Archives of Pulmonology and Respiratory Care
Post-inflammatory pulmonary fibrosis in a discharged COVID-19 patient: Effectively treated with Pirfenidone
Zhan Xi1*, Zhao Zhigang2 and Li Ting3
2Orthopaedic Department, The Fourth Hospital of Wuhan, Wuhan, China
3Orthopaedic Department, Hubei Provincial Hospital of Integrated Chinese and Western Medicine, Wuhan, China
Cite this as
Xi Z, Zhigang Z, Ting L (2020) Post-inflammatory pulmonary fibrosis in a discharged COVID-19 patient: Effectively treated with Pirfenidone . Arch Pulmonol Respir Care 6(1): 051-053. DOI: 10.17352/aprc.000053Introduction
The coronavirus disease 2019 (COVID-19) caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV2) has led to a pandemic situation. According to the WHO classification, COVID-19 patients could be classified with severity as uncomplicated illness, mild pneumonia, severe pneumonia, acute respiratory distress syndrome (ARDS), sepsis, and septic shock [1]. China has controlled the situation and thousands of patients had been discharged. We noticed that most patients of severe pneumonia or ARDS had post-inflammatory pulmonary fibrosis (PPF) during follow-up after the cure of COVID-19. Here we report a case of PPF resulting from COVID-19 effectively treated with Pirfenidone, an antifibrosis medication.
Case report
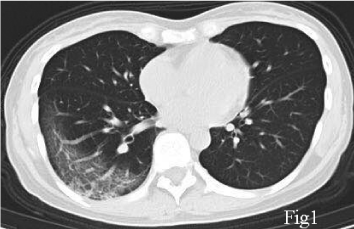
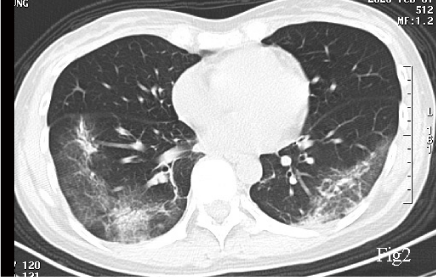
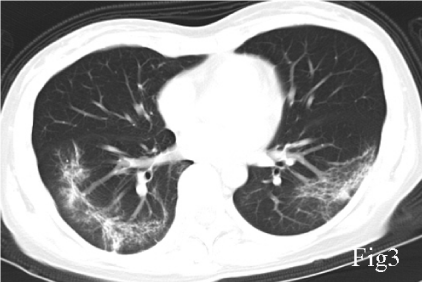
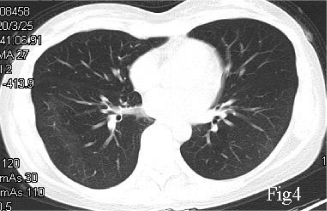
A 40-year-old female, with a chief complaint of febrile (body temperature 38℃) for 5 days, was diagnosed COVID-19 with a positive nucleic acid assay of SARS-CoV2 of her nasopharyngeal swab. Her first CT (Jan 25, 2020), which performed 7 days after symptoms onset, showed a right lower lobe pneumonia, presenting as Ground Glass Opacity (GGO) and reticulation (Figure 1). She was prescribed acetaminophen yet the fever was not controlled and she started to have dyspnea 1 week later, when the second CT scan (Feb 1, 2020) showed that the pulmonary lesion had progressed into a bilateral involvement, with GGO, reticulation, patchy consolidation and subpleural sparing, in accordance with an Organizing Pneumonia (OP) mixed with Nonspecific Interstitial Pneumonia (NSIP) pattern (Figure 2). She was admitted in the Hubei Provincial Hospital of Integrated Chinese and Western Medicine, as a result of the deterioration. Physical examination: T 38.5℃, Bp 110/80mmHg, Respiratory Rate 20/min, Heart Rate 110/min. Laboratory results on hospital day1 reflected leukopenia and lymphopenia, mild thrombocytopenia (White Blood Cell count: 2.7×109/L, Lymphocytes 0.73×109/L, Platelet 100×109/L), and normal levels of creatine and liver enzymes. The oxygen saturation was 88-90% with room air. She was given lopinavir/ritonavir (400 mg and 100 mg, respectively) twice a day for 10 days, intravenous immunoglobin 20g qd for 5 days, and methylprednisolone 40mg qd for 3 days, intravenous Moxifloxacin 0.4 qd for 7 days, and oxygen therapy. With treatment, her body temperature became normal and the oxygen saturation was improved to 93% at room air, and the blood routine showed normal white cell count, the re-test of nucleic assay of nasopharyngeal swab for SARS-CoV2 turned negative, for 2 consecutive times with an interval of 48 hours. However, she still complained about shortness of breath. She was discharged on Feb 14 (hospital day14), and the CT scan on the same day showed minor improvement compared with the second CT scan, still in an OP mixed with NSIP pattern (Figure 3), with obvious reticulation. She was prescribed Pirfenidone 600mg Tid, and well tolerated. The symptoms of dyspnea improved gradually and the saturation was 99% at room air. 6 weeks later, the follow-up CT scan on March 25 showed a substantial improvement (Figure 4). Pirfenidone was ceased.
Discussion
Post-inflammatory pulmonary fibrosis (PPF, ICD-9-CM 515) has been categorized as a general diagnostic code used by providers for parenchymal fibrosis [2], which was not rare after viral pneumonia and caused continuous pulmonary function loss, reported in Middle East Respiratory Syndrome (MERS) patients [3]. GGO and consolidation were the major form of pulmonary involvement in COVID-19 patients [4,5]. With the control of COVID-19 situation in China, we are having thousands of patients discharged, and interstitial abnormalities on CT scan such as reticulation and NSIP pattern could be observed in some cured patients during follow-up, but was not reported yet. As an antifibrotic medication, Pirfenidone was approved for the treatment for Idiopathic Pulmonary Fibrosis (IPF) in China since December 2013. The phase 2 trial of uILD study had confirmed that patients with progressive fibrosing unclassifiable interstitial lung disease could benefit from 24 weeks of pirfenidone treatment as well, the predicted mean change in forced vital capacity (FVC) was significantly lower in patients given pirfenidone than placebo, with an acceptable safety and tolerability profile [6]. Pirfenidone had never been reported to effectively treat PPF, especially in such an unprecedented disease COVID-2019.
Despite the use of short term steroids, the patient still complained about shortness of breath, only minor improvement of interstitial abnormalities on the follow-up CT scan was observed, the reticulation of which suggesting a post-inflammatory pulmonary fibrosis. Due to the side effects of a longer term use of steroids, Pirfenidone was prescribed trying to avoid the progress into irreversible pulmonary fibrosis. The fourth CT scan showed absorption of the interstitial abnormalities including reticulation, GGO and patchy consolidation, indicating the potential use of Pirfenidone as an effective medication for the post-inflammatory pulmonary fibrosis of COVID-19 patients.
- WHO (2020) Coronavirus disease (COVID-19) Clinical management of COVID-19. outbreak. Link: https://bit.ly/2B0vyCl
- Vu A, Vasireddy A, Moua T, Baqir M, Ryu JH (2019) Clarifying the Diagnosis of Post-Inflammatory Pulmonary Fibrosis: A Population-based Study. Eur Respir J 54: 1900103. Link: https://bit.ly/37UeiL5
- Das KM, Lee EY, Singh R, Enani MA, Al Dossari K, et al. (2017) Follow-up chest radiographic findings in patients with MERS-CoV after recovery. Indian J Radiol Imaging 27: 342-349. Link: https://bit.ly/2CuTf6k
- Bernheim A, Mei X, Huang M, Yang Y, Fayad ZA, et al. (2020) Chest CT Findings in Coronavirus Disease-19 (COVID-19): Relationship to Duration of Infection. Radiology 295: 200463. Link: https://bit.ly/31bAcbg
- Shi H, Han X, Jiang N, Cao Y, Alwalid O, et al. (2020) Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis 20: 425-434. Link: https://bit.ly/2YmJlMB
- Maher TM, Corte TJ, Fischer A, Kreuter M, Lederer DJ, et al. (2020) Pirfenidone in patients with unclassifiable progressive fibrosing interstitial lung disease: a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet Respir Med 8: 147-157. Link: https://bit.ly/3fQpknh
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
