Archives of Otolaryngology and Rhinology
Efficacy of olfactory training program in conjunction with nasal steroid irrigation in treating persistent smell loss in COVID-19 patients
Marwa Abdelhafeez1* and Safaa Mostafa Ali Elkholi2
2Department of Rehabilitation Sciences, College of Health and Rehabilitation Sciences Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia
Cite this as
Abdelhafeez M, Ali Elkholi SM (2022) Efficacy of olfactory training program in conjunction with nasal steroid irrigation in treating persistent smell loss in COVID-19 patients. Arch Otolaryngol Rhinol 8(1): 001-006. DOI: 10.17352/2455-1759.000144Copyright
© 2022 Abdelhafeez M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Background: Smell loss is a challenging clinical problem. There is still no evidence-based medical intervention to treat patients with persistent smell loss as a result of Coronavirus Disease 2019 (COVID-19). The aim is to analyze the efficacy of treating patients with persistent smell loss due to COVID-19 using olfactory training (OT) combined with nasal steroid irrigation. A cross-sectional study enrolled 196 COVID-19 patients with persistent smell loss were recruited. All patients underwent two testing sessions. At the first session, patients were examined by a history taking followed by an Otorhinolaryngological examination including, a thorough nasal endoscopy. In addition, an olfactory performance assessment was performed to assess the severity of smell loss. All patients were then treated with OT and nasal steroid irrigation with budesonide for 12 weeks. The second session includes an olfactory performance assessment to assess if there is an improvement in olfactory function.
Methods: A cross-sectional study enrolled 196 COVID-19 patients with persistent smell loss were recruited. All patients underwent two testing sessions. At the first session, patients were examined by a history taking followed by an Otorhinolaryngological examination including, a thorough nasal endoscopy. In addition, an olfactory performance assessment was performed to assess the severity of smell loss. All patients were then treated with OT and nasal steroid irrigation with budesonide for 12 weeks. The second session includes an olfactory performance assessment to assess if there is an improvement in olfactory function.
Results: Patients after treatment showed improvement in overall Threshold-Detection-Identification (TDI) score, odor detection threshold score, odor discrimination score, and odor identification score, which were higher than before treatment. Statistically significant differences before and after treatment results were very pronounced for the overall TDI score (p = 0.014) and odor detection threshold score (p = 0.003). There was no significant difference in odor discrimination (p = 0.08) or odor identification (p = 0.126) scores before and after treatment.
Conclusion: The results of this study support the efficacy of the combination of budesonide nasal irrigation and OT in treating persistent smell loss due to COVID-19 infection.
Introduction
Evolutionarily speaking, olfaction is the most important chemosensory system in the living world [1]. Post-Viral Olfactory Dysfunction (PVOD) is thought to result from a conductive dysfunction caused by mucosal edema, as well as a neurosensory dysfunction due to degeneration of the olfactory epithelium [2]. PVOD has become especially relevant with the onset of the Coronavirus Disease 2019 (COVID-19) pandemic, in which a viral involvement of olfactory neuroepithelium has led worldwide to an unprecedented number of occurrences of smell loss [3,4].
People suffering from COVID-19 with loss of odor have a significant deterioration in their quality of life. Their loss of smell mainly disrupts each day's activities linked to olfactory functions [5]. Studies have shown that the predominance of loss of smell in COVID-19 patients ranges from 59% to 86% [6,7]. Loss of smell debilitates individuals for a diversity of reasons, inclusive of interference with the recognition of circumferential hazards (smoke, toxins, or gas leaks). It can disturb feeding conduct and appetite because of changes in taste and distress of not recognizing rotten food [8].
Loss of smell due to COVID-19 can be more common and severe than with other viral upper respiratory infections. Huart et al found that olfactory function is more affected in patients with COVID-19 than in patients with a common cold. Although many patients regain their sense of smell after a few weeks, a definite proportion of them seem to develop a long-lasting olfactory dysfunction [9].
Loss of smell is a challenging clinical problem with few proven therapeutic options [10]. There is still no evidence-based medical intervention to treat patients with persistent loss of smell as a result of COVID-19; nonetheless, Olfactory Training (OT) is still recommended [11]. With OT, individuals practice repeated sniffing of particular scents for 12 to 56 weeks. The hypothesis for this is that frequent sniffing of odors can stimulate olfactory neurons to regenerate and/or reconstruct synaptic pathways to the cortical area for olfaction. Several studies have confirmed that OT can enhance olfactory sensitivity and function [10,12]. In a large multicenter study by Damm. et al. [13], OT has been shown to be effective, particularly in patients with PVOD. Nguyen, et al. [14]. Demonstrated that when subclinical inflammation is a considerable contributor in persistent smell loss, the addition of topical nasal steroid that are capable of reaching the olfactory cleft can increase the effectiveness of OT. Regarding mechanisms of action, it was postulated that corticosteroids enhance olfactory function and regulate the olfactory receptor neurons’ function through their anti-inflammatory activity (by acting on olfactory Na, K-ATPase) [15]
The present study seeks to investigate the effect of adding budesonide nasal irrigation to OT in patients both with smell loss and without any visible sign of sinonasal inflammation. Therefore, the study aims to evaluate the effectiveness of budesonide nasal irrigation and OT as a treatment for patients with persistent smell loss as a result of COVID-19.
Methodology
Study design & Subjects
A cross-sectional study was carried out in the Otorhinolaryngology department of a university hospital. This study evaluated 196 patients with persistent loss of smell due to COVID-19.
The inclusion criteria established for this research are: Patients with a history of smell loss after COVID-19 confirmed by Sniffin’ Sticks test and those who fulfilled the following criteria were enrolled in the study.
- Patients of both genders aged 18 to 60 years.
- Patients with loss of smell for more than three months due to COVID-19, confirmed by PCR results.
- All patients had no persistent symptoms of COVID-19 infection, except for smell loss.
- Patients with a Threshold-Detection-Identification (TDI) score < 30.75 [16].
- Patients without sinonasal pathology or edema and with a visible and patent olfactory cleft confirmed by nasal endoscopy.
The exclusion criteria are:
- Patients under 18 or over 60 years of age.
- Loss of smell is associated with sinonasal diseases such as sinusitis, allergic rhinitis, or sinonasal tumors.
- Patients with parosmia and/or phantosmia.
- History of head trauma.
- History of neurological or psychiatric diseases.
- Patient does not regularly perform OT or nasal steroid irrigation.
Procedure
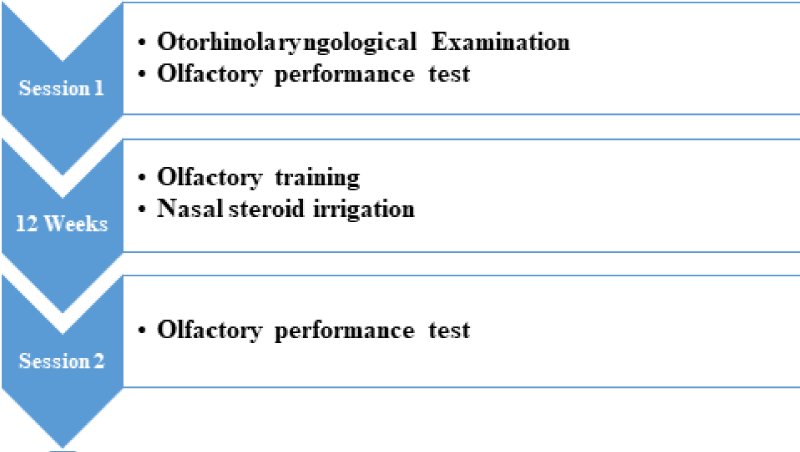
All patients underwent two testing sessions (Figure 1). At the first session, patients were examined by a detailed history taking followed by an Otorhinolaryngological examination. A thorough nasal endoscopy was performed, paying particular attention to the visibility and patency of the olfactory cleft. In addition, an olfactory performance assessment was performed to assess the severity of smell loss.
All patients were then treated with OT and nasal steroid irrigation with budesonide for 12 weeks. In the second session, after completing the OT in conjunction with nasal steroid irrigation, the patients performed an olfactory performance assessment, identical to that of the first testing session, to assess the evolution of olfactory function.
Olfactory performance test
Olfactory function was assessed using the Sniffin’ Sticks battery test (Burghart GmbH, Wedel, Germany). This battery of tests includes three subtests: odor detection threshold; odor discrimination; and odor identification. The Sniffin’ Sticks battery uses pen-like devices to present odors [17,18]. Using commercially available felt-tip pens, the odorants were presented approximately two centimeters in front of both nostrils for two seconds. The patient should not smoke, or swallow anything other than water fifteen minutes before the test. An interval of three to five minutes could be observed between each subtest.
The odor detection threshold for N-butanol is determined using a single-staircase, three-alternative, forced-choice procedure. Three pens were presented to the participant in a random order, two contained odorless solvent (propylene glycol), and the other an odorant in a certain dilution. The patient’s task was to specify the pen with the odorant. The concentration increased if one of the blanks was chosen, and decreased if the correct pen was determined twice in a row. This test must be performed without any visual input: eyes closed or blindfolded. The odor discrimination task consists of 16 triplets of odorants (two pens contain the same odorant; the third pen contains a strange odorant). The task of the patients is to detect the strange pen in a forced-choice procedure. The subject must keep their eyes closed or blindfolded. The odor identification task is composed of 16 common odors presented in a multiple-choice answer format. The patient had to recognize each of the odorants from a list of four descriptors. This test does not require blinded conditions. The results of all three subtests are summed to obtain the overall TDI score.
Olfactory training
OT was carried out at home for 12 weeks [19]. Patients were instructed to take four odors for the OT: (Phenyl Ethyl Alcohol (PEA): rose, eucalyptol: eucalyptus, citronellal: lemon, and eugenol: cloves). Thus, all patients received four brown glass jars (50 ml total volume), filled with 1 ml of the respective odorant (soaked in cotton pads to prevent spilling) and labeled with the odor name. Patients were advised to sniff odors in the morning and evening for about 10 seconds each. In addition, the enrolled patients were advised to keep a diary during the training period to monitor whether the training was carried out regularly. Further, patients received phone calls every three weeks of the study to ensure and encourage continued compliance.
Nasal steroid irrigation
A NeilMedTM (NeilMed, Santa Rosa, CA) squeeze bottle was used. Budesonide respules in a 0.5 mg/2 mL dose were mixed with salt packets, along with 240 ml distilled or filtered water, was used to deliver saline irrigations twice a day for 12 weeks [14].
Statistical analysis
Data were analyzed using the Statistical Package of Social Science (SPSS), software version 26.0 (SPSS Inc., 2019). The normality of data distribution was checked using the Shapiro-Wilk-W test. Paired sample t-test was used to analyze olfactory performance test data before and after treatment. The level of significance was set at 0.05.
Ethical consideration
All ethical considerations were fulfilled before the study. Ethical approval has been requested from the local institutional ethics committee (IRB No. 21-0225). Informed consent was obtained before the start of the study. Thus, the participants were informed about the objectives of the study and the steps in carrying it out, as well as about its risks and benefits. Furthermore, the confidentiality of the data was guaranteed.
Result
Baseline characteristics of the study group
This study included 196 patients who had persistent smell loss after COVID-19. The study sample consisted of 128 (65.3%) females and 68 (34.7%) males. The average age was 35.1 ± 10.3 years.
Measurements of olfactory performance
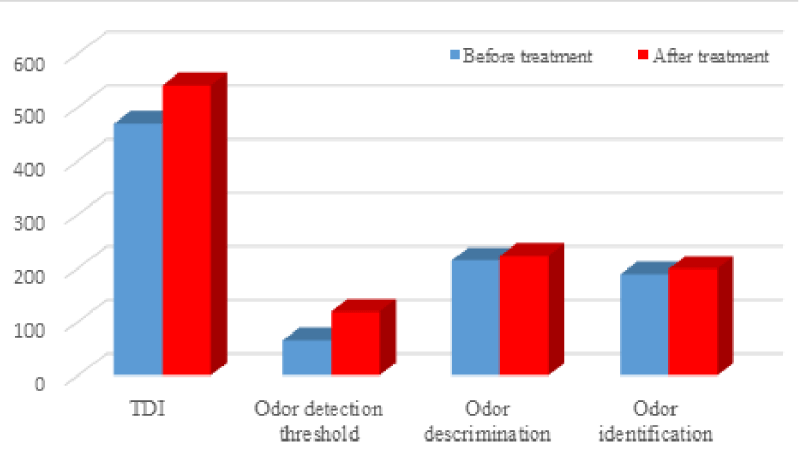
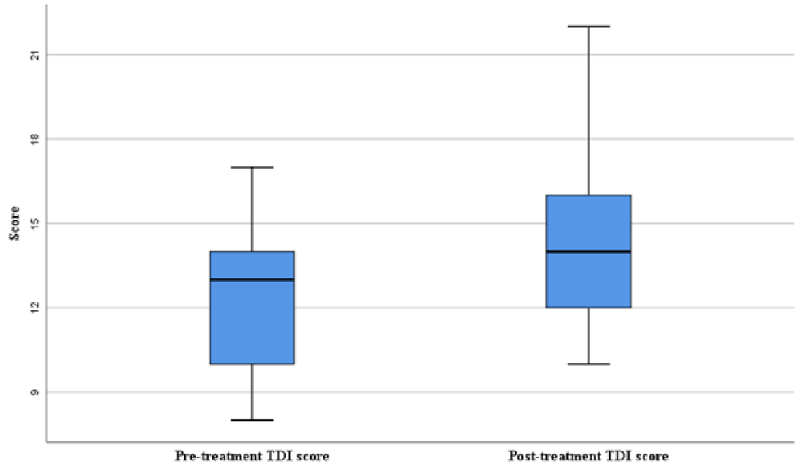
All patients received OT and nasal steroid irrigation for 12 weeks. Patients after treatment showed improvement in overall TDI score (Figures 2,3), odor detection threshold score, odor discrimination score, and odor identification score that were higher than before treatment (Figure 2).
When a paired sampling t-test was used for comparison (Table 1), statistically significant differences before and after treatment outcomes were very pronounced for the overall TDI score (p = 0.014) and odor detection threshold (p = 0.003), there was no significant difference in odor discrimination (p = 0.08) or odor identification (p = 0.126) scores before and after treatment.
Discussion
As the COVID-19 pandemic persists, an increasing number of patients are suffering from loss of smell, which is now a well-known symptom of the disease [11]. Loss of smell significantly impacts the quality of life of COVID-19 patients; therefore, its diagnosis and treatment should be prioritized [8]. Unfortunately, the lack of a clear understanding of the molecular mechanisms of olfactory dysfunction has resulted in limited treatment options [10].
The olfactory system exhibits unique neural plasticity that is not found in other parts of the central nervous system, with neurogenesis in the neuroepithelium and parts of the olfactory tract continuing throughout a person’s life [20]. This suggests that certain interventions could help restore the sense of smell by awakening olfactory neurons or modulating neural function. OT is an intervention that improves olfactory function through frequent sniffing and/or exposure to strong odors [10]. However, the gold standard for treating loss of smell remains unknown. Oral corticosteroids can often improve olfactory function; however, their effect is often short-lived and disappears with the discontinuation of treatment [21]. In addition, the use of corticosteroids is associated with clinically significant side effects and is not suitable for all patients [22]. Therefore, the use of oral corticosteroids is strongly contraindicated by the World Health Organization [11]. However, long-term improvement has been reported with the use of intranasal steroids [21].
The results of the present study suggest that budesonide nasal irrigation enhances the effects of OT by suppressing any asymptomatic inflammation that may cause or exacerbate loss of smell and prevent appropriate neuronal regeneration [14]. To our knowledge, no study in the literature has used budesonide nasal irrigation in combination with OT to treat persistent loss of smell associated with COVID-19.
In the present study, significant improvements were noted in the overall TDI score (p = 0.014) and odor detection threshold score (p = 0.003) after OT in combination with nasal steroid irrigation (Table 1). On the other hand, no significant differences were noted in the odor discrimination score and odor identification score after this treatment (p = 0.08 and p = 0.126, respectively; Table 1). Similarly, Kollndorfer et al [23]. Found that OT led to a significant progress in the odor detection threshold (p = 0.028) and that there was no significant difference in the odor discrimination score (p = 0.916) and odor identification score (p = 0.673). Several previous researches have described debits in odor identification and discrimination abilities and improvements in odor detection threshold [24,25]. Animal studies have revealed that repetitive short-term inhalation of odors could lead to enhanced growth of olfactory receptor neurons and increased expression of olfactory receptors in response to odor inhalation [26,27]. In contrast, in other studies, OT was found to be ineffective in 50% - 85% of study subjects [19,28].
The odor detection threshold is thought to represent a basic olfactory function, while odor identification and odor discrimination abilities represent higher olfactory functions [29]. Hedner, et al. [30]. Examined cognitive factors and their relationship with odor identification and discrimination abilities and the odor detection threshold. They discovered that cognitive factors have a greater influence on odor identification and discrimination abilities. Accordingly, the results of the present study indicated that OT enhances the most basic olfactory function and is likely to influence higher olfactory functions, such as odor identification and discrimination abilities, after long periods of OT.
Recently, Miwa et al and Hura et al reported that OT is the recommended treatment for loss of smell. OT has been associated with minimal deleterious effects and has been found to be most effective in improving olfactory function, especially with early treatment and high treatment compliance. The only inconvenience is the need for providing continuous daily training for several months [2,31]. The positive effect of odor exposure on odor sensitivity may be associated with changes not only at the level of the olfactory epithelium but also at the level of the olfactory bulb or at higher processing levels [32]. Interestingly, previous studies have demonstrated a decrease in the metabolism of the olfactory centers of the brain in patients with PVOD, suggesting that OT induces changes in the functional connection of olfactory, somatosensory and integrative pathways in the brain [23,33].
Nguyen and Patel [14]. Reported that budesonide nasal irrigation in combination with OT was superior to OT lonely in enhancing olfactory function in individuals with loss of smell (p = 0.039). Notably, Fleiner. et al. [34]. Reported additional improvement in olfactory function with a combination of topical corticosteroid spray and OT in comparison with OT alone. The present research varies from the study of Fleiner et al [34]. Since the present study included patients with loss of smell without any signs of chronic sinonasal inflammation. This is important to ensure that any improvement in olfactory function caused by steroid irrigation is not confounded by the treatment of the underlying sinonasal disease.
In the present study, it was hypothesized that although inflammation may not be very obvious in COVID-19 sufferers with smell loss, the loss of smell can be produced by subsidiary microscopical inflammation of the olfactory nerves or epithelium. Previous researches have revealed elevated levels of numerous proinflammatory cytokines or accelerated activity of inflammatory mediators related with smell loss [35]. In addition, a study on patients with hyposmia revealed significantly higher levels of interleukin-6 (IL-6), which is a proinflammatory cytokine, in the nasal discharges of patients than in those of controls [36]. The correct mechanism by which those cytokines can cause olfactory dysfunction is not fully recognized. Though, the combination of an increase activity of inflammatory mediators or their levels proposes that inflammation is a crucial player associated with smell loss [14]. This probably elucidate why the combination of budesonide irrigation with OT enhanced the patient outcomes in the present research.
Recently, Magnetic resonance imaging studies have shown signs of inflammation of the olfactory clefts in COVID-19 patients with smell loss compared with healthy controls, indicating a possible role for anti-inflammatory drugs such as steroids [37]. The idea of corticosteroid treatment for PVPD is not new, but studies with a high level of evidence are insufficient, and the few studies available provide contradictory results. Miwa. et al. [2]. Emphasized that steroid treatment may be effective in post-viral smell loss only in the early stages of the disease. On the other hand, Heilmann et al [38]. Found no significant improvement in olfactory function after the use of a nasal steroid spray. This finding may be due to the fact that the delivery mode of the nasal steroid during irrigation is also likely to contribute to its effectiveness. Distinct from nasal steroid sprays, nasal irrigation can be fully disseminated throughout the whole nasal cavity. A study of the method of nasal irrigation utilizing radioisotopes contrast displayed that both the anterior and the posterior nasal cavity are well irrigated, and the sinuses can also be irrigated [39]. In disparity, nasal steroid sprays are less efficient at involving the whole nasal cavity and are predominantly poorly suited for distribution to the superior or posterior nasal cavity [40]. In individuals with smell loss, nasal irrigation with steroid can extend to the olfactory cleft area to apply its anti-inflammatory effects, which makes it more efficient than a nasal steroid spray [14].
This study has some limitations. First, it lacks a placebo-trained control group to better represent the treatment effects. Second, there are possible differences in patient adherence to olfactory training (OT) and inevitable small differences in how they conduct their OT at home, despite regular follow-up.
At the time of the study design, the recommended training period for OT was 12 weeks [19]. However, a multicenter follow-up study [13]. Found an excellent improvement in olfactory performance after a 32-week training period. This will be of interest for future research examining the long-term effects of OT, which may shed light on the effect of OT on higher olfactory function. In addition, future research may include a placebo-trained control group to investigate the potential effects of OT in conjunction with nasal steroid irrigation.
Conclusion
In the present study, a significant improvement was noted in the overall TDI score and the odor detection threshold score. The present results support the efficacy of a combination of budesonide nasal irrigation and OT in treating persistent smell loss in COVID-19 patients.
Financial disclosure information
The authors have no funding or financial relationships to disclose.
Conflicts of interest
The authors have no conflicts of interest to report.
The authors are grateful for the help of all patients who took part in this work.
- Kalogjera L, Dzepina D (2012) Management of smell dysfunction. Curr Allergy Asthma Rep 12: 154-162. Link: https://bit.ly/3eIKLIH
- Miwa T, Ikeda K, Ishibashi T, Kobayashi M, Kondo K, et al. (2019) Clinical practice guidelines for the management of olfactory dysfunction- secondary publication. Auris Nasus Larynx 46: 653-662. https://bit.ly/3eMWbuN
- Vaira LA, Salzano G, Deiana G, De Riu G (2020) Anosmia and ageusia: common findings in COVID-19 patients. Laryngoscope 130: 1787. Link: https://bit.ly/3sQ9cw3
- Spinato G, Fabbris C, Polesel J, Cazzador D, Borsetto D, et al. (2020) Alterations in smell or taste in mildly symptomatic outpatients with SARS-CoV-2 infection. JAMA 323: 2089-2090. Link: https://bit.ly/3eN35jY
- Elkholi SMA, Abdelwahab MK, Abdelhafeez M (2021) Impact of the smell loss on the quality of life and adopted coping strategies in COVID-19 patients. Eur Arch Otorhinolaryngol 278: 1-8.Link: https://bit.ly/3pQbGZw
- Menni C, Valdes A, Freydin MB, Ganesh S, Moustafa JE, et al. (2020) Loss of smell and taste in combination with other symptoms is a strong predictor of COVID-19 infection. MedRxiv. Link: https://bit.ly/3pNSoUn
- Lechien JR, Chiesa-Estomba CM, De Siati DR, Horoi M, Le Bon SD, et al. (2020) Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study. Eur Arch Otorhinolaryngol 277: 2251-2261. Link: https://bit.ly/3mRXVrc
- Keller A, Malaspina D (2013) Hidden consequences of olfactory dysfunction: a patient report series. BMC Ear Nose Throat Disord 13: 1-20.Link: https://bit.ly/3EK12rb
- Huart C, Philpott C, Konstantinidis I, Altundag A, Trecca EMC, et al. (2020) Comparison of COVID-19 and common cold chemosensory dysfunction. Rhinology. Link: https://bit.ly/3zt3Kk4
- Pekala K, Chandra RK, Turner JH (2016) Efficacy of olfactory training in patients with olfactory loss a systematic review and meta‐analysis. Int Forum Allergy Rhinol 6: 299-307. Link: https://bit.ly/3eN5JWW
- Le Bon SD, Konopnicki D, Pisarski N, Prunier L, Lechien J R, et al. (2021) Efficacy and safety of oral corticosteroids and olfactory training in the management of COVID-19-related loss of smell. Eur Arch Otorhinolaryngol 1-5.Link: https://bit.ly/3HySpBx
- Patel ZM (2017) The evidence for olfactory training in treating patients with olfactory loss. Curr Opin Otolaryngol Head Neck Surg 25: 43-46. Link: https://bit.ly/3sQblb5
- Damm M, Pikart LK, Reimann H, Burkert S, Göktas Ö, et al. (2014) Olfactory training is helpful in postinfectious olfactory loss: a randomized, controlled multicenter study. Laryngoscope 124: 826-831. Link: https://bit.ly/3sSMVOf
- Nguyen TP, Patel ZM (2018) Budesonide irrigation with olfactory training improves outcomes compared with olfactory training alone in patients with olfactory loss. Int Forum Allergy Rhinol 8: 977-981. Link: https://bit.ly/3HmDjPI
- Fong KJ, Kern RC, Foster JD, Zhao JC, Pitovski DZ (1999) Olfactory secretion and sodium, potassium-adenosine triphosphatase: Regulation by corticosteroids. Laryngoscope 109: 383-388. Link: https://bit.ly/3EQiNW7
- Oleszkiewicz A, Schriever VA, Croy I, Hähner A, Hummel T (2019) Updated Sniffin’ Sticks normative data based on an extended sample of 9139 subjects. Eur Arch Otorhinolaryngol 276: 719-728.Link: https://bit.ly/31nRpky
- Hummel C, Zucco GM, Iannilli E, Maboshe W, Landis BN, et al. (2012) standardization of international olfactory tests. Eur Arch Otorhinolaryngol 269: 871-880. Link: https://bit.ly/3FQrbGi
- Kobal G, Klimek L, Wolfensberger M, Gudziol H, Temmel A, et al. (2000) Multicenter investigation of 1,036 subjects using a standardized method for the assessment of olfactory function combining tests of odor identification, odor discrimination and olfactory threshold. Eur Arch Otorhinolaryngol 257: 205-211. Link: https://bit.ly/3mTeeE5
- Hummel T, Rissom K, Reden J, Hähner A, Weidenbecher M, et al. (2009) Effects of olfactory training in patients with olfactory loss. Laryngoscope 119: 496-499. Link: https://bit.ly/32Ys8Ok
- Wilson DA, Best AR, Sullivan RM (2004) Plasticity in the olfactory system lessons for the neurobiology of memory. Neuroscientist 10: 513-524. Link: https://bit.ly/3JCGOTZ
- Blomqvist EH, Lundblad L, Bergstedt H, Stjarne P (2003) Placebo-controlled randomized double-blind study evaluating the efficacy of fluticasone propionate nasal spray for the treatment of patients with hyposmia/ anosmia. Acta Otolaryngol 123: 862-868. Link: https://bit.ly/3qNdkKJ
- Buchman AL (2001) Side effects of corticosteroid therapy. J Clin Gastroenterol 33: 289-294. Link: https://bit.ly/3FV3tsr
- Kollndorfer K, Fischmeister FPS, Kowalczyk K, Hoche E, Mueller CA, et al. (2015) Olfactory training induces changes in regional functional connectivity in patients with long-term smell loss. Neuroimage Clin 9: 401-410. Link: https://bit.ly/3t7T3lV
- Larsson M, Finkel D, Pedersen NL (2000) Odor identification: influences of age, gender, cognition, and personality. J Gerontol B Psychol Sci Soc Sci 55: 304-310. Link: https://bit.ly/34iEOjJ
- Zucco GM , Amodio P, Gatta A (2006) Olfactory deficits in patients affected by minimal hepatic encephalopathy: a pilot study. Chem Senses 31: 273–278. Link: https://bit.ly/3ENq9cU
- Youngentob SL, Kent PF (1995) Enhancement of odorant-induced mucosal activity patterns in rats trained on an odorant. Brain Res 670: 82-88. Link: https://bit.ly/31nRDrU
- Hudson R, Distel H (1998) Induced peripheral sensitivity in the developing vertebrate olfactory system. Ann N Y Acad Sci 855: 109-115. Link: https://bit.ly/3zizI2e
- Patel ZM, Wise SK, DelGaudio JM (2017) Randomized controlled trial demonstrating cost-effective method of olfactory training in clinical practice essential oils at uncontrolled concentration. Laryngoscope Invest Otolaryngol 2: 53-56. Link: https://bit.ly/3ziGy7O
- Kollndorfer K, Kowalczyk K, Hoche E, Mueller CA, Pollak M, et al. (2014) Recovery of olfactory function induces neuroplasticity effects in patients with smell loss. NEURAL PLAST. Link: https://bit.ly/3mR5NJI
- Hedner M, Larsson M, Arnold N, Zucco GM, Hummel T (2010) Cognitive factors in odor detection, odor discrimination, and odor identification tasks. J Clin Exp Neuropsychol 32: 1062-1067. Link: https://bit.ly/3pMLMFH
- Hura N, Xie DX, Choby GW, Schlosser RJ, Orlov CP, et al. (2020) Treatment of post-viral olfactory dysfunction: an evidence-based review with recommendations. Int Forum Allergy Rhinol 10: 1065-1086. Link: https://bit.ly/3Htufsi
- Livermore A, Laing DG (1996) Influence of Training and experience on the perception of multicomponent odor mixtures. J Exp Psychol Hum Percept Perform 22: 267-277. Link: https://bit.ly/3eMuLVW
- Kim YK, Hong SL, Yoon EJ, Kim SE, Kim JW (2012) Central presentation of postviral olfactory loss evaluated by positron emission tomography scan: a pilot study. Am J Rhinol Allergy 26: 204- 208. Link: https://bit.ly/3pN6Tru
- Fleiner F, Lau L, Goktas O (2012) Active olfactory training for the treatment of smelling disorders. Ear Nose Throat J 91: 198-215. Link: https://bit.ly/3mNn46k
- Victores AJ, Chen M, Smith A, Lane AP (2018) Olfactory loss in chronic rhinosinusitis is associated with neuronal activation of c-Jun N-terminal kinase. Int Forum Allergy Rhinol 8: 415-420. Link: https://bit.ly/3JC35RL
- Henkin RI, Schmidt L, Velicu I (2013) Interleukin 6 in hyposmia. JAMA Otolaryngol Head Neck Surg 139: 728-734. Link: https://bit.ly/3sP54wg
- Altundag A, Yıldırım D, Sanli DE, Cayonu M, Kandemirli SG, et al. (2020) Olfactory cleft measurements and COVID-19–related anosmia. Otolaryngol Head Neck Surg. Link: https://bit.ly/32N6vk5
- Eliezer M, Hautefort C (2020) MRI evaluation of the olfactory clefts in patients with SARS-CoV-2 infection revealed an unexpected mechanism for olfactory function loss. Acad Radiol 27: 1191. Link: https://bit.ly/3sP5bba
- Wormald Pj, Cain T, Oates L, Hawke L, Wong I (2004) A comparative study of three methods of nasal irrigation. Laryngoscope 114: 2224-2227. Link: https://bit.ly/3FOJvja
- Bateman ND, Whymark AD, Clifon NJ, Woolford TJ (2002) A study of intranasal distribution of azelastine hydrochlorideaqueous nasal spray with different spray techniques. Clin Otolaryngol Allied Sci 27: 327-330. Link: https://bit.ly/3sQhsMm
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
