Annals of Marine Science
Habitats and Characteristics of Sardina pilchardus, off the Moroccan Atlantic coast
Abdelaziz Mounir1,2*, Hassan Alahyane1,3, Nor-eddine Chouikh4, Nawal Hichami5 and Safaa Bendami1,2
2The Natural History Museum of Marrakech, Research Centre on Biodiversity, Cadi Ayyad University, Bd Allal El Fassi, Marrakech, Morocco
3Laboratory “Aliments, Environnement et santé “, Department of Biology, Faculty of Sciences and Technics, B.P. 549, Cadi Ayyad University, Marrakech 40000, Morocco
4Department of Environmental Engineering, Higher School of Technology Khénifra, B.P. 170, Sultan Moulay Slimane University, Morocco
5Laboratory of Biotechnology & Sustainable Development of Natural Resources. Polydisciplinary Faculty of Beni Mellal, Sultan Moulay Slimane University, Mghila PO Box. 592, Beni Mellal 23000, Morocco
Cite this as
Mounir A, Alahyane H, Chouikh NE, Hichami N, Bendami S (2022) Habitats and Characteristics of Sardina pilchardus, off the Moroccan Atlantic coast. Ann Mar Sci 6(1): 007-020. DOI: 10.17352/ams.000027Copyright License
©2022 Mounir A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.This paper is an overview of the main characteristics of Sardina on the Moroccan Atlantic coast. Sardine occupies an important place in the socio-economic context of Morocco and is the main species exploited on Moroccan coasts. It is the most important species in terms of catches and biomass. The small pelagic stocks in the area are exceptional wealth and as many opportunities in an international context, marked both by the scarcity of the resource and the need to make the sea c contribute more to food security and economic growth. The aim of this work is the study some features of S. pilchardus in the Moroccan waters.
Introduction
Sardine occupies an important place in the socio-economic context of Morocco and is the main species exploited on Moroccan coasts. It is the most important species in terms of catches and biomass. Morocco is the world’s leading producer of sardines and the leader in the international market for canned sardines.
The small pelagic stocks in the area are exceptional wealth and as many opportunities in an international context, marked both by the scarcity of the resource and the need to make the sea contribute more to food security and economic growth.
With a coastline of 3500 km (Mediterranean and Atlantic) and a significant diversity of its marine wealth, Morocco has a real fishing potential of which small pelagic, mainly sardine species, anchovies, mackerel, and horse mackerel, represent in quantitative terms, the main resources exploited and total almost 83% of the catches. And sardine constitutes 63% of these catches. It is thus one of the most exploited pelagic species on the Moroccan coasts.
The biological wealth of Moroccan waters, particularly the Atlantic, is more determined by the favorable nature of the hydro-climatic regime that prevails and the configuration of the continental shelf of Moroccan coasts. These two factors, of paramount importance, will govern the production of the fishing sector of Morocco.
Upwellings are the main source of enrichment in North West African coastal ecosystems and small pelagic fish make up the bulk of biomass. Their dynamics are closely associated with those of environmental factors such as the intensity and seasonal or interannual variability of upwellings, the aspects of primary or secondary production as well as the hydrodynamic characteristics of the environment.
Currently, the management of Moroccan sardine fisheries is based on the assumption of a Mediterranean stock and three separate Atlantic stocks: A first stock north between Cape Spartel and El Jadida. A second, central stock between Safi and Laayoune, and a third, south stock between Boujdor and Lagouira.
In general, in order to better implement a fisheries management strategy, it is important to properly identify management units as a lack of knowledge of population structure complicates and reduces the quality of management plans. It is within this framework that this work, whose objective is to examine the structure of the sardine population and to try to highlight the existing spatial variability in the linear growth of the sardine on the Moroccan coastline.
The biology of the Sardine has long since attracted the attention of naturalists, but it still remains bad known despite the many studies she has been the subject of. At the beginning of the century, in the Atlantic as in the Mediterranean, the sardine has been the subject of many studies we will quote:
For the Atlantic Ocean: [1-10].
For the Mediterranean: works by: [11-23]. These authors have contributed to his knowledge, they have shown the close relationship between hydro-climatic factors and the origin of zooplankton production necessary for sardine nutrition. This is how we know that the color of the seawater and the surface temperature are essential indicators of the abundance of this resource.
In Algeria, several works like those of Bouchereau (1981) in Oran, [24-26].Mouhoub (1986), Brahmi, et al. (1998) in Algiers, and Bedairia (2009 and 2011) in Annaba [27,28]. Treated the biology and the physico-chemical parameters of the marine environment. Their results, although they do not measure the magnitude of the impacts of climate change on the decline of pelagic resources, they justify their interest in local markets, especially sardines whose average price has risen from 50 to 400 dinars per kilogram between 2005 and 2010 [29]. Knowledge of biological parameters is essential for a good analysis of population dynamics and for the management of stocks. To make a contribution to the study of S. pilchardus biology of LEA, our overview has been based are 4 essential axes in the field of fisheries.
Firstly, the biometric axis offers us the possibility of such a study.
Knowing that the measurable characters of the individuals of the same species are susceptible to certain variations and that these variations make it possible to distinguish within the species, races, varieties, or other secondary groups, it will be important to choose the best differential characters, to establish the average type for each of these particular groups which, thus characterized, can be easily identified and followed.
Secondly, data on the age and growth of fish are essential for understanding the life history traits of species and populations (longevity, age at recruitment, age at sexual maturity, breeding periods, migration, mortality) and studying the demographic structure of populations and their populations dynamic, as for example in estimation models based on an age structure. Applications in ecology and paleoecology include the study of the adaptive responses of populations to environmental pressures, of natural origin (climatic variations) or anthropogenic (fishing, pollution, development of the coastal strip). Given the current state of depletion of many aquatic resources, the demand for sclerochemical data reliable resources is increasing to assist decision-making in fisheries management and sustainable use of aquatic resources.
Thirdly, the reproductive study allows sexual determinism, the mode of reproduction (gonochoric, functional, or rudimentary hermaphroditism and parthenogenesis), the type of external fertilization (oviparity) or internal (ovoviviparity or viviparity), the duration of laying and the number of eggs emitted (fertility), to know other parameters necessary for the study of the stock (period reproduction and egg-laying, size of first sexual maturity and size structure according to sex).
The purpose of the study of the reproductive cycle is to characterize the main phase’s evolution of the sex glands, which is established mainly on three types of morphological, weight, and histological criteria.
Finally, the quality and quantity of fish food are among the factors the most important exogenous factors directly affecting growth and, indirectly, maturation, health status (fitness), and fish mortality [30].
Traditionally, data on the qualitative and quantitative composition of food consumed by fish can be used in research fisheries. Indeed, their incorporation into appropriate mathematical models makes it possible to assess in the natural environment the biomass of the prey consumed by the different predators [31].
Presentation of the species ‘Sardina pilchardus Walbaum 1792’
European pilchard is a small coastal pelagic species that lives in dense schools abundant during the day at depths of 30 to 55 m and at night at 15-40 m depths [32]. In the southern zone, sardines are found in waters up to 200 m deep and winter temperatures range from 13.8 to 18.2 °C and 16.7 to 18.7 °C in summer [33]. In addition to diel movements, sardines perform seasonal vertical and horizontal migrations in search of optimal feeding, temperature, and reproduction conditions. Their distribution is conditioned by the temperature of the water. The 10 ° C isotherm marks, more or less, the northern limit of the sardine populations and the isothermal 20 ° C the southern limit [34]. The maximum size of the sardine is 25 cm in the Atlantic and 22 cm in the Mediterranean. The common size is 10 to 20 cm [32].
A. The morphology
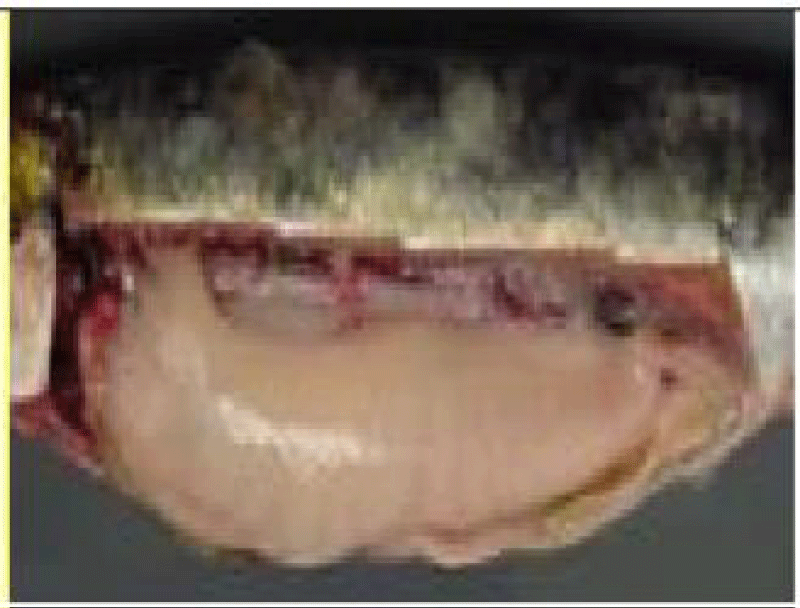
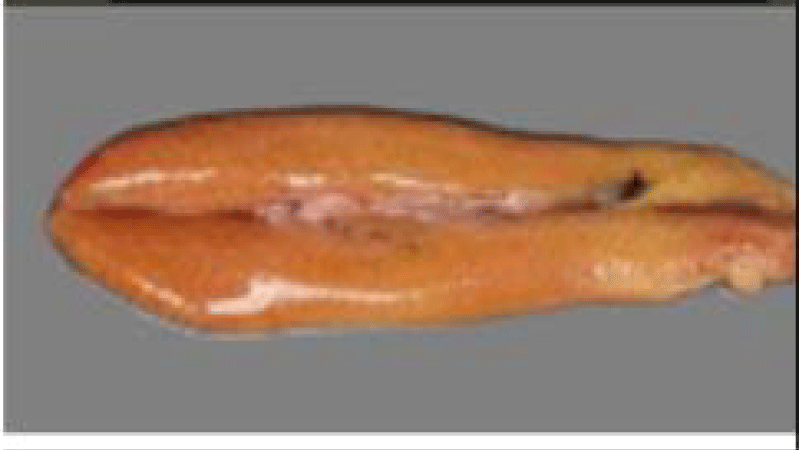
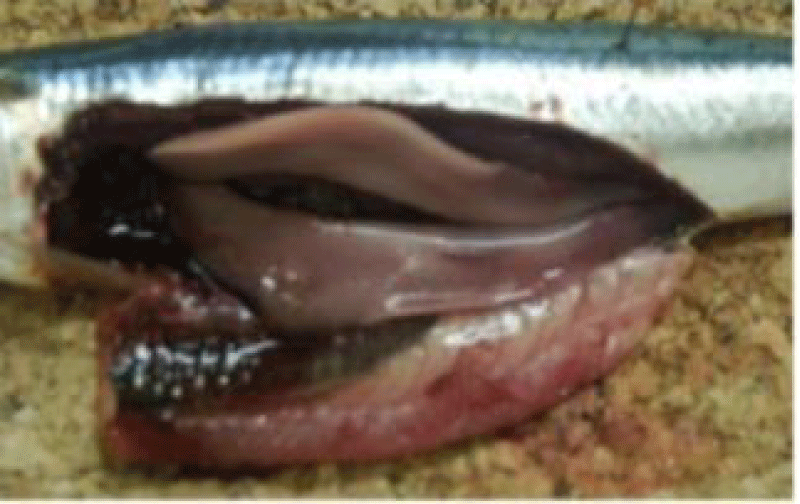
Sardina pilchardus is easily distinguished from other congeners, visible striations adorning the operculum;
vAbout 80 scales, radially fluted lids; Ventral fins located behind the anterior edge of the dorsal fin; Mouth not close to the posterior border of the eyes.
The dorsal fin starts in front of the pelvic and the anal fin ends with two longer rays than the others [35].
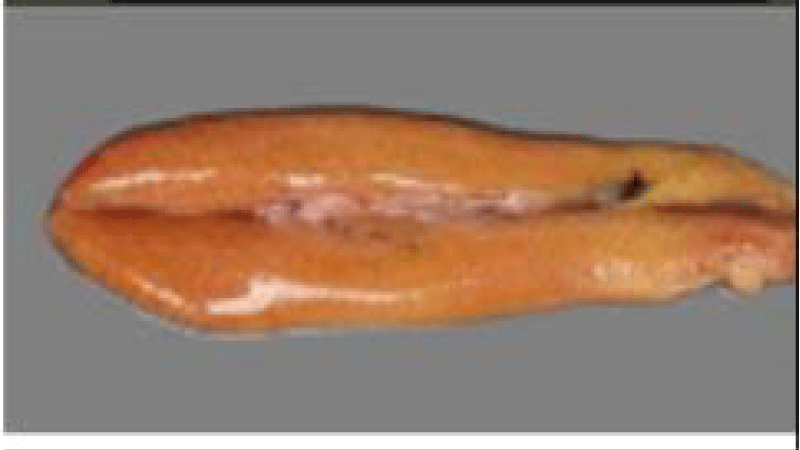
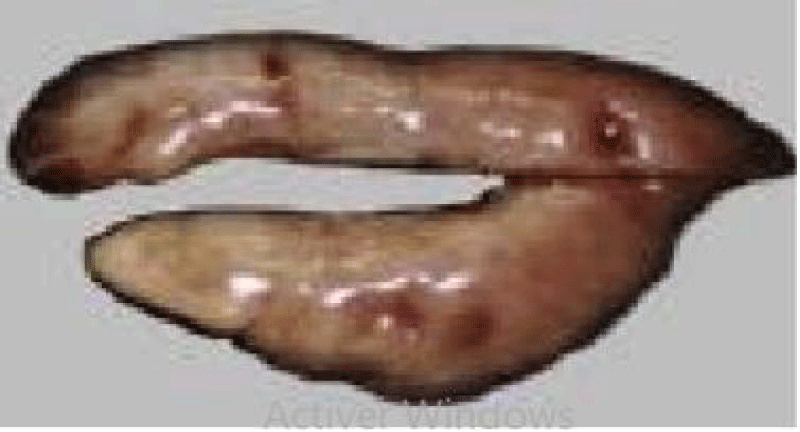
Sardine is an elongated and fusiform-shaped fish of oval section that is laterally compressed and has a pointed snout and terminal mouth, with smooth and deciduous scales (Figure 1). The size of the Sardine can reach 27 cm; 90% of which is reached during the first year of its life cycle. Growth in subsequent years is much lower despite the longevity of up to 14 years [36]. In the North West Africa region, the size of the Sardine increases from North to South this is probably related to a trophic richness of the environment and a temperature generated by the upwelling to which these ribs are subjected.
B. Description and taxonomy
The most common species of Sardine correspond to the genus Sardina and constitute the group of small pelagics that dominate temperate and subtropical waters. Sardines belong to a complex taxonomic group that includes marine or freshwater pelagic fish such as shad and herring. The two main types of Sardine are distributed in the different upwelling zones of the world, where water is cold to temperate and where primary production is important [36,37]. Sardina pilchardus (Walbaum, 1792), also called Sardine commune.
C. Ecology of Sardina pilchardus:
(a) The diet: Sardines are usually microphagous filter feeders (zooplankton and/or phytoplankton). They occupy an intermediate position in the food chain and thus undergo predation at all stages of their development by individuals of the same species (larval stages) as by tuna, Scombridae, marine mammals, and some birds.
Sardines are planktonic, but the type of food ingested varies with age. Larvae feed mainly on phytoplankton (diatoms especially), while juveniles and adults have a more varied diet. During upwelling seasons, sardines can feed almost exclusively on phytoplankton and zooplankton. The digestive tract length of sardine is 1.5 times the length of the body, which is much larger than that of the other strictly zoophagous Clupeidae (0.5 times the length of the body).
This lengthening would be an adaptation to a phytoplankton diet of sardines.
The adult sardine feeds mainly on small crustaceans such as copepods, mollusk larvae, and fish eggs and fry [38], with a relative importance of these prey according to the sector of the season [39]. The composition of its diet reflects the planktonic composition of the environment [40].
(b) Geographical distribution: Sardina pilchardus is found in the North Atlantic, the Mediterranean, and the Black Sea. Its distribution extends along the Atlantic coast from the dogger-bank in the North Sea to the Saharan coast in Mauritania, with the remaining populations in the Madeira Islands and the Canary Islands [36]. Its location and abundance are strongly influenced by hydroclimatic conditions. The 10-13 °C isotherm marks [37,41].
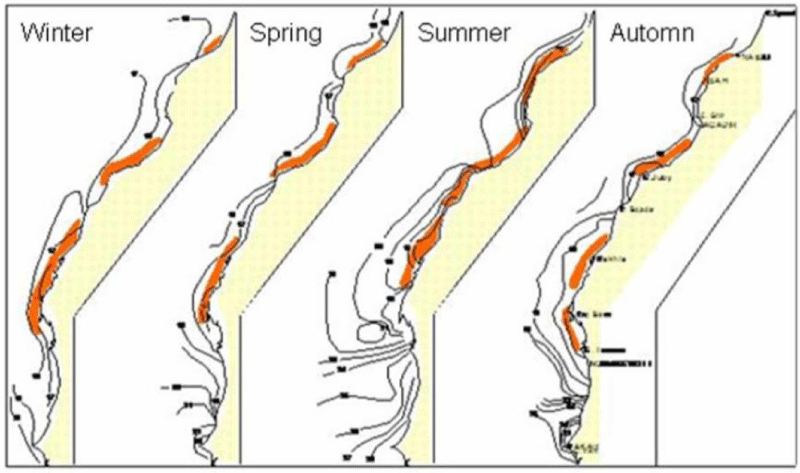
The range of the Sardine has periodically seen its limits expand or retract according to water temperature anomalies. In the mid-1960s and 1970s, the southern limit of the species extinction extended to Senegal, coinciding with an intensification of upwelling in this area and receding into the North in the late following [42] (Figure 2).
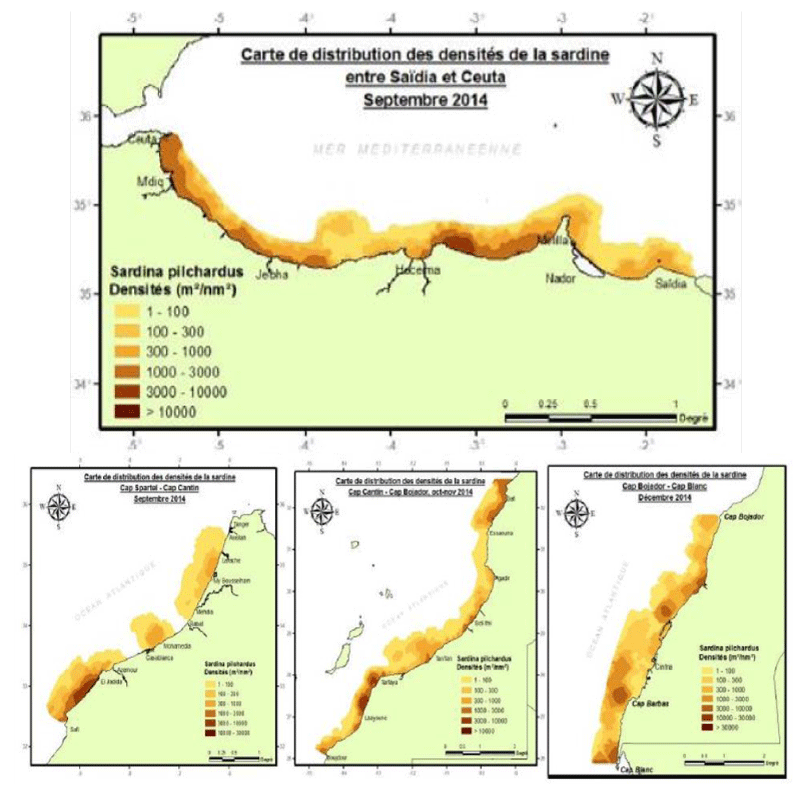
The spatial distribution of sardine along Moroccan coasts
At the level of the Mediterranean zone, sardines show a continuous distribution along the Moroccan coast. The highest densities are at Betouya Bay, especially near RasTarf, and along the coastal strip of the western zone between Jebha and Fnideq. In the North Atlantic, sardines are distributed in three distinct strata; the highest density is along the coastal fringe between Cape Cantin and El Jadida, followed by a less dense stratum extending between Mehdia and Assilah and a smaller one between Casablanca and Mohammedia. At the level of the Central Atlantic, the distribution of the sardine is continuous all along the coast. The most important concentrations are located north between Cape Cantin and Essaouira and are the continuation to the south of the high concentration detected north of Safi. The distribution is continuous between Agadir and Tarfaya; the highest densities are in the coastal fringes on both sides of Sidi Ifni and Tan Tan. Further south, very high densities are found between Tarfaya and Laayoune on bottoms ranging from the coast up to 100 meters offshore, while the detections are very coastal in the southern part of Laayoune. In the southern zone, the sardine shows a continuous distribution along the coast, from Cape Bojador to Cape Blanc. The densest concentrations are detected between Lakraa and Dakhla, at Cape Barbas, and at Cape Blanc [43]. (Figure 3).
c. Habitat: Sardina pilchardus can live at a depth of 180 m but is generally found at depths of 35 to 55 m during the day and 13 to 35 m at night [44]. Elderly sardines live further offshore than young ones [25]. In the Mediterranean, it lives on the level of the continental shelf, and in winter it is on the bottoms of 35 to 100 m. The compact beds that it forms during the day from 25 to 55 m, tend to disperse at night on the bottoms of 15 to 35 m.
(d) Behavior: Sardine is a gregarious species, but has a tendency to disintegrate at night [36]. Its banks may be composed of individuals of different ages and sex but of equivalent size [45]. In the case of high abundance, the schools may be mono-specific. On the other hand, if the Sardine is less abundant, the schools will be composed of several species of small pelagics, notably Anchovies and or Chinchards [45]. The Sardine makes vertical migrations during the day, these are conditioned by the light intensity and the amount of food. It is usually present at depths of 30 to 55 m during the day and rises between 15 and 35m the night after the zyoplankton nychthemeral migration [36]. It also performs horizontal migrations during the day by approaching the coast during the night [36]. In addition to its daily migrations, the Sardine makes larger trips depending on the season. In the Mediterranean, Sardines are not migratory because the ecological conditions of temperature and salinity are more stable.
On the other hand, she is a migrant in the Atlantic; where reproduction, nutrition, temperature, and salinity are factors influencing its migration, there is also another factor which is the age of the fish [41].
D. Reproductive biology of Sardine
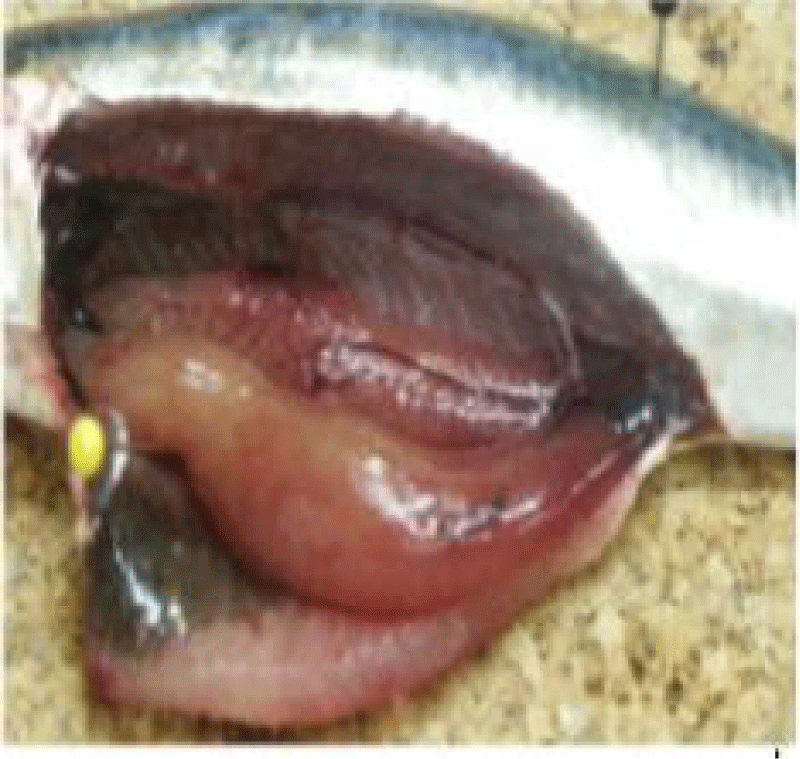
(a) The breeding cycles: Sardines present a life cycle that is essentially characterized by rapid growth, short life span, and rapid maturation associated with high fecundity and high mortality, especially in the larval phase [46]. It lives on the continental shelf at a maximum depth of 150m and its presence is often associated with that of anchovy, Engraulis encrasicolus [47].
Sardine reaches sexual maturity in the first two years of life. In general, the life cycle of a fish can be schematized by two phases, the larval phase and the adult phase, linked by two biological phenomena: recruitment and reproduction.
(b) The laying of the Sardine: Sardines lay mainly between September and June on the Atlantic coasts and in the Mediterranean, and from October to June on the African coasts [9,36,48], It is strongly correlated with environmental factors, such as temperature and hydrodynamics [22]. It occurs between 12 ° C and 18 ° C and extends over most of the continental shelf [9,8,49].
In the North-East Atlantic, Sardines preferentially lay eggs in winter, and in spring, the duration of oviposition increases from the north (1 to 2 months) to the south (6 months) [8,9,50].
In the Mediterranean, laying also extends over 6 months with a maximum in winter [51].
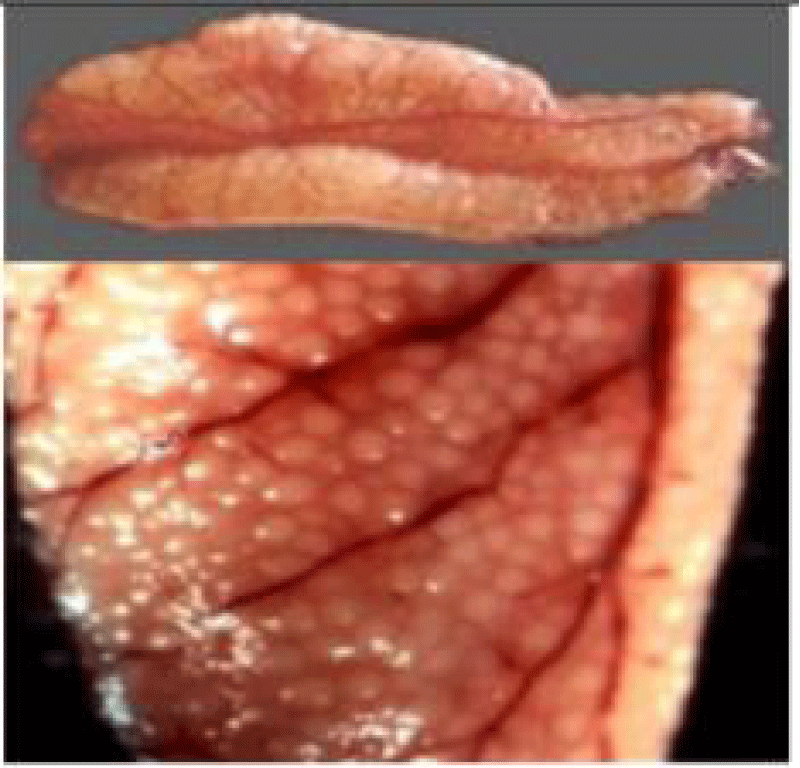
Sardines are highly fertile and each female can release up to 35,000 pelagic eggs [36]. However, larval mortality is important and strongly influences recruitment. The strategy used to compensate for the high potential mortality is based on an allocation of energy to reproduction, thus favoring the mass production of eggs [45,52]. The larval phase lasts 60 days and the larvae live between 10 and 40 m in depth and disperse more widely at night [22].
The Sardine is oviparous, she does not mate. However, there is a rapprochement of the sexes in bands [22]. Ovules and laitances are emitted in water, a single spermatozoon among the thousands of sperm that will penetrate through a micropyle to ensure fertilization [45].
c. Maximum breeding and laying period: The Sardine breeding season on the European Atlantic coasts lasts ten months, from September to June. The clutches are low from September to October, then they intensify quickly until the end of December to reach their maximum in early January. However, they remain quite strong until February, then decrease rapidly to end between May and June.
Spawning areas and nurseries are located in areas where production is able to meet the larval energy requirements and where surface drift does not cause excessive advective losses.
Eggs of sardine are present throughout the Moroccan coast [9,53,54].
The monthly monitoring of the gonado-somatic ratio (RGS) over several years has shown that in the Mediterranean zone, sardines breed on average between January and April and November. While in the Atlantic, sardine oviposition occurs throughout the year but mainly in winter and secondarily in spring and summer [9,48,54]. The maximum emission period varies according to the years between January and March in the Mediterranean region and between November and March in the central and southern Atlantic. Concentrations are found between Larrache and Casablanca, Cape Cantin and Cape Sim, Cape Ghir and Sidi Ifni, Cape Draa and Cape Juby, and between Cape Bojdor and Cintra Bay, and Cape Barbas and Cape Blanc.
Temperature is the most important external factor for sardine reproduction. It is to its variations that we must attribute the seasonal and geographical differences observed. The thermal margin in which spawning occurs is rather narrow, 15 ° to 20 °C, the optimum being between 16 and 18 °C. The spawning is weak and sparse if the winter is cold, it is strong and to a limited extent if the winter is [53]. Emission temperatures of sardine eggs are localized in the thermal window between 16 ° and 19 ° C in winter and between 18 ° and 20 ° C in summer [9,55], The laying takes place in an environment with precise thermal conditions and in an environment in which food is abundant. In fact, larval survival is dependent on the quantity and quality of food [56]. In contrast, salinity is not a determining factor for sardine oviposition [7]. Its average relative fertility is 401 oocytes per act of spawning and per gram of females in the area of Laayoune [48].
The size at first sexual maturity (L50) estimated in sardines at Moroccan (Mediterranean and Atlantic) coasts varies according to the year and the area considered. It fluctuates between 10 and 17 cm in total length, which corresponds to individuals in age group 0. A gradient of increase in the size of first sexual maturity as a function of latitude is observed from north to south of the Atlantic: 11.5 cm (Mediterranean), 12.4 cm (North Atlantic), 13.2 cm (Central Atlantic), and 17 cm (South Atlantic) [57].
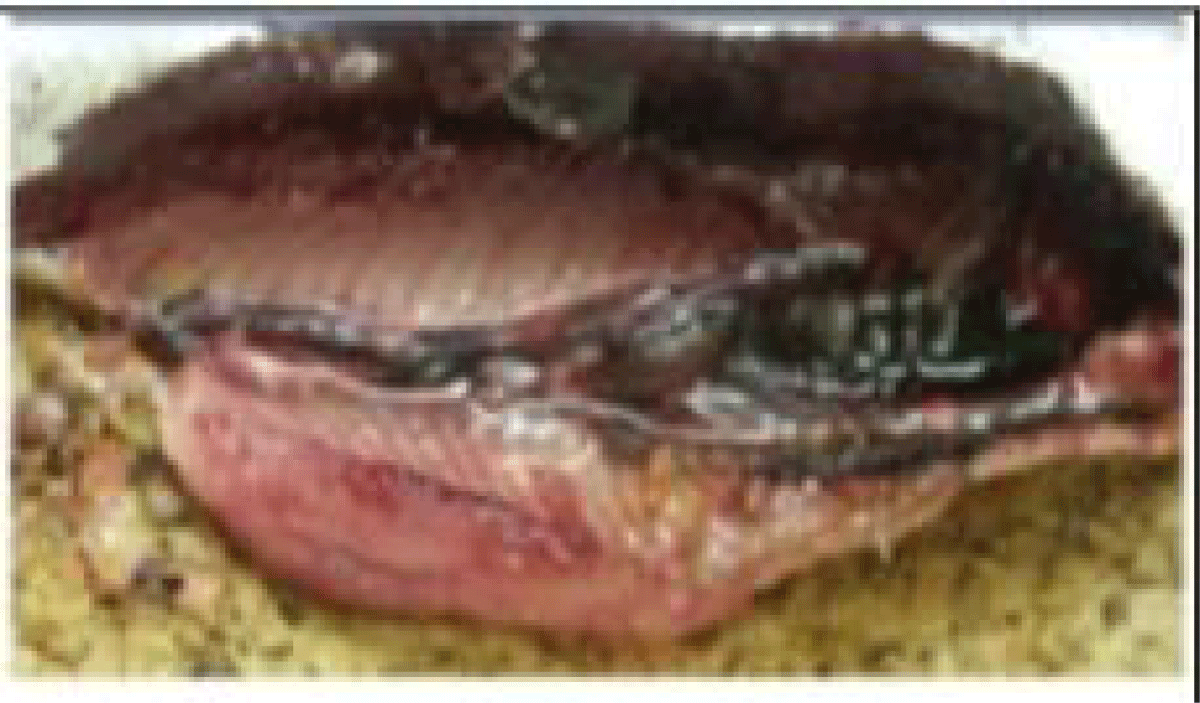
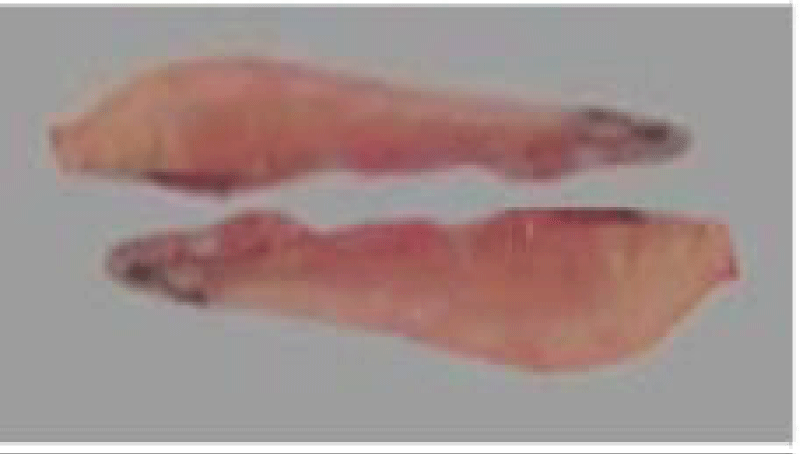
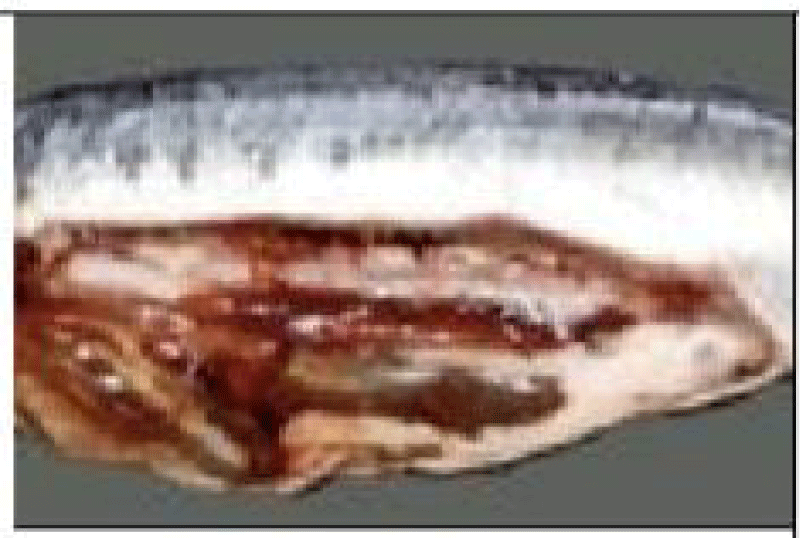
(d) Sexual maturity stages: The determination of sardine sexual maturity stages is based on macroscopic observation of female and male gonads, taking into account their color, consistency, vascularity, thickness, and transparency of the gonad wall [58]. The superficial vasculature, thickness, and transparency of the ovarian wall (possibility of observing the eggs through this wall), the shape and volume occupied by the gonads in the abdominal cavity, these morphological criteria make it possible to follow the evolution of the male and female gonads during the reproductive cycle. Using a macroscopic sexual developmental scale comprising five stages [59] (Tables 1,2).
E. Food interest of Sardine
The choice Sardine, as a food matrix, is a fish that is widely consumed, and very vulnerable to microbial alteration and lipid oxidation process. The nutritional quality of Sardine is largely related to the composition of these lipid deposits especially polyunsaturated chain fatty acids. In addition, the oxidation compounds formed may be harmful to fish and consumer health.
Proteins: Fish is a source of protein of high biological value as important as meat. Fish proteins are able to meet the growing needs of the child. Their digestive utilization coefficient of about 95% is explained. The lowest proportion of collagen in the connective tissue impairs digestibility. The presence of short and fine muscle fibers leads to better penetration of the digestive juices.
(a) Lipids: The lipid content is very variable depending on the species. Lean fish contain less than 5 % fat (cod, merlon, ray, etc.).
- Semi-fat fish contain 5 to 10% fat (herring, mackerel, salmon, etc.).
- Fatty fish: lipid contents exceed 10 %, such as Sardine, tuna, eel, etc.
The lipids of fish consist mainly of triglycerides having a high proportion of unsaturated fatty acids. Fish have the particularity of having low levels of cholesterol. The Sardine, like most Clupeids, goes through successive periods of fattening and weight loss. Those fattening correspond to the storage of large quantities of fat.
c. Vitamins: The flesh of the fish is rich enough in vitamins citing: - The water-soluble vitamins: the flesh of the fish contains appreciable quantities such as vitamins B1 and B2. Fatty fish are rich in vitamins A and vitamin D (1.5 ppm, and 0.7 ppm, respectively), which qualifies them as a very valuable food because they increase the resistance to infectious diseases, especially through their vitamin A intake. They provide children, adolescents, and the elderly with the vitamin D necessary for sufficient calcification of the skeleton.
d. Mineral salts: Fish, in general, is a food rich in minerals. Those in the sea are relatively rich in iodine.
F. Economic interest
Small pelagics are of great socio-economic importance, and their catches account for about 40% of the world’s catch of fish. However, the stocks and dynamics of these species are not known. The difficulty in studying them is related to the fact that these species often have irregular abundances probably due to high variability of recruitment, high mortality, excessive fishing [60], or following climatic or hydrological variations [61].
The common Sardine, Sardina pilchardus, is a highly exploited species in North West Africa and in the North Atlantic, it also presents, important fluctuations of the stocks [62]. As with other species and despite increasing exploitation, neither biogeography, geographical and seasonal limits, nor the dynamics and movements of sardine populations are well apprehended.
G. Linear growth
Sardine has rapid growth, especially in its juvenile phase, but it varies according to the period, the spawning area, and the sex [63]. The intensity of growth can be rapid in the spring and slowed or even halted during the winter [64]. The sardine has a very long growth in weight and weight during its first year of life and then the speed decreases when the age increases and this is whatever the region studied.
The size of the sardine can reach 27 cm of which 90% is reached during the first year of its cycle. Growth in subsequent years is much lower despite the longevity of up to 14 years [36]. In the North West Africa region, the size of the sardine increases from north to south [65]. This is probably related to a trophic richness of the environment and the temperature generated by the upwelling to which these ribs are subjected. The sardine reaches sexual maturity during the first two years of life. Growth and sexual maturity show wide variations throughout the range [66- 69].
H. Interactions of sardine and the Moroccan marine environment
The study of the relations of the fishery resources with their environment generally brings essential elements for the understanding of Spatio-temporal dynamics of these resources, their ecosystem interconnections, their strategies of reproduction, and adaptations specific to the natural and anthropic changes. For these purposes, the spatial and temporal dynamics of the sardine stock on the Atlantic coast of Morocco have been the subject of several studies which have led to the following results:
a. The Latitudinal Dynamics of Sardines: During the fall and winter seasons, fish tend to migrate south along the prevailing currents from north to south. Thus, at the central zone level, the highest densities are at the Tan Tan-Boujdor area (zone B), while the southern zone has the most dominant distribution zones in the southern region. From Dakhla to Cape Blanc, with an extension to the south that invades Mauritanian waters and often reaches Cape Timris [57] (Figure 4).
During warm seasons, the fish migrates in the opposite direction to the north. The highest densities in the central zone are located in the north between Agadir and Cap Cantin, while those in the southern zone stand out at Dakhla and reach Cap Boujdor [57].
b. Characterization of spawning and nursery areas: The Spatio-temporal distribution of eggs and larvae depends on the place of spawning, first, and then hydrological and trophic conditions that influence the distribution of ichthyoplankton. Variation in the oceanographic environment over an annual time scale can cause interannual changes in the distribution of adult fish, as well as in egg products [9].
Although analyzes of the maturity stages of sardine commercial catches have highlighted the main spike in spawning between November and March (Report of the small pelagics of [70]. The sardine oviposition seems to take place almost all the year.
Indeed, the campaigns carried out between 1994 and 1999 cover the months of January to April and June to August. In addition, and according to subsequent oceanographic surveys of INRH, laying eggs were also observed in May (2003), September (2004), October-November (2003) and November-December (2007) (Internal report of INRH, 2008). This peculiarity of the southern stock sardine compared to the central stock could be considered as an adaptation to the hydrodynamic characterizing the southern zone; the population would breed in an area where upwelling is almost permanent throughout the year and would also face the adverse effects of the hydrodynamics of the area (dispersal, drift, and loss to the open sea) [57].
On the other hand, the characterization of sardine spawning areas has resulted in a seasonal spatial pattern that confirms the seasonal dynamics of the pre-described species. In fact, the areas with the highest densities of cold-season eggs in autumn and winter are between Dakhla and Cap Barbas, while the highest densities of eggs recorded in the hot season are higher to the north, between Dakhla and latitude 25 ° N [57].
It was also shown that the main sardine spawning area, in the southern Moroccan Atlantic, was between Dakhla (24 ° N) and south of Boujdor (25 ° N), while the main nursery was located south, between Cintra Bay (23 ° N) and Dakhla (24 ° N) with some overlap in the Dakhla region [9].
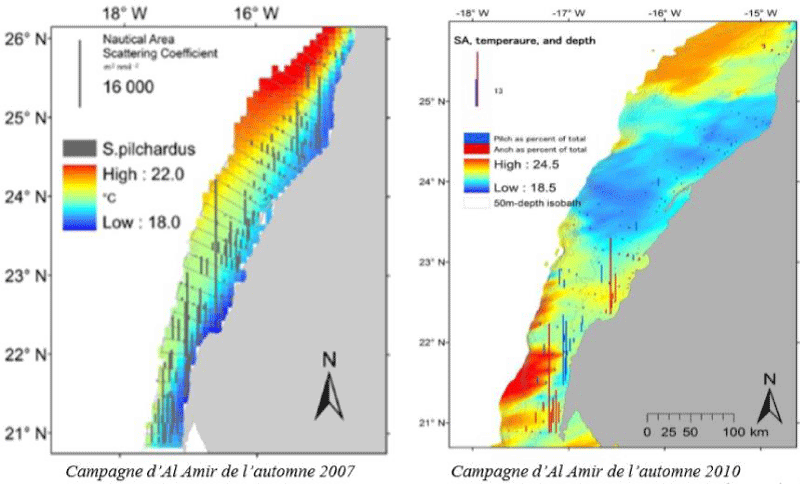
c. Spatial correlations of surface temperature and sardine densities: Surface temperature (SST) is one of the fundamental factors that characterize the state of the marine environment of small pelagics. To study the effect of SST on sardine density two cases are presented below: the case of a situation of “availability” of sardines, in autumn 2007, where the biomass exceeded 4 million tons, and a “scarcity” situation in autumn 2010, when biomass declined to about 2 million tonnes [57].
In the case of the year 2007, stratification of the temperature gradient is manifested between the coast and the sea at the level of the whole zone; the coldest temperatures (17.5 °C) occur near the coast and the warmer ones are located further offshore, especially at Boujdor (22 °C). In the two regions north and south of latitude 23 ° N, a strong negative correlation is observed between SST and sardine bank densities along the radial longitude between the coast and the open sea. The sardines detected are indeed more concentrated and denser in the cold waters near the coast, especially in the southern part has more densities in the autumn season (50% of the biomass is concentrated between Cape Blanc and Cape Barbas in 2007) [57]. The sardines present in autumn 2007 consisted generally of adult individuals (> 14cm). The near-absence of young individuals is confirmed by the low recruitment index evaluated in 2007 by Russian surveys in the region, by the acoustic survey carried out the following season in the summer of 2008, and by the reduction of about 25% of the biomass in the fall 2008 assessment [57]. (Figure 5).
In 2010, the stratification of temperature between the coast and the sea is much less obvious and the average value of the SST is higher than in 2007 (up to 24.5 ° C in 2010, surveyed offshore between Cape Blanc and Cape Barbas). Between latitudes 25 ° N and 23 ° N, the waters are relatively cold, but the temperatures are homogeneous between the coastal fringe and the continental slope. Detections of sardine are also spread in this zone to offshore isobaths (more than 100 meters), but the recorded densities are generally very low. South of latitude 23 ° N, the region knows a strong intrusion of the warm waters of the sea, especially between Cap Blanc and Cap Barbas. The spatial concentrations of sardine in this area are confined to a few very small patches of cold coastal waters, but densities at these points are very high [57].
This phenomenon of aggregation at the level of patches still tolerable to ensure the survival of the species explains its strategy of adaptation in extreme hydrological conditions by taking refuge in certain well-privileged areas [71].
The sardines detected in the south in 2010 are dominated by young individuals (< 15 cm). Their presence confirms the improved recruitment observed since 2009 in the results of the Russian campaigns conducted aboard the N / R Atlantniro [57].
I. The growth
Sardine has rapid growth, especially in its juvenile phase, but it varies according to the period, the spawning area, and the sex [72]. The intensity of growth can be rapid in the spring and slowed or even halted during the winter [73]. The sardine has a very long growth in weight and weight during its first year of life and then the speed decreases when the age increases and this is whatever the region studied.
The size of the sardine can reach 27 cm of which 90% is reached during the first year of its cycle. Growth in subsequent years is much lower despite the longevity of up to 14 years [36]. In the North West Africa region, the size of the sardine increases from north to south [74]. This is probably related to a trophic richness of the environment and the temperature generated by the upwelling to which these ribs are subjected. The sardine reaches sexual maturity during the first two years of life. Growth and sexual maturity show wide variations throughout the range [67- 69].
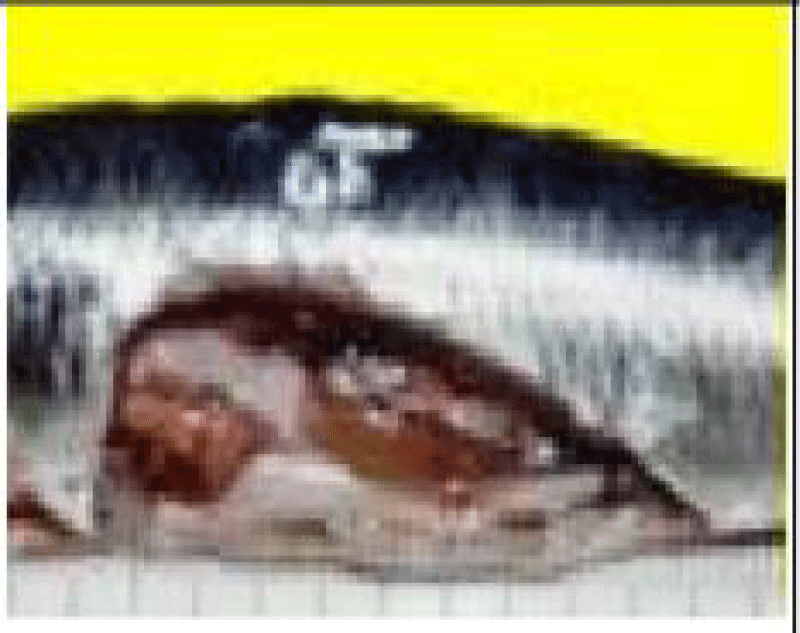
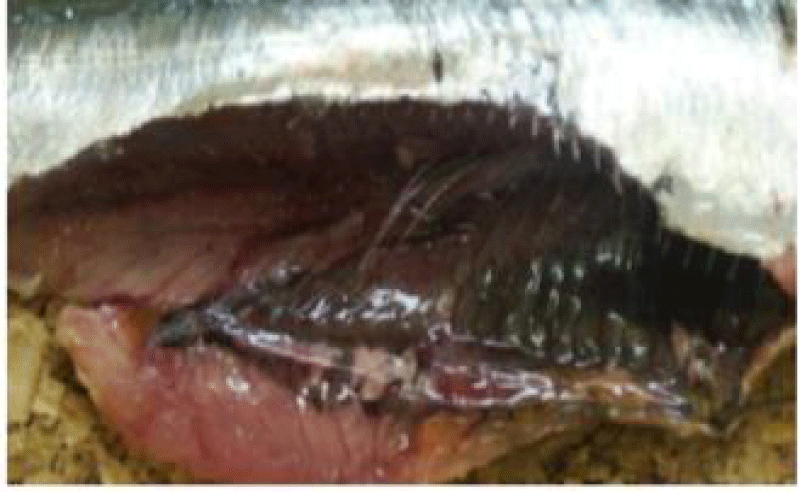
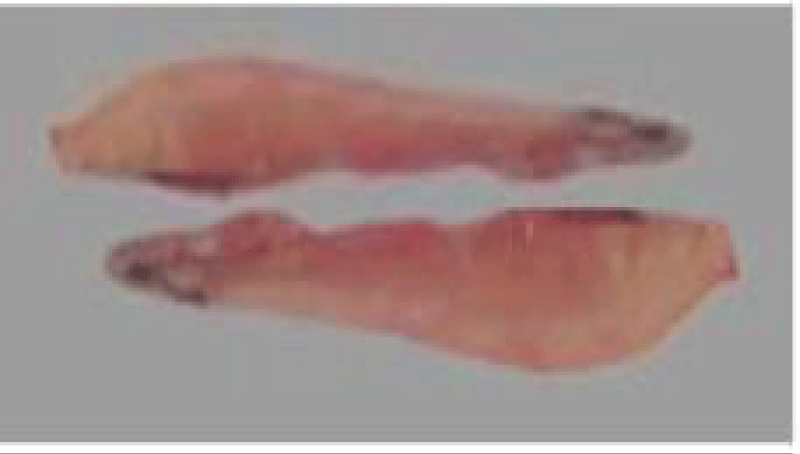
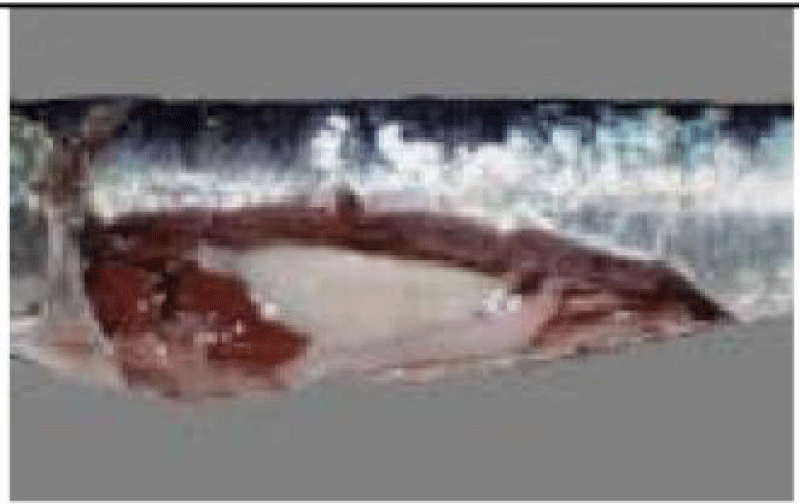
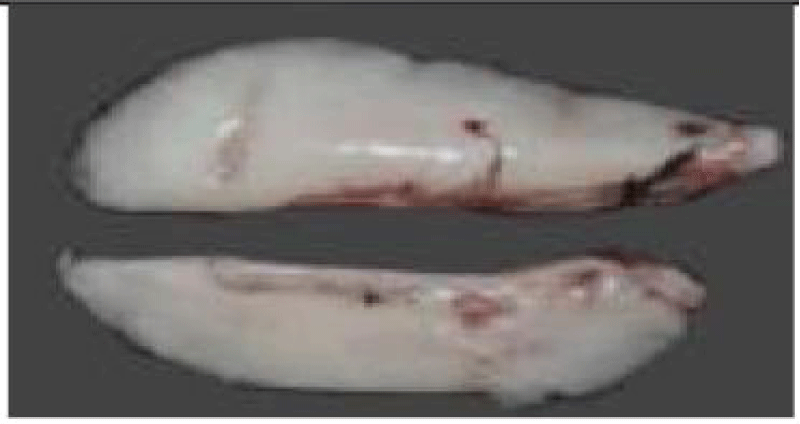
a. Otoliths: Description, Function, Reading, and Interpretation: All bone fish (Osteichthyens) have otoliths: small calcareous corpuscles in the membranous labyrinth inside the neurocranium. This labyrinth is on each side of the brain. It consists of three semicircular canals terminating at their base in an enlarged pocket, the utriculus, communicating in turn with two other diverticula, the sacculus, and the lagaena. Each of these three enlarged pockets contains an otolith, which floats freely in the endolymph whose pocket cavity is filled.
They help maintain and regulate muscle tone [75]. Each fish has 3 pairs of otoliths, lapilli, astericus, and sagittae. The latter is the largest and is most often used to interpret the age of bony fish.
Otolith growth begins with one or more primordia (crystals) secreted by the inner ear cells [76]. They then merge to form the nucleus. It is from this central area that the otolith develops. The extension of the central zone depends on the spawning period and the date of birth of the fish [77].
The otolith is increased by the deposition of material on its surface in the form of concentric layers in a cyclic manner and as a function of the internal rhythm of calcium metabolism and protein synthesis [78].
The growth of the otolith is not uniform in all directions but is preferentially along the anteroposterior axis. During the autumn and winter seasons, the low contribution of crystals results in the formation of a translucent layer mainly containing organic material giving it a hyaline appearance.
From the spring, many calcium crystals are deposited and creating an opaque zone, called (the rapid growth zone) and will continue during the summer. Each year, therefore, there are two distinct growth zones, which allow the estimation of age, by conventionally counting areas of slow growth.
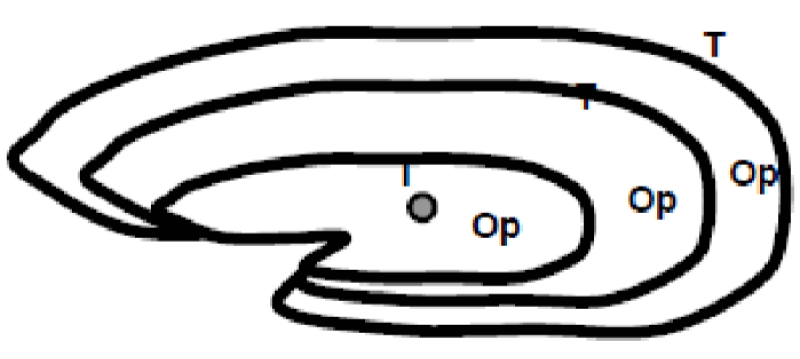
Thanks to the annual growth zones visible in otoliths, it is possible to calculate the age of a fish (Figure 6). These data are used to determine the age structure of a population and to estimate the effect of overfishing on this population. The annulus structure of otoliths is used to estimate the growth rate of very young fish. This structure also makes it possible to measure the duration of the reproduction cycle. In sardines, sagittae have an ellipsoidal, laterally compressed shape with a convex (outer) distal surface and a concave (internal) proximal surface carved out of a sulcus called sulcus acoustics. The anterior part is characterized by two advancements: the longest corresponds to the rostrum and the shortest to the antiroster. The ventral and dorsal edges may be smooth or ornamented with more or less numerous indentations depending on the individual and their age.
Environmental factors can lead to a slowdown or even a stop of growth which results in the formation of an intermediate ring: the pseudo-ring or false ring. There is another irregularity in the arrangement of the rings. It occurs mainly in adult sardines, from the third year. This is the duplication of the winter rings.
Other difficulties in reading otoliths are found in sardines and are related to the problem of identification of the first winter mark and that of the analysis of the edge of the otolith in the elderly (the last rings become less and less identifiable).
In this study, age determination was made according to the recommendations of international guidelines developed during sardine age reading standardization workshops [79]. January 1st is assigned as the theoretical date of birth for all sardines.
When otoliths were taken from fish caught in the first half of the year (January to June), the age group of these fish corresponds to the number of hyaline zones present, in the number of winter fish. The winter on the otolith corresponds to the zone of slow growth. (Figure 7). When the fish were collected during the second half of the year, their age group corresponds to the number of fully formed hyaline zones. Thus, if the edge of the otolith is a hyaline zone, it will not be counted.
J. The exploitation
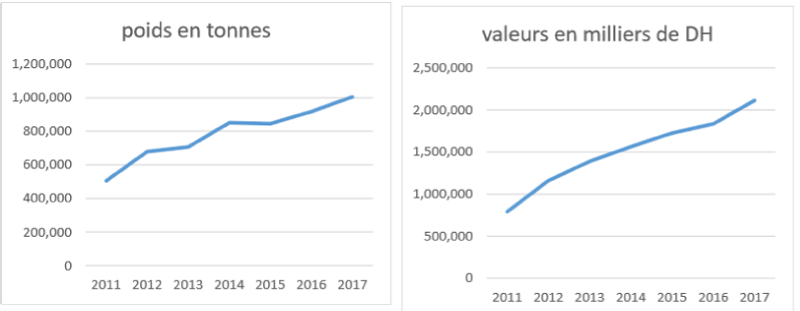
a. The production: Total sardine production reported during the 2011-2017 period varies according to the year and according to the means of access to the resource. The average catch in recent years is 786 881 tonnes.
This production increases from one year to another, reaching its maximum of 1,005,152 tons in 2017.
The sardine catch reported in 2017 in the Moroccan zone increased by 9.54% compared to 2016, from a catch of 917,554 tons to a catch of 1,005,152 tons. (Figure 8).
b. Development units: On the Moroccan coasts, sardines are found in the Mediterranean and in the Atlantic from Cape Spartel to Cape Blanc, but their distribution is discontinuous [58]. There are four stocks:
- The Mediterranean stock: distributed between Sebta and Saidia
- The North Atlantic stock: lies between Cape Spartel and El Jadida.
- The central Atlantic stock: extends from Safi to Boujador, this stock is subdivided into two zones:
Zone A: between Safi and Sidi Ifni.
Zone B: between Sidi Ifni and Cape Bojador
- South Atlantic stock: from Cape Bojador to Lagouira. It constitutes Zone C.
The southern limit of its distribution is conditioned by the position of the thermal front of the tropical waters.
Currently, two sardine stocks are genetically identified along the Moroccan Atlantic coast regardless of the season of study. A stock in northern Morocco includes sardines from Agadir, Safi, Larache, and Sidi Ifni, and another stock in southern Morocco stretch from Tarfaya to Dakhla [80]. The transition zone between the two stocks is between Sidi Ifni and Tarfaya, this zone is occupied by the southern stock in winter and the northern stock in summer. As a result, the limit between the two stocks is in winter at Sidi Ifni and in summer this barrier is at the Tarfaya level.
c. The current state of stocks: The Central sardine stock, as a fishing unit, is considered over-exploited. This stock is very dependent on recruitment that fluctuates with environmental changes. In 2010, a significant decrease in sardine biomass was the lowest in the last decade with a population structure dominated by small sizes. This situation was accompanied by the availability of anchovy in this area and a rise of the tropical sardinella, even reaching the northern zone.
The southern stock of sardines remains, however, not fully exploited. However, the sardine biomass in this area shows significant variations that are not attributable solely to fishing mortality but also to a response of the intrinsic dynamics of the stock to environmental factors. A precautionary approach is strongly recommended with respect to harvest levels and should be subject to annual adjustments based on indicators of stock status in terms of abundance and demographic composition of the population [70].
K. Aggression factors of sardines
There are several aggression factors of the Sardine, such as:
- Pollution: Human activities are responsible for the introduction into the marine environment of a large number of substances. According to UNEP (United Nations Environment Program), 80% of marine pollution is land-based and anthropogenic. The most common and most visible form of pollution is oil pollution caused by oil tanker accidents and the cleaning of bunkers at sea.
- Parasites: Marine domain parasites belong to several distinct taxa. Almost all free-living species carry parasites; according to Price (1980), parasites, therefore, contribute significantly to biodiversity by the number and variety of existing species.
Marine parasites can be divided into microparasites and macroparasites, based on their size. Microparasites include viruses, bacteria, fungi, protozoa, and myxozoa. Macroparasites are larger multicellular organisms: mostly Helminths and Arthropods. According to Cressey (1983), there are 3000 species of crustaceans described as parasites or semi-parasites; he brings back; moreover, and an incalculable number remains to be discovered.
- Biaz R, Rami M. Available data on pelagic fish from the northern Moroccan coast. FAO Fisheries Reports (FAO). 1978; 204.
- CECAF. Fisheries in the Eastern Central Atlantic. Report of the working group on the unification of age determination of the sardine Sardina pilchardus (Walbaum,1792). COPAC/TECH/78/8/Dakar (fr.). 1978; 9.
- Guerault D. Linear growth of the Bay of Biscay sardine. Its long-term variations. ICES. C. M/H. 1980; 40:9.
- Idrissi M, Zouiri M. Biostatistical data available on sardines and anchovies in the Moroccan Mediterranean. Report of the 4th CGPM technical consultation. Sidi Fredj, Algeria. 1985; 16-21:347; 99-105.
- Bencherifi S, Idrissi M, Lakhnigue A. Demographic structure of the stock of sardines Sardina pilchardus populating the Mediterranean coasts of Morocco. Report of the sixth technical consultation of the General Fisheries Council. 1989.
- Amenzoui K, Variability of the biological characteristics of sardines. Sardina pilchardus (Walbaum, 1792) exploited in the Safi areas. Agadir and Laâyoune (Moroccan Atlantic coasts). Doctoral thesis, Faculty of Sciences. Mohammed V University Rabat. 2010
- Bernal M et al. Sardine spawning off the European Atlantic coast: Characterization of spatiotemporal variability in spawning habitat. Prog. Oceanog. 2007; 74:10-227.
- Coombs SH et al. Spawning season and temperature relationships for sardine Sardina pilchardus in the Eastern North Atlantic. J. Mar. Biol. Ass. UK. 2006; 86:1245-1252.
- Ettahiri O, Berraho A, Vidy G, Ramdani M, Dochi T. Observation on the spawning of Sardina and European pilchardlla off the south Maroccan Atlantic coast (21-26°N). Fish. Res. 2003; 60:207-222p.
- Gushchin AV et Corten A. Feedind of pelagic fiche in waters of Mauritania: European Anchovy. Engraulis encasicolus, Europea Sardine, Sardina pilchrdus, Round Sardinelle, Sardinella aurita, and Fat Sardenella, S. maderensis. Journal of inchthylogy. 2015; 55:77-85.
- Fage L. Engraulidae, Clupeidae. Rep. Dan. Ocean. II; 9: 1920;1908-1910.
- Andreu B et Pinto JS. Histological and biometric characteristics of the sardine ovary (S. pilchardus) during maturation, laying and recovery, origin of the oocytes. Invest. Fishery. 1957; 6:3-38.
- Mozzi C, Duo A. Growth and age of sardines from the upper Adriatic, landed in Chioggia. Italy. proc. Gen. Fish. count. Tells me. 1959;5:105-112.
- Lopez J. Edal of the sardine, Sardina pilchardus, Walbaum, (1792) of Barcelona. Invest. Pesp. Barc; 1963; 23:133-157.
- Boulva J. New data on the sardine Sardina pilchardus (Walbaum, 1792). FAO, Rep. Peaches. 1975.
- Alvarez F. Growth studies of Sardina pilchardus (Walbaum, 1792) in Galician waters. N. W. Spain. ICES C.M. 1981/ H: 29. 1980; 11.
- Quignard JP, Kartas F. Observation on the sardine Sardina pilchardus (Walbaum, 1792), teleost fish from the Tunisian coasts during the winter of 1973-1974. (Numerical characters; height-weight relationship; sexual state). Report. ICES. 1976; 23 (8): 21- 25.
- Kartas F. The Clupeidae of Tunisia. Biometric and biological characteristics. Comparative study of the populations of the eastern Atlantic and the Mediterranean. State Doctorate Thesis, University of Tunis, Faculty of Sciences. 1981; 608.
- Lieonart J. The Catalunay Valencia fishery: global description and approach to the bases for achievement: Final report. Commission of the European Communications. DGXIV. 1990; 1634.
- Khemiri S. Reproduction, age and growth of the three species of pelagic teleosts of the Tunisian coasts: Engraulis encrasicalus, Sardina pilchardus and Boopsloops. Doctoral thesis from ENSAR, National School of Agronomics of Rennes Specialty in Fisheries. 2006.
- Nikolioudakis N et al. Diel feeding intensity and daily ration of the sardine Sardina pilchardus. Mar. Ecol. Prog. Ser. 2011; 437:215-228.
- Olivar MP, Salat J et Palomera I. Comparative study of spatial distribution patterns of the early stages of anchovy and pilchard in the NW Mediterranean Sea. Marine Ecology Progress Series 2001; 217:111-120.
- Silva A. Morphometric variation among sardine Sardina pilchardus populations from the northeastern Atlantic and the western Mediterranean. ICES Journal of Marine Science 60. 2003; 1352-1360.
- Bouchereau JL. Contribution to the study of the biology and exploited dynamics of Sardina pilchardus (Walbaum, 1792) in the bay of Oran (Algeria). 3rd cycle doctoral thesis, Aix-Marseille II University. 1981; 117.
- Mouhoub R. Contribution to the study of the biology and dynamics of the exploited population of the sardine Sardina pilchardus (Walbaum, 1792) from the Algiers coasts. Master's thesis, USTHB. Algiers. 1986; 163.
- Brahmi B, Bennoui A, Oualiken A. Estimation of the growth of the sardine Sardina pilchardus (Walbaum, 1792) in the central region of the Algerian coast. Marine populations dynamics, 1998, vol. 35 In: Lleonart J. (ed.). Dynamics of marine populations, Cahiers Options Méditerranéennes, CIHEAM. 1998; 35:57 – 64.
- Bedairia A, Djebar AB. A preliminary analysis of the state of exploitation of the sardine. Sardina pilchardus (Walbaum, 1792), in the Gulf of Annaba. East Algerian. Animal Biodiversity and Conservation. 2009; 3(2):89.
- Bedairia A. Biologie, écologie et exploitataion de la sardine, Sardina pilchardus (Walbaum, 1792) dans le littoral Est - Algérien. Thèse de Doctotat. Université Badji Mokhtar. Annaba. Algérie. 2011; 203.
- DPRH; Direction de Pêche et des Ressources Halieutiques de Jijel, 2018
- Wootton RJ. Ecology of Teleost Fishes. Chapman and Hall, New York. 1990; 404 .
- Jennings S, Kaiser MJ, Reynolds JD. Marine Fisheries Ecology. Oxford: Blackwell Science. 2001; 417.
- Holden MJ, Raitt DFS. Manual of fisheries science. Part 2-Methods of resource investigation and their application. Documents Techniques FAO sur les Peches (FAO)-Documentos Tecnicos de la FAO sobre la Pesca (FAO). 1974.
- Inrh. State of Stocks and Moroccan Fisheries 2002; 14.
- Furnestin J. Biology of Mediterranean Clupeidae. Life and environment. 1952; 4 (1):37–56.
- Bauchot ML. Fiches d’identification des espèces pour les besoins de la pêche (Révision 1). Méditerranée et mer Noire. Zone de pêche 37. Vertèbres. Rome. FAO; 1987; 2:761-1530.
- Whitehead PJP. FAO species catalogue. Clupeoid fishes of the world (Suborder Clupeoidei). Part 1. Chirocentridae, Clupeidae and Pristigasteridae. United Nations Development Program, Rome. -X-Y-Z- Bibliographie 210. 1985; 7
- Parrish RH, Mallicoate DL. Variation in the condition factors of California pelagic fishes and associated environmental factors. Fish. Oceanogr. 1995; 4(2): 171-190.
- Demirhindi U. Nutrition of the sardine (Sardina pilchardus Walb.). Proceedings and Technical Papers of the General Fisheries Council for the Mediterranean 1961; 6:253–259.
- Varela M, Larrañaga A, Costas E, Rodríguez B. Stomach contents of sardines (Sardina pilchardus, Walbaum) during the Saracus 871 campaign on the Cantabrian and Galician platforms in February 1987. Boletin del Instituto Espanol de Oceanografía 1988; 5:17–28.
- Conway DVP, Coombs SH, Fernandez ML de Puelles, Tranter PRG. Feeding of larval sardines, Sardina pilchardus (Walbaum), off the north coast of Spain. Bol. Inst. Esp. Oceanogr. 1994; 10: 165-175.
- Furnestin ML. Chaetognaths and zooplankton of the Moroccan Atlantic sector. Review of the work of the Institute of Maritime Fisheries. 1957; 21(1-2): 1-356.
- Fréon P, Stéquert B, Boëly T. Fishing for coastal pelagic fish in West Africa from the Bissagos Islands to the north of Mauritania: description of the types of exploitation. ORSTOM ser. Ocenaogr. 16(3 - 4): 209–228.
- Inrh, 2014, Etat des Stocks et des Pêcheries Marocaines 2014, 14.
- Fisher W, Bauchot ML, Shneider M. FAO Species Identification Sheets for Fishery Purposes (Revision 1). Mediterranean and Black Sea. Fishing area 37. Vertebrae. Rome. FAO. 1987; 2: 761–153.
- Cury P. Small pelagics in upwelling systems: patterns of interaction and structural changes in “wasp-waist” ecosystems. ICES Journal of Marine Science: Journal du Conseil. 2000; 57: 603–618.
- Rochet MJ, Cornillon PA, Sabatier R, Pontier D. Comparative analysis of phylogenetic and fishing effects in life history patterns of teleost fishes. Oikos. 91: 255-270.
- Abad R. Acoustic estimation of abundance and distribution of sardine in the Nortwestern Mediterranean. Fisheries research. 1998; 34: 239 - 245.
- Amenzoui K, Ferhan-Tachinante F, Yahyaoui A, Kifani S, Mesfioui AH. Analysis of the cycle of reproduction of Sardina pilchardus (Walbaum, 1792) off the Moroccan Atlantic coast. Comptes Rendus Biol. 2006; 329:892–901.
- Bernal M. Sardine spawning off the European Atlantic coast: Characterization of spatiotemporal variability in spawning habitat. Prog. Oceanog. 2007; 74: 210-227.
- Riveiro I, Guisande C, Lloves M, Maneiro I, Cabanas JM. Importance of parental effects on larval Survival in Sardina pilchardus. Marine Ecology Progress. Series. 2000; 205: 249-258.
- Abad, R. and Giraldez, A. (1993). Reproduction, facteur de condition et première taille de maturité chez la sardine, Sardina pilchardus (Walb.1792), Au large de la côte de Malaga, mer d'Alboran (1989-1992). Bulletin de l'Institut espagnol d'océanographie (Espagne).
- Bakun A. Patterns in the ocean: ocean progresses and marine population dynamics. Upwelling of California Sea Grant, San Diego, CA, USA, in cooperation with Centro de Investigaciones Biologicas de Noroeste, La Paz, Baja California Sur, Mexico, 1996: 323.
- Furnestin J, Furnestin ML. The Moroccan sardine and its fishing: Trophic and genetic migrations in relation to hydrology and plankton. Rapp. P.V CIEM. 159:165-175. 1970.
- Berraho A. Spatialized relationships between environment and ichthyoplankton of small pelagics off the Moroccan Atlantic coast (central and southern areas). Mohamed V University, Rabat, 2007; 261.
- Ettahiri O. Study of the planktonic phase of the sardine, Sardina pilchardus and the anchovy, Engraulis encrasicolus from the Moroccan Atlantic coasts. Doc. of university. University of Western Brittany, 262.
- Palomera I, Olivar P, Salat J, Sabatés A, Coll M, García A, Morales-Nin B. Small pelagic fish in the NW Mediterranean Sea: an ecological review. Progress in Oceanography. 2007; 74: 377–396.
- Inrh, bulletin halieutique, 2015: 17-21.
- Belvèze H. Biology and population dynamics of Sardine (Sardina pilchardus) populating the Moroccan Atlantic coasts and proposals for fisheries management. Doc. State. University of Western Brittany. 1984; 532.
- FAO. Fisheries in the Central – Eastern Atlantic. report of the Working Group on the unification of the age determination of sardines (Sardina pilchardus walb.). CECAF Technical report, 78/8/Dakar, 1978; 8.
- Schwartzlose RA, Alheit J, Bakun A, Baumgartner TR, Cloete R, Crawford RJM. Worldwide large-scale fluctuations of sardine and anchovy populations. S Afr J Mar Sci. 1999; 21: 289–347. doi: 10.2989/025776199784125962
- Lluch-Belda D, Crawford RJM, Kawasaki T, MacCall AD, Parrish RH, Schwartzlose RA. World-wide fluctuations of sardine and anchovy stocks: the regime problem. S Afr J Mar Sci. 8: 195–205. doi: 10.2989/02577618909504561
- ICES. Report of the workshop on sardneotolith age reading. ICES CM 1997/H: 7.
- Forest A. Non-quota fishery resources in the North East Atlantic: review of knowledge and analysis of management development scenarios. Ifremer Eds, tome. 2001; 2: 215.
- Lee JY. The Gulf of Lion sardine (Sardina pilchardus Sardina Regan), Review of the Works of the Institute of Maritime Fisheries. 1961; 25.
- FAO. Report of the working Group on the assessment of small-pelagic fish off northwest Africa. FAO Fisheries Report, 849: 3.
- Monteiro C, Jorge IM. Age abd growth of Sardine pilchardus (Walb.) From the Portuguese coast (ICES Div.IXa). ICES Document C.M. 1982/H: 19. 1982.
- Pérez N, Proteiro C, Alvarez F. Contribution to the knowledge of the biology of the Galician sardine. Boletin de l’Instituto Espanolde Oceanografia. 1985; 2: 27-37.
- Alemany F, Alvarez F. Grouth differences among sardine (Sardina pilchardus Walb.) populations in western Mediterranean. Sci Mar. 1993; 57:229-234.
- FAO. Sardine otolithworkshop.FAO Fisheries Report. 2001; 685: 49.
- Inrh, fisheries bulletin, 2013: 12-19.
- Kifani S, Gohin F. Dynamics of upwelling and spatio-temporal variability of the distribution of the Moroccan sardine Sardina pilchardus (Walbaum, 1792). 1991.
- Forest A. Non-quota fishery resources in the North East Atlantic: review of knowledge and analysis of management development scenarios. Ifremer Eds, tome. 2:215.
- Lee JY. Note complémentaire sur les sardinelles Sardinella aurita (Valenciennes, 1847) du golfe du Lion. Rapp Comm Int Mer Médit. 1961; 16(2):335–339.
- FAO. 2007. Report of the FAO Working Group on the Assessment of Small Pelagic Fish off Northwest Africa. Casablanca, Maroc, 4-9 Décembre 2006. Rapport du Groupe de travail de la FAO sur l’évaluation des petits pélagiques au large de l’Afrique nord-occidentale. Casablanca, Maroc, 4-9 Décembre 2006. FAO Fisheries Report/FAO Rapport sur les pêches. N°. 848. Rome, 2006. 107p.
- Lowestein O. The sence organs: the accousticolatralis system. In: “the physiology of fishes” 2:115-186. Brown M.E.ed Acad press. 1957; 526.
- Campana SE, Neilson JD. Microstructure of fish otoliths. Can J Fish Aquat Sci. 1985; 42:1014-1032.
- Deniel C. Les poissons plats (Téléostéens, Pleuronectiformes) en Baie de Douarnenez. Reproduction, croissance et migration des Bothidae, Scophthalmidae, Pleuronectidae el Soleidae. Thèse d’état, Université de Bretagne Occidentale. 1981; 476.
- Morales B. Parametros biologicos dels almonete Mullussurmuletus (L. 1758) en Mallorca. Bol Inst Esp Oceanogr. 1991; 7(2): 139-147.
- FAO. Report of the FAO Working Group on the Assessment of Small Pelagic Fish off Northwest Africa. Casablanca, Maroc, 4-9 Décembre 2006. Rapport du Groupe de travail de la FAO sur l’évaluation des petits pélagiques au large de l’Afrique nord-occidentale. Casablanca, Maroc, 4-9 Décembre 2006. FAO Fisheries Report/FAO Rapport sur les pêches. N°. 848. Rome, 2006; 107.
- Chlaida M, Kifani S, Lenfant P, Ouragh L. First approach for the identification of sardine populations Sardina pilchardus (Walbaum 1792) in the Moroccan Atlantic by allozymes. Marine Biology. 2005;149: 169–175.

Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.







 Save to Mendeley
Save to Mendeley
