Annals of Pancreatic Disorders and Treatment
Successful Conservative Management of Pancreatico-Colonic Fistula
Hazem Ben Ameur1*, Haitham Rejab1, Wiem Feki2, Moez Sahnoun1, Rafik Mzali1 and Zeineb Mnif2
2Department of Radiology, Hedi Chaker’s Teaching Hospital, 3029 Sfax, Tunisia
Cite this as
Ben Ameur H, Rejab H, Feki W, Sahnoun M, Mzali R, et al (2017) Successful Conservative Management of Pancreatico-Colonic Fistula. Ann Pancreat Disord Treatm 1(1): 001-003. DOI: 10.17352/apdt.000001Background: Internal fistula with adjacent viscera during acute pancreatitis is a rare complication. Due to their anatomic localization, colon, duodenum or stomach may be involved. Pancreatico-colonic fistula is associated with a higher septic risk and surgery is usually mandatory.
Results and main findings: We report a new case in 71-year-old man diagnosed by CT with rectal water soluble contrast media and managed conservatively. To our knowledge, this is the second case in the literature treated in this way.
Conclusion: Conservative treatment may be offered to selected patients who have a rapid improvement of symptoms associated with a significant regression of the collection.
Abbreviations
CRP: C-Reactive Protein; CT: Computed Tomography; ERCP: Endoscopic Retrograde Cholangio-Pancreatography
Introduction
Internal fistula with adjacent viscera during acute pancreatitis is a rare complication. Due to their anatomic localization, colon, duodenum or stomach may be involved [1]. Pancreatico-colonic fistula is associated with a higher septic risk and surgery is usually mandatory [1,2]. Through a new case and a brief review of the literature, we purpose to discuss the diagnostic and therapeutic modalities of this particular entity.
Results
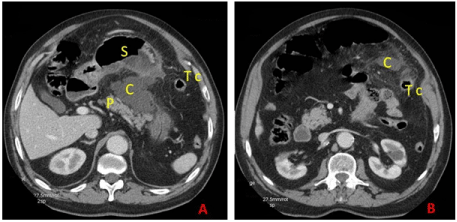
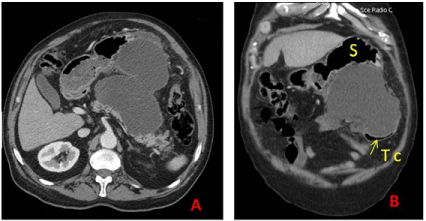
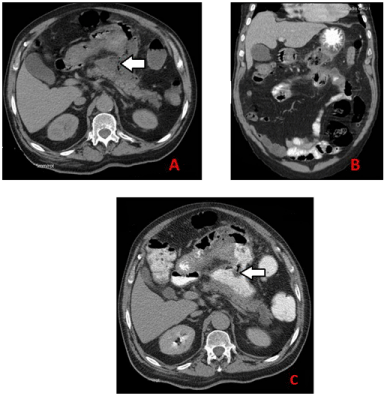
A 71-year-old man, with a previous history of alcohol abuse, arterial hypertension, asthma and arrhythmia, was admitted to our department for acute abdominal pain with vomiting. Physical examination revealed an epigastric mass. Biological parameters showed serum lipase level at 2430 IU/ml, a white blood cell count at 20100 /mm3, a blood glucose level at 8.37 mmol/l and C-reactive protein (CRP) rate at 287 mg/l. The diagnosis of acute pancreatitis was then retained. Abdominal CT grade was judged to be grade E by showing two fluid collections: the greatest one measuring 80 x 40 mm was located in the lesser sac (Figure 1A) and the other was located in the tail of the pancreas (Figure 1B). Conservative management including i.v. fluids and analgesics was carried out. During the follow-up, the patient experienced persistent symptoms, increasing of the epigastric mass size and onset of fever at 38.5°C. A second CT performed on the 21th day showed an increase in the size of the lesser sac collection reaching 170 x 120 mm without any sign of infection (Figure 2A). This collection was in close contact with the stomach and the transverse colon exerting a mass effect (Figure 2B). A percutaneous drainage was scheduled within 48 hours (the interventional radiologist was not available before this date). During this interval, the patient developed diarrhea of necrotic materials with anal irritation and concomitantly, a significant regression of the size of the epigastric mass was observed. At biology, there was ascension of CRP to 411 mg/l and an accelerated erythrocyte sedimentation rate at 150. CT performed at 48th hour showed a significant reduction in the size of the fluid collection with presence of gas (Figure 3A). A pancreatico-enteric fistula was strongly suspected. The use of oral contrast media did not demonstrate any fistulous tract (Figure 3B). However, rectally administrated water soluble contrast media revealed a large fistula between the collection and the transverse colon (Figure 3C). Subsequently, the patient improved rapidly with antibiotic therapy (apyrexia and resolution of the diarrhea) and he well tolerated a low fat diet. We then decided to continue conservative management. The patient was discharged on 45th day. He was seen regularly and continued to progress well. A repeat CT performed 3 months later showed complete resolution of the collection.
Discussion
Colonic complications during acute pancreatitis are rare and account for 1 to 3.3% [3]. They include a localized ileus with pseudo obstruction, obstruction, necrosis, hemorrhage, ischemic colitis and fistula [3]. Incidence of pancreatico-colonic fistula increases with the severity of pancreatic inflammation [2]. It seems to be comprised between 3 and 10% of severe acute pancreatitis [4-7]. The interval between the onset of acute pancreatitis and appearance of pancreatico-colonic fistula is ranging from 10 to 90 days [8]. In fact, the fistula is more common in presence of a well-defined collection or pseuodocyst, like in our patient, than in poorly organized pancreatic necrosis [9].
Pancreatico-colonic fistula should be suspected in lower gastrointestinal bleeding (which results from erosion of vessels in the bowel wall), sepsis, or diarrhea especially of necrotic or purulent materiel, as in our patient [1,3,10-12].
Pancreatico-colonic fistula may be objectified by injection of contrast media into collection or pseudocyst during endoscopic or percutaneous drainage of them (cystography) [2,9]. ERCP is also useful to diagnosis but only if there is ductal disruption with communication between pancreatic necrosis or collection and main pancreatic duct [13]. On CT scan, an edematous and indistinct large bowel wall and the presence of air in the pancreatic bed is very suggestive for pancreatico-colonic fistula [1]. Confirmation is then obtained by using rectal water soluble contrast media which allow to directly showing the fistula tract, like in our case [11,14]. This examination represents a simple, inexpensive, and efficient modality in comparison with previous ones. Colonoscopy may also identify pancreatico-colonic fistula, but it has been reported by only a few authors and cautions could be taken regard of risk of bowel perforation [1,15].
Surgery remains the treatment of choice of pancreatico-colonic fistulas regarding the risks of colonic perforation, spontaneous or persistent infection, or overwhelming sepsis and massive hemorrhage [1,2,9,10,12,16]. Surgical procedures should be selected according to patient condition and the degree of pancreatic inflammation. For the treatment of colonic fistula, the best surgical procedure is the resection of the responsible segment of the colon [2]. If patient is unfit, diverting colostomy or ileostomy is recommended [2 9]. The treatment of pancreatic necrosis consists of debridement and wide drainage while that of pseudocyst is internal or more often external drainage [9]. Postoperative mortality is high ranging from 17% to 67% [6,17]. So, alternative treatments have been reported which may be endoscopic or percutaneous. Various endoscopic procedures were described. Drainage of pseudocyst either transmurally (endoscopic cystogastrostomy or cystoduodenostomy) or transpapillary (if there is connection with main pancreatic duct) permits rapid closure of the colonic fistula, thus avoiding the need for surgical intervention [9,13,16]. Transpapillary stent insertion in the ductal disruption provides disconnection of pseudocyst [9]. Hwang et al., have reported closing the fistula hole endoscopically using hemoclips and biological glue with good outcome [15]. Sezer et al., reported percutaneous drainage of pancreatic abcess which was fistulized in the ascending colon [18]. Nevertheless, all these techniques are feasible for only well-defined pseudocyst or collection and by an experienced operator. In addition, they require further evaluation before proposing them as a standard.
Unlike fistula to the upper gastrointestinal tract which may be benefit by providing spontaneous drainage of pancreatic collection or pseudocyst, pancreatico-colonic fistula has a higher infectiouvs risk and conservative treatment is theoretically inconceivable [1,19]. However, this was previously reported by Green et al. [10]. Our case is the second one in the literature. We advocated conservative treatment because symptoms were spectacularly improved and an almost complete spontaneous drainage of pancreatic collection into colon was observed.
Conclusion
The diagnosis of pancreatico-colonic fistula complicating acute pancreatitis could be made by means of cystography, ERCP or CT with rectal contrast media. Conservative treatment may be offered to selected patients who have a rapid improvement of symptoms associated with a significant regression of the collection or pseudocyst. In the other cases, surgery or eventually endoscopic or percutaneous treatment is required.
- Kochlar R, Jain K , Gupta V , Singhal M , Kochhar S , et al. (2012) Fistulization in the GI tract in acute pancreatitis. Gastrointest Endosc 75: 436-440. Link: https://goo.gl/rqnzRm
- Suzuki A , Suzuki S , Sakaguchi T , Oishi K , Fukumoto K , et al. (2008) Colonic Fistula Associated with Severe Acute Pancreatitis: Report of Two Cases. Surg Today 38: 178–183. Link: https://goo.gl/MdFvuo
- Adams DB, Davis BR, Anderson MC (1994) Colonic complications of pancreatitis. Am Surg 60: 44-49. Link: https://goo.gl/Ue7XPg
- Kriwanek S, Armbruster C, Beckerhinn P, Dittrich K, Redl E (1996) Improved result after aggressive treatment of colonic involvement in necrotzing pancreatitis. Hepatogastroenterology 43: 1627–1632. Link: https://goo.gl/cNcvZl
- Ho HS, Frey CF (1995) Gastrointestinal and pancreatic complications associated with severe pancreatitis. Arch Surg 130: 817–823. Link: https://goo.gl/CSVy99
- Bouillot JL, Alexandre JH, Vuong NP (1989) Colonic involvement in acute necrotizing pancreatitis: result of surgical treatment. World J Surg 13: 84–87. Link: https://goo.gl/ltUpSn
- Van Minnen LP, Besselink MGH, Bosscha K, Van Leeuwen MS, Schiper MEI, et al. (2004) Colonic involvement in acute pancreatitis: a retrospective study of 16 patients. Dig Surg 21: 33–38. Link: https://goo.gl/zKGVzT
- Negro P, D’Amore L, Flati G, Gossetti F, Guadagni S, et al. (1991) Colonic involvement in pancreatitis: six cases more. Int Surg 76: 122–126. Link: https://goo.gl/RpvS3o
- Howell DA, Dy RM, Gerstein WH, Hanson BL, Biber BP (2000) Infected Pancreatic Pseudocysts With Colonic Fistula Formation Successfully Managed by Endoscopic Drainage Alone: Report of Two Cases. Am J Gastroenterol 95: 1821–1823. Link: https://goo.gl/eNo1Ew
- Green BT, Mitchell RM, Branch MS (2003) Spontaneous Resolution of a Pancreatic–Colonic Fistula after Acute Pancreatitis. Am J Gastroenterol 98: 2809-2810. Link: https://goo.gl/99EJfx
- Tüney D, Altun E, Barlas A, Yegen C (2008) Pancreatico-Colonic Fistula after Acute Necrotizing Pancreatitis. Diagnosis with Spiral CT Using Rectal Water Soluble Contrast Media. JOP 9: 26-29. Link: https://goo.gl/i7E34M
- Santos JC Jr , Feres O, Rocha JJ, Aracava MM (1992) Massive lower gastrointestinal hemorrhage caused by pseudocyst of the pancreas ruptured into the colon. Report of two cases. Dis Colon Rectum 35: 75–77. https://goo.gl/EKJKgJ
- Fujii K , Suzuki K , Goto A , Nakahata K , Matsunaga Y , et al. (2011) Pancreatic-colonic fistula successfully treated with endoscopic transpapillary nasopancreatic drainage. Endoscopy 43: E154-155. Link: https://goo.gl/VrL7Rt
- De Backer AI, Mortelé KJ, Vaneerdeweg W, Ros PR (2001) Pancreatocolonic fistula due to severe acute pancreatitis: imaging findings. JBR-BTR 84: 45-47. Link: https://goo.gl/tGZlDh
- Hwang SO, Lee TH, Park JW, Park SH , Kim SJ (2010) Endoscopic management of multiple colonic fistulae secondary to acute pancreatitis. Gastrointest Endosc 71: 395-397. Link: https://goo.gl/JHgwIf
- Urakami A, Tsunoda T, Hayashi J, Oka Y , Mizuno M (2002) Spontaneous fistulisation of a pancreatic pseudocyst into the colon and duodenum. Gastrointest Endosc 55: 949–951. Link: https://goo.gl/JLF2NB
- Alexander ES, Clark RA, Federle MP (1982) Pancreatic gas: indication of pancreatic fistula. AJR Am J Roentgenol 139: 1089–1093. Link:
- Sezer A , Sagiroglu T , Temizoz O , Yagci MA , Umit GE , et al. (2011) Spontaneous fistulization of a pancreatic abscess to colon and duodenum treated with percutaneous drainage. Surg Laparosc Endosc Percutan Tech 21: e138-140. Link: https://goo.gl/tua2Dh
- Levy I, Ariche A (1999) Complete recovery after spontaneous drainage of pancreatic abscess into the stomach. Scand J Gastroenterol 34: 939-941. Link: https://goo.gl/qgj6oK
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
