with COC consumption should be investigated.
Postmortem analysis of biochemical markers might aid in characterizing causes of death. Hypothermia has been associated with increased ketone levels in blood and other biological fluids, increased cortisol levels in postmortem serum and urine, and increased adrenaline concentrations in urine [10]. The concentrations of β-hydroxybutyrate in vitreous humor and blood are correlated and high levels were found in fatal ketoacidosis cases associated to diabetes mellitus [11], adrenaline/noradrenaline ratios were increased long agony in comparison to short agony death cases in blood, liquor, urine and vitreous humor [12] and high serum catecholamines levels were associated to fatal methamphetamine abuse (adrenaline and dopamine) and hyperthermia (dopamine) [13]. In addition, hydroxytryptophol (5-HTOL)/5-hydroxyindolacetic acid (5-HIAA) ratio increases after alcohol consumption and is a biomarker for differentiating between ethanol produced from glucose and other substrates by putrefactive process, since ethanol alters the catabolism of serotonin to metabolites 5-HIAA and 5-HTOL [14-16].
Current literature shows that biochemical markers in various biological specimens are useful to complement autopsies and toxicological findings. On the other hand, few studies have found association between neurotransmitters in brain with COC consumption. It was referred increased dopamine transporter binding sites in COC consumers [17] and decreased dopamine transporter levels in excited delirium cases, most of them cocaine and other psychostimulants consumers [18]. Brain is an isolated compartment and polar molecules do not cross the blood brain barrier [19], thus cathecolamines and indoleamines concentrations are directly related to alterations in this compartment. Considering COC actions on dopaminergic pathways in CNS, the present study compared the differences in DA and 5-HT levels between brain samples of COC consumer group and COC free control group.
Material and Methods
Postmortem brain and blood samples from COC consumers (n=18) and non COC consumers (COC-free control subjects) (n=21) were obtained during routine autopsy between April 2009 and May 2010 at the Medical Legal Institute of São Paulo. Subjects’ age range was between 15 and 55 years old and maximal post mortem interval was 36 hours. Autopsy findings, family and police report were recorded.
The COC free group was composed by individuals whose urine samples were negative for cocaine and benzoylecgonine (BE) by immunoassay test (screening test) and by mass spectrometry. In addition, the negativity to cocaine and benzoylecgonine was tested in whole blood, vitreous humor and brain tissue by High Performance Liquid Chromatography with Diode Array Detector (HPLC-DAD) method.
COC and BE were quantified in brain tissue (BT) and whole blood (WB) by HPLC-DAD method, as previously published [9]. Basal ganglia (whole structure: caudate, putamen and globus pallidus) and prefrontal cortex (circa 25 g) samples were collected from the right brain hemisphere. Samples were stored at -20ºC and transferred to laboratory in no more than 12 hour after collection. In laboratory samples were homogenized and stored at -80ºC until the time of analysis.
Blood alcohol levels were measurement by head space gas chromatographic method at Forensic Laboratory of Medical Legal Institute of São Paulo (Limit of Detection=0.01 g/L).
DA, DOPAC, HVA, 5-HT and 5-HIAA were quantified in brain samples, basal ganglia (BG and prefrontal cortex (PC) by reverse-phase High Performance Liquid Chromatography with Electrochemical Detection (HPLC-ED), (model 6A, Shimadzu instrument), as described by Felicio et al. [20]. Briefly, brain tissue (100 mg) were homogenized in 0.1 M perchloric acid (2.0 mL) with a micro-ultrasonic cell disrupter (Kontes, Vineland, NJ, USA), stored at 4 °C overnight, and centrifuged at 10,000 rpm/30 min at 4°C. The supernatant (20 µL) was injected into the HPLC-ED. The chromatographic conditions were reversed phase with C18 column, mobile phase pH 3.0 at isocratic mode was a mix of citrate buffer 0.02 M: methanol (92:8 v/v), ethylenediamine tetraacetic acid (EDTA) and 1-heptanesulfonic acid, the flow was 1.0 mL/min, column temperature was 55ºC and detector was operated at 0.08V. Dihydroxybenzylamine (DHBA), 27.8 ng/mL, was used as internal standard. The limit of detection and limit of quantification were 10 ng/g and 20 ng/g for all analytes. Obtained values are expressed as ng/g tissue wet weight.
Results
The study population (n=39) represented 1% of autopsies performed between April 2009 and May 2010, most individuals were young, age between 15 and 24 years old and male (Table 1). The age mean of COC free group (mean = 31, SEM ± 2) shows no significant difference from COC consumer group (mean = 28, SEM ± 2) (Figure 1). Many death circumstances were referred, such as, homicide, sudden death, running over, suicide, electrocution, natural causes, etc. All sudden death and suicide by precipitation cases were found in the cocaine consumer group.
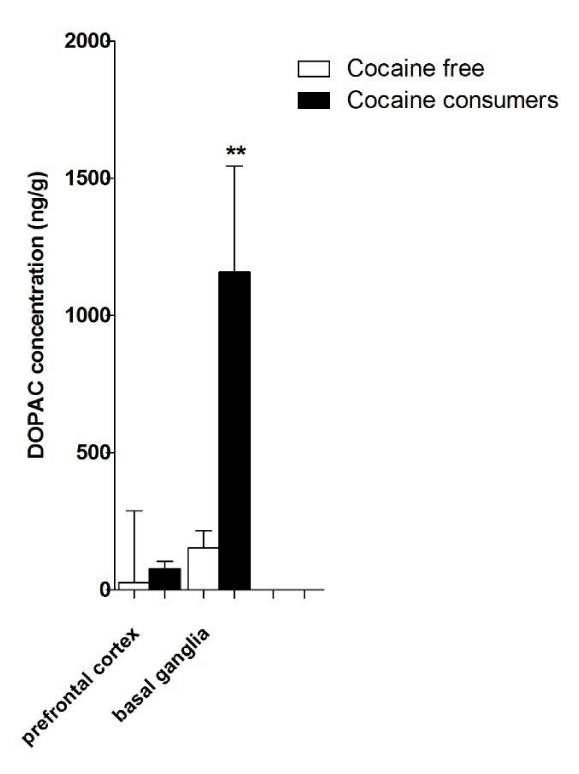
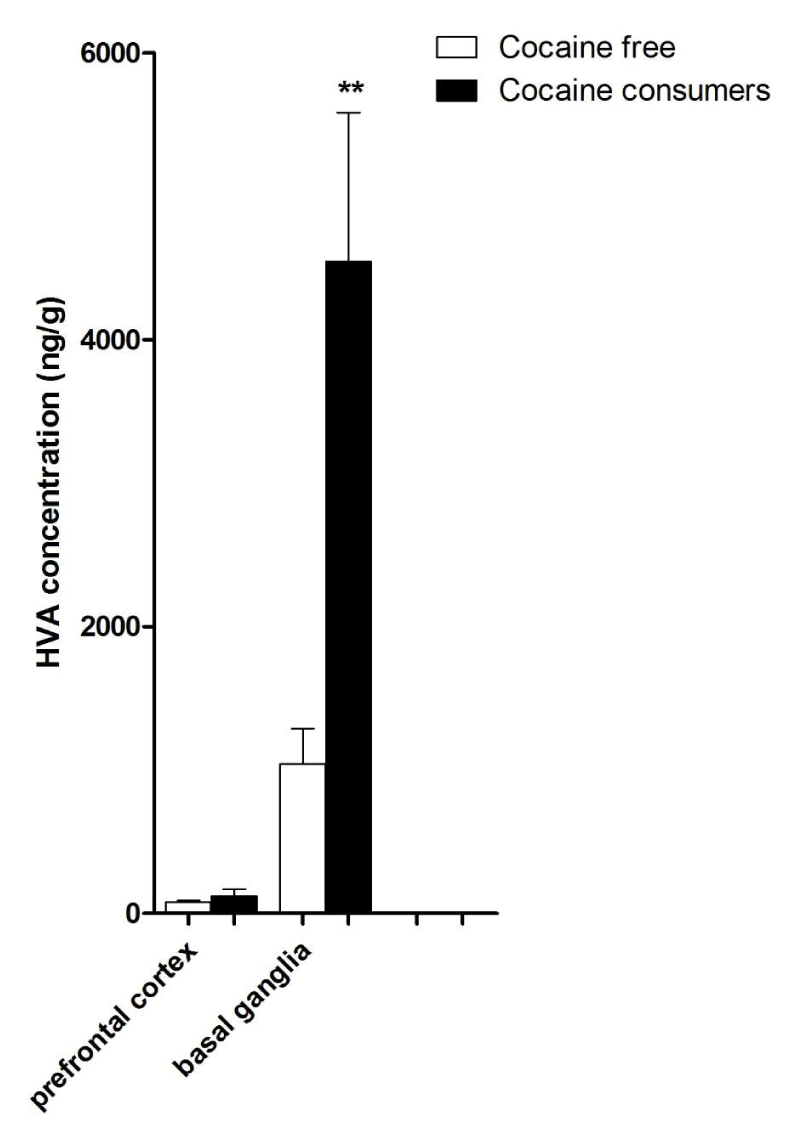
Neurotransmitters were measured in both groups and the mean concentrations are shown in Table 2. Table 3 shows cocaine, benzoylecgonine and catecholamine levels in samples of COC consumer group and Table 4 shows catecholamine levels measured in cocaine non consumer group. Dopamine and its metabolites mean concentrations were higher in COC consumer group and Student t test showed significant differences for DA, DOPAC and HVA in basal ganglia in regard to cocaine free group (Figures 2-4). Correlation tests were performed, COC levels in basal ganglia showed no correlation with DA (Pearson r=0.3759, p=0.1242) and DOPAC (Pearson r=-0.1156, p=0.6477) levels, but significant correlation was found between COC and HVA levels in basal ganglia (Pearson r=0.4996, p=0.0348). Similarly, COC levels in whole blood showed no correlation with DA (Pearson r=0.3995, p=0.1005) and DOPAC (Pearson r=-0.1248, p=0.6216) levels in basal ganglia, but significant correlation was found between COC levels in whole blood and HVA levels in basal ganglia (Pearson r=0.4971, p=0.0358).
Discussion
In clinical toxicology practice the toxic agent in a biological matrix is frequently evaluated together with biochemical markers, as for example, carbamate and organophosphate pesticides intoxication are associated with cholinesterase inhibition and lead exposition is associated with alterations in parameters of hemoglobin production. On the other hand, the biochemical profiles at autopsy may show considerable variations due to various factors involving preexisting disorders, cause of death, survival periods, agony time and postmortem changes. Such changes depend on environmental factors and also on the chemical properties, distribution and location of analytes [12,21]. For instance, catecholamines were demonstrated as a parameter that suffers increase during survival period [13,21] and a great individual variability was found in catecholamine concentrations of different biological specimens, such as, femoral vein blood, heart blood, urine, vitreous humor and liquor [12]. Nonetheless, knowing the postmortem biochemical profiles may be useful in interpreting cases in which toxicology results do not explain the death, especially when drug levels do not characterize a classical overdose. An illustrative case is a 37-year-old man, regular consumer of heroin, cocaine and methadone who died after using cocaine. A friend stated that the man consumed crack cocaine with him on the previous day and on day of the death he stayed in bed, was extremely tired and vomited several times, while his friend did not suffer from any physical problems. The toxicological analysis showed low levels (not sufficient to cause death) of morphine, mirtazapine buprenorphine, nordiazepam and oxazepam in blood and cocaine in urine confirmed its use. Although potential pharmacological interactions of multiple drugs could have contributed to the death, biochemical parameters analyzed in biological specimens, such as ketone, isopropyl alcohol, glucose and glycated hemoglobin, histological and immunohistochemical analyses suggested severe diabetic ketoacidosis as the cause of death [22]. Despite the difficulty to interpret postmortem biochemical changes, our study shows different neurochemistry profiles in two groups, one formed by 18 known cocaine consumers and another composed by 21 non cocaine consumers. Neurotransmitters concentrations in both groups were higher in basal ganglia than in prefrontal cortex. Basal ganglia show more dopaminergic and serotonergic pathways than the prefrontal cortex and our findings indicated that the distribution pattern in vivo is sustained after death. DOPAC and HVA higher concentrations relative to DA, and 5-HIAA higher concentrations compared to 5-HT can indicate neurotransmitter degradation. 5-HT is highly sensitive to degradation by oxidative mechanism, and as there were no differences in 5-HT and 5-HIAA levels between groups and these parameters did not seem useful markers to interpret cocaine related deaths.
DA is also sensitive to degradation, when neurochemistry is studied in rats it is recommended fast brain structures collection after decapitation to prevent analytes degradation [20], but in autopsy routine time control is impossible because each procedure spends different time and the population presents different post mortem intervals. Although the high neurotransmitter metabolites concentrations probably resulting from postmortem degradation, DA, DOPAC and HVA concentration means showed significant difference between the two groups (COC consumers and COC non consumers). Higher catecholamine concentrations were show in cases of overdose and shooting after violent behavior and resisting arrest (COC consumer group, Table 3) and in two cases involved electric shock in COC non consumers group, the first with history of cardiopulmonary resuscitation after subarachnoid hemorrhage and the second electrocution (Table 4).
Cocaine increases dopamine levels in dopaminergic pathways and its mechanism action can be associated with increased dopamine and its metabolites levels in cocaine consumers group. A postmortem study conducted with 34 cocaine users and 36 control subjects showed that striatal dopamine transporters binding sites were increased in cocaine users [17], indicating that it is possible to associate cocaine use with biochemical parameters related to its action mechanism in cadaveric samples. The association between neurotransmitter levels in postmortem brain and drug consumption was also demonstrated in a study performed with chronic heroin users. Research showed a reduction in DA, HVA and DOPAC concentrations in caudate, putamen and nucleus accumbens of chronic heroin users with postmortem interval of 13 ± 2 hours (n=9). It was suggested that decreased dopaminergic activity in the nucleus accumbens was a compensatory response to prolonged dopaminergic stimulation by heroin [24]. Cocaine also stimulates the dopaminergic system, however in our study the COC consumer population was a heterogeneous group and it was not possible to identify chronic users, thus hindering a comparison. Possibly, for this reason, the correlation between cocaine and dopamine and between its metabolites concentrations was confused. It was observed a weak correlation between cocaine levels in whole blood and HVA levels in basal ganglia and the same was found between cocaine levels in basal ganglia and HVA in basal ganglia. The variability related to the cause of death, cocaine dose, frequency of use and interval between cocaine use and death certainly impair the correlation studies. An illustrative case is a sudden death with COC chronic use history (Table 3), young man, 18 years old, 170 cm height, 60 kg weight, autopsy findings were enlarged heart (cardiomegaly), lungs congested with petechiae and brain edema, the toxicological findings were high COC and BE concentrations in BG and WB that can be associated to tolerance phenomena and COC levels are 4 times higher than BE levels in BG which indicating a recent cocaine use [9]. Although COC levels in blood (6.95 µg/mL) indicated an overdose, the catecholamine levels were relatively low in comparison with a massive acute intoxication presented in a body packing case (Table 3). Thus dopamine levels can be an additional marker to interpret cocaine related deaths in complement of cocaine levels. Mash et al. (2009) demonstrated reduction in the dopamine transporter levels for all victims of excited delirium, including non-drug abuse consumers (drug abuse free cases). Excited delirium (ED) has been showed generally after immobilization with a conductive energy device and is a syndrome characterized by delirium and agitation, combativeness, unexpected strength and elevated temperature. Decrease in DA transporter levels in an ED group composed mostly by COC and other psychostimulants consumers’ would be the cause of extracellular DA levels increased [18].
It is well known that alcohol intensifies the neurotoxicity and cadiotoxic effects of COC by cocaethylene production which is more liposoluble than COC. Curiously, two cases with high alcohol blood levels, 0.7 and 3.73 g/L, showed also high HVA levels, 15.295 and 6.236 µg/g, respectively (Table 3). Off course other more studies are necessary to any inference.
Although dopamine depletion is a theory proposed to explain dysphoric aspects of cocaine abstinence after overstimulation of DA neurons and excessive synaptic metabolism of the neurotransmitter in chronic use [25], DA receptors and transporters density depends on duration of use and abstinence [26]. Age it also seems other variable associated with dopamine activity, Volkows et al. (1998) showed reductions in dopamine D2 receptor and a significant correlations between D2 receptor availability and age in the caudate and putamen in living healthy individuals (age range=24-86 years old) [27], but Eidelberg et al. (1993) demonstrated that uptake rate constants of a marker to assess DA presynaptic nigroestriatal function do not decline with normal aging in living healthy individuals (age range=27-77 years old) [28]. In our study most of population was young and there was no significant difference between the age mean of the two groups (COC free and COC consumer).
While our results clearly indicate an association between high DA levels and its metabolites in basal ganglia and cocaine positivity, further studies are needed to investigate the practical application in legal medicine. In the present study the sample population was small (N=39 individuals) and non COC consumers (COC-free control subjects) were classified by the urine, blood and brain analysis that indicate the recent use of cocaine, less than 2 days before the death for a half-life of up to 7.3 hour by intranasal administration [29]. Although in future studies hair analysis should be performed to improve the neurochemistry interpretation, controlling all the variable that can be influence neurochemistry is impossible in the context of legal medicine whose purpose is to clarify facts by the vestiges of the corpse and usually its whole life history is not available.
Considering the biological processes of life and death occur in multivariate and integrated way, the contribution of present study was to demonstrate that the comparison of neurochemistry markers and toxicological findings can be valuable to clarify cocaine related deaths.
- World Drug Report (2013) United Nations Office on Drugs and Crimes, Vienna. Link: https://goo.gl/yYKFzg
- Abdalla RR, Madruga CS, Ribeiro M, Pinsky I, Caetano R, et al. (2014) Prevalence of Cocaine Use in Brazil: Data from the II Brazilian National Alcohol and Drug Survey (BNADS). Addict Behav 39: 297-301. Link: https://goo.gl/2ji3oF
- Carboni E, Spielewoy C, Vacca C, Nosten-Bertrand M, Giros B, et al. (2001) Cocaine and Amphetamine Increase Extracellular Dopamine in the Nucleous Accumbens of Mice Lacking the Dopamine Transporter Gene. J Neurosci 21: 1-4. Link: https://goo.gl/R0gIi4
- Darke S, Kaye S, Duflou J (2005) Cocaine-related fatalities in New South Wales, Australia 1993-2002. Drug Alc. Depend 77: 107-114. Link: https://goo.gl/2jtJ5j
- Jenkins AJ, Levine B, Titus J, Smialek JE (1999) The interpretation of cocaine and benzoylecgonine concentrations in postmortem cases. Forensic Sci Int 101: 17-25. Link: https://goo.gl/Tg3cfz
- Takekawa K (2005) Cocaine and its metabolites. In Suzuki O Watanabe K Drugs and Poisons in Humans a Handbook of Practical Analysis. Heidelberg: Springer 2: 207-218. Link: https://goo.gl/hKaHys
- Spiehler VR, Reed D (1985) Brain concentrations of cocaine and benzoylecgonine in fatal cases. J Forensic Sci 30: 1003-1011. Link: https://goo.gl/wpnvVx
- Bertol E, Trignano C, Di Milia MG, Di Padua M, Mari F (2008) Cocaine-related deaths: An enigma still under investigation. Forensic Sci Int 176: 121-23. Link: https://goo.gl/FP4tfl
- Carvalho VM, Fukushima AR, Fontes LR, Fuzinato DV, Florio JC, et al. (2013) Cocaine postmortem distribution in three brain structures: A comparison with whole blood and vitreous humour. J Forensic Leg Med 3:143-145. Link: https://goo.gl/jyw9Gq
- Palmieri C, Bardy D, Letovanec I, Mangin P, Augsburger M, et al. (2013) Biochemical markers of fatal hypothermia. Forensic Sci Int 226: 54-61. Link: https://goo.gl/2bmBqI
- Osuna E, Vivero G, Conejero J, Abenza JM, Martínez P, et al. (2005) Postmortem vitreous humor b-hydroxybutyrate: its utility for the postmortem interpretation of diabetes mellitus. Forensic Sci Int 153: 189-195. Link: https://goo.gl/hWoq9O
- Wilke N, Janssen H, Fahrenhorst C, Hecker H, Manns MP, et al. (2007) Postmortem determination of concentrations of stress hormones in various body fluids – is there a dependency between adrenaline/noradrenaline quotient, cause of death and agony time? Int J Legal Med 121: 385-394. Link: https://goo.gl/wI25Bb
- Zhu BL, Ishikawa T, Michiue T, Li DR, Zhao D, et al. (2007) Post mortem serum catecholamine levels in relation to the cause of death. Forensic Sci Int 173: 122-129. Link: https://goo.gl/YCw5eI
- Helander A, Beck O, Jones AW (1992) Urinary 5HTOL/5HIAA as biochemical marker of postmortem ethanol synthesis. Lancet 340: 1159. Link: https://goo.gl/GGuxmY
- Helander A, Beck O, Jones AW (1995) Distinguishing ingested ethanol from microbial formation by analysis of urinary 5-hydroxytryptophol and 5-hydroxyindolacetic acid. J Forensic Sci 40: 95-98. Link: https://goo.gl/hZjyFB
- Johnson RD, Lewis RJ, Canfield DV, Dubowsky KM, Blank CL (2005) Utilizing the urinary 5-HTOL/5-HIAA ratio to determine ethanol origin in civil aviation accident victims. J Forensic Sci 50: 670-675. Link: https://goo.gl/LGEfbb
- Little KY , McLaughlin DP , Zhang L , McFinton PR , Dalack GW , et al. (1998) Brain dopamine transporter messenger RNA and binding sites in cocaine users: a postmortem study. Arch Gen Psychiatry 55: 793-739. Link: https://goo.gl/RUlcX8
- Mash DC, Duque L, Pablo J, Qin Y, Adi N, et al. (2009) Brain biomarkers for identifying excited delirium as a cause of sudden death. Forensic Sci Int 190: e13-e19. Link: https://goo.gl/CrSVGn
- Laterra J, Keep R, Betz LA, Goldstein GW (1999) Blood-Brain Barrier in Siegel GJ, Agranoff BW, Albers W, Ficher SK, Uhler MD. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6th. Philadelphia: Lippincott-Raven. Link: https://goo.gl/FcR4SX
- Felicio LF, Florio JC, Sider LH, Cruz-Casallaz PE, Bridgest RS (1996) Reproductive Experience Increases Striatal and Hypothalamic Dopamine Levels in Pregnant Rats. Brain Res Bull 40: 253-256. Link: https://goo.gl/gwkNRM
- Maeda H, Zhu, BL, Ishikawa T, Quan L, Michiue T (2009) Significance of postmortem biochemistry in determining the cause of death. Leg Med 11: S46-49. Link: https://goo.gl/p3L7nX
- Palmieri C, Lesta MM, Sabatasso S, Mangin P, Augsburger M, et al. (2012) Usefulness of postmortem biochemistry in forensic pathology: Illustrative case reports. Leg Med 14: 27-35. Link: https://goo.gl/06Z4Am
- Maruyama Y, Oshima T, Nakajima E (1980) III. Simultaneous determination of catecholamines in rat brain by reversed-phase liquid chromatography with electrochemical detection. Life Sci 24: 1115-1120. Link: https://goo.gl/xifPTl
- Kish SJ, Kalasinsky KS, Derkach P, Schmunk GA, Guttman M, et al. (2001) Striatal Dopaminergic and Serotonergic Markers in Human Heroin Users. Neuropsychopharmacology 24: 561-567. Link: https://goo.gl/CEQ6Ed
- Dackins CA, Gold MS (1985) New Concepts in Cocaine Addiction: The Dopamine Depletion Hyphotesis. Neurosci Biobehav Rev 9: 469-467. Link: https://goo.gl/9pJHQR
- Beveridge TJR, Smith HR, Nader MA, Porrino LJ (2009) Abstinence from Chronic Cocaine Self-Administration Alters Striatal Dopamine Systems in Rhesus Monkeys. Neuropsychopharmacology 34: 1162-1171. Link: https://goo.gl/l0kT0l
- Volkow ND, Gur RC, Wang GJ, Fowler JS, Moberg PJ, et al. (1998) Association between Decline in Brain Dopamine Activity with Age and Cognitive and Motor Impairment in Healthy Individuals. Am J Psychiatry 155: 344-349. Link: https://goo.gl/oGRlKG
- Eidelberg D, Takikawa S, Dhawan V, Chaly T, Robeson W, et al. (1993) Striatal 18F-DOPA Uptake: Absence of an aging Effect. J Cereb Blood Flow Metab 13: 881-888. Link: https://goo.gl/jd7iKS
- Cone EJ (1998) Recent discoveries in pharmacokinetics of drugs of abuse. Toxicol Letters 102: 97-101. Link: https://goo.gl/Qvx3KE
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
