Annals of Environmental Science and Toxicology
Fluoride Sources, Toxicity and Its Amelioration: A Review
Vijay K Bharti*, Arup Giri and Krishna Kumar
Cite this as
Bharti VK, Giri A, Kumar K (2017) Fluoride Sources, Toxicity and Its Amelioration: A Review. Ann Environ Sci Toxicol 2(1): 021-032. DOI: 10.17352/aest.000009In recent scenario, fluorosis is now going to be a severe problem throughout the globe due to toxic effects of fluoride (F) on both plants and animals. F presents in the halogenated group of the periodic table and has the characteristics of electronegativity. Natural geological sources and increased industrialization have contributed greatly to the increasing incidence of fluoride-induced human and animal health issues. In animals and human beings, it exerts adverse effects mainly through the attenuation of antioxidant defense mechanism and chelation of enzymatic cofactors. Thereafter, it causes metabolic disorders through interacting with various cellular processes such as gene expression, cell cycle, metabolism, ion transport, hormonal secretion, endocytosis, apoptosis, necrosis, and oxidative stress. These effects lead to dental mottling, skeletal dysfunctions including crippling deformities, osteoporosis, and other vital organs dysfunction. It was found that, water is the main source of fluoride intake to plants and animals, which further may go into food chain of human beings through consumption of high fluoride content plant and animal origin food. Several preventive and control measures have been developed to ameliorate the fluoride toxicity, like application of synthetic chemicals, plants bioactive molecules, and plant products like fruit pulp, seed mixture, and plant buckle products. Therefore, this article presents up-to-date information on the fluoride sources, toxicity and different amelioration measures to reduce fluoride level directly from water as well as application of different natural/synthetic products/molecules to ameliorate the toxic effects of fluoride in in-vivo models.
Introduction
In the halides group of the periodic table, fluoride (F) has great importancy due to its smallest size and most electro negativity. Although the mechanisms of F in biological forms are remains unclear but it has the unique chemical and biochemical properties for the size and reactivity [1-3]. It is ubiquitously present in soil, water, plants and air. In the animal body, F makes its presence through water and food. But, some of the recent studies indicate that, most of the F comes from pharmaceutical drugs (20%) and through agrochemicals (30-40%) [4,5]. The variability and presence of fluoride depends upon the location. It was found that F is present in the soil within the range of 10-1000 parts per million (ppm). However, in water it ranges from 0.5 to 2000 ppm. This incident depends upon the sources of water [6,7]. According to World Health Organization (WHO), F exposure to animals above the 1.5 ppm, set at chronic fluoride toxicity. Through water exposure, this type of toxicity is going to endemic in most of the countries across the world [8]. In USA, the normal level of F in drinking water is 4 mg/L [9]. But, in the European country, it is 0.8 ppm [10]. In India, most of the states are showing the greater level of F in drinking water [11]. Fluoride exerts its effects on plants also [12]. It attenuates all the cells and tissues, impaired the stomatal conductance. Simultaneously, it acts as the metabolic and reproductive inhibitor, impaired photosynthesis and respiration pathways. Ultimately, F caused even to plants death [13-18]. In animals, fluoride intoxication causing skeletal impairment, called as skeletal fluorosis. Recently, high fluoride intake has been associated with dental cancer and tumors of other organs. First clinical symptoms appeared like reduced in food intake and loss of body weight gain. After attenuating the antioxidant defence mechanism, F also affect to the gastrointestinal tract, brain, muscle etc.. [19-22]. To ameliorate these effects, several types of synthetic chemicals, herbal drugs, plant bioactive molecule, and plant natural products have been incorporated in the medicinal documentary. For example, melatonin, pineal proteins (epiphyseal proteins), quercetin, curcumin, ascorbic acid, lipoic acid, flavonoids, polyphenols have been found great role against the F toxicity [23-26]. The present review critically discusses on the fluoride sources, worldwide levels and its toxic effects on plants and animals. Furthermore, the article discusses the recent ameliorative steps developed through synthetic chemicals, plant bioactive molecules, and plant natural products.
Biochemistry of fluoride
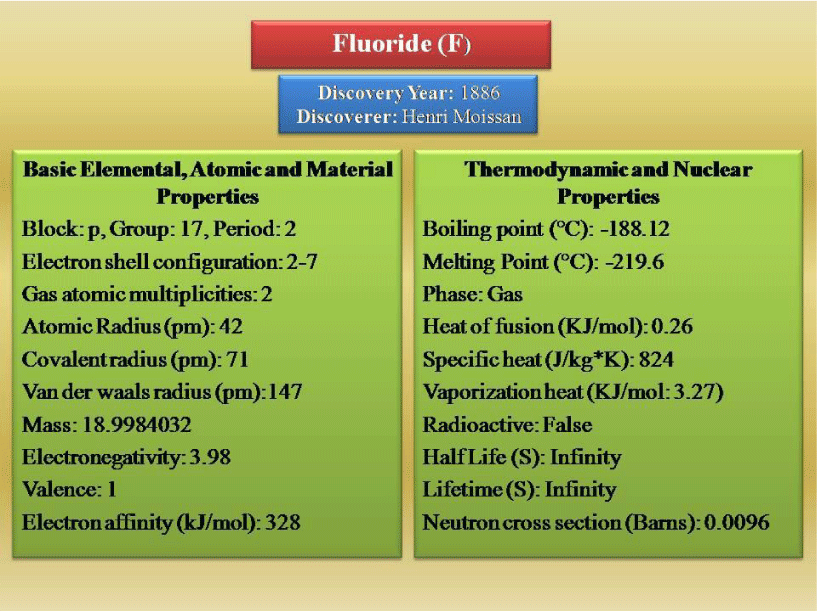
In the halides group of the periodic tables (group VII), among all other molecules, fluoride has the great importancy due to it’s smallest size and most electro negativity. Although, the mechanisms of F in biological forms are remains unclear but it has the unique chemical and biochemical properties for the size and reactivity [1-3]. It is 13th most abundant element and distributed widely throughout the earth in soil, water, and food. F, a pale yellow colored gas, has atomic number 9 and atomic weight of 18.9984 at standard temperature and pressure [27]. The brief about the F, have been mentioned in the Figure 1 [28]. It has the tendency to exist in the Free State as diatomic molecules. Due to electromotivity characteristics, these can react with less electromotive elements or chemical groups. Fluoride compounds are formed when the element fluoride combines with other chemical elements. It does not occur in a free state in nature [28]. Fluoride however has many unique chemical properties. These properties had a great impact on the special biochemical physiological effects. For these reason, F can affect the metabolism and mechanisms of action within the living system [29]. In addition to the chemical properties and isotopic nature of fluorine has had an important impact on our understanding of the metabolism, toxicity, and therapeutic effects of fluoride. 19-F is one of the isotopes of F and occurs naturally. This isotope has the extremely short half-life.
Sources of fluoride
Natural and anthropogenic sources are the two main ways through which F entered in the environment [30].
Natural sources
Soil: The normal total fluoride content of soil ranges from 150-400 mg/kg. F level in the clay soil is 1000 mg/kg [31]. F contamination to soil is because of the utilization of phosphorus fertilizers which have total 1-1.5% fluorine [32]. Contaminated soil with F, show it’s toxicity after the inhalation of soil contaminants which have vapourized or through the contaminated ground water after the F leaching from the soil [33-35].
Water: Water containing the F concentration up to 1.0 mg/L is safe. Whereas, the F levels in between 1.1 and 2.5 mg/L are marginally contaminated. However, above 2.6 mg/L F level is determined as the highly contaminated [31]. It was found that the level of F in ground water is higher than the surface water as the F percolates from the soil to ground water through leaching process. There are several factors which are responsible for the presence of F in natural ground water from the soil. Among them, geological factors, consistency of the soil, nature of rocks, pH and temperature of the soil, chelating action of other elements, depth of wells, leakage of shallow groundwater, and chemical and physical characteristics of water [36]. Water is an important source of F exposure to human beings and animals.
Forage, grasses and grains: At the vicinity of industrialized area, it was found that forages and grasses contain the higher level of F than the other area. Some studies also found that, grasses and forages has the higher level of F than the industrialized area. It is due to the fluoride rich dust, ash, raining factors for which plants could be affected far from the industry. Plants contamination depends upon several factors like the amount of F released in to the atmosphere, distance between the F source and contaminated area, type of vegetation, height of plants, atmospheric condition, and seasons etc. [37-39]. It has been established the relationship between the F level in soil and plants of F will be increased by 3 ppm for each 100 ppm increase in soil F up to the 2200 ppm [39].
Volcanic activities: Due to volcanic eruption, animals and plants kingdom have been affected throughout the globe (Table 1). Volcanic ash contains high level of F and contaminations of F to the geochemical cycle are frequent. From the volcanic eruption, F has been released in the form of hydrogen fluoride. Erupted F may covered several places and stay for many years. After decaying and leaching, F caused severe casualty to domestic and wild animals [6,40,41].
Anthropogenic sources
Anthropogenic fluoride contamination happens by human activities like industrialization, motorization, fluoride containing pesticides, fluoridation of drinking water supplies, dental products, refrigerants, and fire extinguishers [46,47]. F contamination due to airborne sources also occurred. The mean F concentration in normal areas (unpolluted/non-industrialized) is generally less than 0.1 μg/m3. The levels may be slightly higher in the vicinity of industries, but should not exceed 2–3 μg/m3 [6]. In many countries, coal burning for household purposes was documented as the main source of F causing endemic fluorosis [48,49]. Industrial release fluoride-rich fumes and effluents into the environment also caused casualty in livestock sector like cattle, buffaloes, sheep, goats, camels etc. [50-55]. There are several reports documenting mineral mixture supplements as a major source of fluoride toxicity in livestock [56]. Moreover, incorporation of modern creation and utilization of chemicals in different sectors like hydrogen fluoride (HF), calcium fluoride (CaF), sodium fluoride (NaF), fluorosilicic corrosive (H SiF), sodium hexafluorosilicate (Na SiF), sulfur hexafluoride (SF), and phosphate manures are the main sources of fluoride.
Global scenario of fluoride levels
Around the globe, twenty three nations are belongs to the critical region regarding the fluoride level. Among them India is also present. Billions of people are affected due to fluoride exposure. In India, twenty million people are severely affected by fluorosis and 40 million people are exposed to risk of endemic fluorosis [57]. Level of fluoride in drinking water throughout the globe has been tabulated in the Table 2.
Fluoride toxicity
In Animals: Chronic exposure to F induces an array of deleterious impacts in livestock animals, experimental animals, as well as humans also [6,97,98]. First symptoms of chronic F toxicity in animals are reduced feed intake and body weight gain (BWG) loss [19,22]. Prolonged exposure to F causes fluorosis, leading to a progressive degenerative disease, dental mottling and several types of skeletal dysfunctions [4]. Main mechanism of these deformities, after exposure of F is mainly the generation of different types of ROS production (Table 3). Experimental evidence (Tables 4,5) has indicated that exposure to fluoride results in oxidative stress both in vitro and in vivo in soft tissues such as liver, kidney, brain, lungs etc. Fluoride inhibits the activities of antioxidant enzymes like superoxide dismutase, glutathione peroxidase and catalase and reduces levels of glutathione. Glutathione reduction leads to overproduction of reactive oxygen species at the mitochondrial level, resulting in damage of cellular components. Besides, production of excessive reactive oxygen species results in oxidation of macromolecules, membrane phospholipid breakdown, lipid peroxidation, mitochondrial membrane depolarization and apoptosis (Tables 4,5). Neurodegeneration also occurred due to the F exposure. Several studies indicated that hippocampus of rat brain can lead to the degenerate due to the imbalance between oxidant– antioxidant balance. F crossed the blood brain barrier (BBB) easily and induces neural cell degeneration [24,99-101]. All the effects of fluoride are summarized in the Tables 4,5.
Amelioration of fluoride toxicity
Recently, various studies have been conducted in various fields like development of different techniques to reduce the fluoride level from the water sources directly, use plant metabolites on the experimental animals, and use of different chemical/molecule (melatonin, pineal protein, quercetin etc.). In case of different techniques, several natural and chemical adsorbents such as red soil, charcoal, brick, Waste tea ash, fly-ash, serpentine, alum, Activated carbon, Al-Fe (hydr) oxides, sulfate-doped Fe3O4/Al2O3 nanoparticles, aluminum salts etc have been used (Table 6). On the other hand, use of leaves, seeds, fruit pulps, plant juices of Azadirachta indica, Ficus religiosa, Acacia catechu, Peltiphyllum peltatum and tamarind seeds etc. are also using to reduce the toxic effects of fluoride and summarized in the Table 7. Additionally, some synthetic chemical molecules like melatonin, pineal protein, lycopene, and quercetin, etc. also have the great role to reduce the fluoride induced toxicity. All are summarized in the Table 8.
Conclusions
Through this review, it is summarized that having the electronegativity, fluoride is ubiquitously present in the environments. In some countries it is within the range, whereas most of the countries which have been reviewed showed more than the permissible level as per guideline recommended by WHO. Among different sources, water is the important source of fluoride exposure. Hence, water purification techniques should be developed for safe and economic method for portable water. High fluoride exposure affects human beings and animals health through oxidative stress, immune suppression, apoptosis, and affecting nutrient utilization. Hence, ameliorative measures are important to prevent their endemicity and disease progress. Meanwhile, plant bioactive molecules, several synthetic molecules, and pineal gland secretions have shown protective effect against fluoride toxicity. However, more extensive studies are required for wide application of these molecules as therapeutics agents.
- Jentsch TJ, Stein V, Weinreich F, Zdebik AA (2002) Molecular structure and physiological function of chloride channels. Physiol Rev 82: 503–568. Link: https://goo.gl/28dJtp
- Edwards JC, Kahl CR (2010) Chloride channels of intracellular membranes. FEBS Lett 584: 2102–2111. Link: https://goo.gl/CGDqTs
- Zimmermann MB (2011) The role of iodine in human growth and development. Semin Cell Dev Biol 22: 645–652. Link: https://goo.gl/F5pIa0
- Nabavi SM, Nabavi SF, Eslami S, Moghaddam AH (2012) In vivo protective effects of quercetin against sodium fluoride-induced oxidative stress in the hepatic tissue. Food Chem 132: 931–935. Link: https://goo.gl/IHEO8G
- Jagtap S, Yenkie MK, Labhsetwar N, Rayalu S (2012) Fluoride in drinking water and defluoridation of water. Chem Rev 112: 2454–2466. Link: https://goo.gl/9fdnB3
- Weinstein LH, Davison A (2004) Fluorides in the environment: effects on plants and animals. CABI Publishing, Cambridge. Link: https://goo.gl/bPwLXC
- WHO (2006). World Health Organization, Fluoride in Drinking water, Fawell J, Bailey K, Chilton J, Dahi E, Fewtrell L, and Magara Y, Eds., IWA Publishing, Alliance House, 12 Caxton Street, London SW1H 0QS, UK, 41–75. Link: https://goo.gl/aIhF6h
- WHO (2003). Background Document for Preparation of WHO Guidelines for Drinkingwater Quality. Fluoride in Drinking-water. Geneva: WHO. Link: https://goo.gl/4YSqwa
- U.S. Environmental Protection Agency. (2003) Ground Water and Drinking Water. Drinking Water Contaminants (Online 2003). Link: https://goo.gl/8qPCS2
- European Commission. (2011) “Critical review of any new evidence on the hazard profile, health effects, and human exposure to fluoride and the fluoridating agents of drinking water,” Scientific Committee onHealth and Environmental Risks (SCHER). Link: https://goo.gl/fn283a
- Planning Commission, India. (2007) Eleventh five-year plan approach paper. Rural watersupplyandsanitation. Link: https://goo.gl/VgUWJk
- Franzaring J, Klumpp A, Fangmeier A (2007) Active Biomonitoring of Airborne Fluoride near an HF Producing Factory Using Standardized Grass Cultures. Atmos Environ 41: 4828- 4840. Link: https://goo.gl/GWku0c
- Alves ES, Moura BB, Domingos M (2008) Structural Analysis of Tillandsia usneoides L. Exposed to Air Pollutants in São Paulo City-Brazil. Water Air Soil Pollu, 189: 61-68. Link: https://goo.gl/tZOEpo
- Reddy MP, Kaur M (2008) Sodium fluoride induced growth and metabolic changes in Salicornia brachiata Roxb. Water Air Soil Pollut 188: 171-179. Link: https://goo.gl/TUhXeP
- Lovelace CJ, Miller GW (1967) In vitro effects of fluoride on tricarboxylic acid cycle dehydrogenases and oxidative phosphorylation: Part I. J Histochem Cytochem 15: 195-201. Link: https://goo.gl/4pAqBe
- Melchior NC, Melchior JB (1956) Inhibition of yeast hexokinase by fluoride ion. Sci 124: 402-403.
- Lee C, Miller GW, Welkie GW (1965) The effects of hydrogen fluoride and wounding on respiratory enzymes in soybean leaves. Air Water Pollut Int J 10: 169-181.
- Miller JE, Miller GW (1974) Effects of fluoride on mitochondrial activity in higher plants. Physiol Plant 32: 115–121. Link: https://goo.gl/sguKvD
- Khandare AL, Kumar PU, Shankar HN. (2007) Effect of calcium deficiency induced by fluoride intoxication on lipid metabolism in rabbits. Fluoride 40: 184–189. Link: https://goo.gl/UWS01J
- Coetzee PP, Coetzee LL, Puka R, Mubenga1 S (2003) Characterization of selected South African clays for defluoridation of natural waters. Water SA 29: 331–338. Link: https://goo.gl/GniZ6V
- Dunipace AJ, Edward JB, Wilson ME (1998) Chronic fluoride exposure does not cause detrimental, extra skeletal effects in nutritionally deficient rats. J Nutr 128: 1392–1400. Link: https://goo.gl/N8G15g
- Lohakare J, Pattanaik A, Khan SA (2010) Effect of long-term fluoride exposure on growth, nutrient utilization and fluoride kinetics of calves fed graded levels of dietary protein. Biol Trace Elem Res 138: 148–162. Link: https://goo.gl/lzBFk5
- Heber D (2010) Pomegranate. Chapter 30. In Nutrition and Health: Bioactive Compounds and Cancer. Edited by J.A. Milner and D.F. Romagnolo. Humana Press, c/o Springer Science and Business Media LLC, New York 725-734. Link: https://goo.gl/QoGK92
- Nabavi SF, Eslami SH, Moghaddam AH, Nabavi SM (2011) Protective effects of curcumin against fluoride-induced oxidative stress in the rat brain. Neurophysiol 43: 287-291. Link: https://goo.gl/VRqZVv
- Nabavi SM, Nabavi SF, Eslami S, Moghaddam AH (2012a) In vivo protective effects of quercetin against sodium fluoride-induced oxidative stress in the hepatic tissue. Food Chem 132: 931-935. Link: https://goo.gl/eQA1JV
- Nabavi SF, Nabavi SM, Abolhasani F, Moghaddam AH, Eslami S (2012b) Cytoprotective effects of curcumin on sodium fluoride induced intoxication in rat erythrocytes. Bull Environ Contam Toxicol 88: 486-490. Link: https://goo.gl/xxeyxK
- Finger GC (1961) Fluorine Resources and Fluorine Utilization. Adv Fluorine Chem 2: 35-54.
- Giri A, Bharti VK, Angmo K, Kalia S, Kumar B (2016) Fluoride versus Oxidative stress, Immune System and Apoptosis in Animals: a Review. Int J Bioass 5: 5163-5173. Link: https://goo.gl/1QiNES
- Department of Health and Human Services. (1991) Report of the subcommittee on fluoride of the Committee to Coordinate Environmental Health and Related Programs, USPHS. Review of fluoride: benefits and risks. Public Health Service. Link: https://goo.gl/SF4MCG
- Cengeloglu Y, Esengul K, Ersoz M (2002) Removal of Fluoride from aqueous Solution by Using red mud. Sep Pur Tech 28: 81-86. Link: https://goo.gl/Dn1unl
- Susheela AK (1999) Fluorosis management programme in India. Curr Sci India 77: 1050–1256. Link: https://goo.gl/V4VjHH
- Bombik E, Bombik A, Gorski K, Saba L, Bombik T, et al. (2011) Effect of Environmental Contamination by Fluoride compounds on selected horse tissues. Polish J Environ Stud 20: 37-43. Link: https://goo.gl/Ev5Ob6
- Begum A (2012) Soil Profiles and Fluoride Adsorption in Intensely Cultivated Areas of Mysore District, Karnataka, India. Chem Sci Trans 1: 410-414. Link: https://goo.gl/d1YElI
- Blagojevic S, Jakovljevic M, Radulovic M (2002) Content of Fluorine in Soils In The Vicinity of Aluminium Plant in Podgorica. J Agricul Sci 47: 1‑8. Link: https://goo.gl/pzHtdc
- Ericson B, Hanrahan D, Kong V (2014) The world’s worst pollution problems; the top ten of the toxic twenty. Link: https://goo.gl/4saMAJ
- Li C, Gao X, Wang Y (2014) Hydrogeochemistry of high-fluoride groundwater at Yuncheng Basin, northern China. Sci Total Environ 508C: 155–165. Link: https://goo.gl/FA4STx
- NRC (1960) The Fluorosis problem in livestock production. A report of the NRC committee on animal nutrition. Publication 824, National Research Council, Washington. Link: https://goo.gl/EFXplv
- Radostits OM, Gay CC, Blood DC, Hinchcliff KW (2000) Veterinary Medicine, a textbook of the diseases of cattle, sheep, pigs, goats and horses, 9th edn. WB Saunders Company Ltd, London. Link: https://goo.gl/ixXkHL
- Mascola JJ, Barth KM, McLaren JB (1974) Fluoride intake of cattle grazing fluoride-contaminated forage, as determined by esophageal-fistulated steers. J Anim Sci 38: 1298–1303. Link: https://goo.gl/xdgvvv
- Araya O, Wittwer F, Villa A (1993) Evolution of fluoride concentration in cattle and grass following a volcanic eruption. Vet Hum Toxicol 35: 437–440. Link: https://goo.gl/IDlfgN
- Bellomo S, Aiuppa A, D’Alessandro W, Parello F (2007) Environmental impact of magmatic fluorine emission in the Mt. Etna area. J Volcanol Geoth Res 165: 87–101. Link: https://goo.gl/vi0hp9
- Thorarinsson S (1979) On the damage caused by volcanic eruptions with special reference to tephra and gases. In: Sheets PD, Grayson DK (eds) Volcanic activity and human ecology. Academic Press, New York, 125–159.
- SEAN (1989) Lonquimay, continued tephra emission: cattle sickened by ash. Scientific Event Alert Network Bull, Smithsonian Institution 14: 2–3.
- Armienta MA, de La Cruz-Reyna S, Cruz O, Ceniceros N, Aguayo A, et al. (2011) Fluoride in ash leachates: environmental implications at popocatepetl volcano, central Mexico. Nat Hazards Earth Syst Sci 11: 1949–1956. Link: https://goo.gl/Y2qCZr
- Flueck WT, Smith-Flueck JA (2013) Severe dental fluorosis in juvenile deer linked to a recent volcanic eruption in Patagonia. J Wildlife Dis 49: 355–366. Link: https://goo.gl/oQO8DP
- WHO (2002) World Health Organization, Geneva, Fluorides, Environmental Health Criteria, 227.
- Paul ED, Gimba CE, Kagbu JA, Ndukwe GI, Okibe FG (2011) Spectrometric Determination of Fluoride in Water, Soil and Vegetables from the Precinct of River Basawa, Zaria, Nigeria. J Basic Appl Chem 1: 33-38. Link: https://goo.gl/c95FyM
- WHO (2000) Fluorides. In: Chapter 6.5 Air quality guidelines, 2nd edn. WHO regional office for Europe, World Health Organization, Copenhagen.
- Guijian L, Liugen Z, Duzgoren-Aydin NS, Lianfen G, Junhua L et al. (2007) Health effects of arsenic, fluorine, and selenium from indoor burning of Chinese coal. Rev Environ Contam Toxicol 189: 89–106. Link: https://goo.gl/Sf7JAD
- Swarup D, Dwivedi SK (2002) Environmental pollution and effects of lead and fluoride on animal health. Indian Council of Agricultural Research, Pusa, New Delhi. Link: https://goo.gl/3d5B5B
- Swarup D, Dey S, Patra RC, Dwivedi SK, Ali SL (2001) Clinico-epidemiological observation of industrial bovine fluorosis in India. Indian J Anim Sci 71: 1111–1115. Link: https://goo.gl/rPNGGc
- Patra RC, Dwivedi SK, Bhardwaj B, Swarup D (2000) Industrial fluorosis in cattle and buffalo around Udaipur, India. Sci Total Environ 253: 145–150. Link: https://goo.gl/is32K0
- Sahoo N, Singh PK, Ray SK, Bisoi PC, Mahapatra HK (2003) Fluorosis in sheep around an aluminium factory. Indian Vet J 80: 617–621. Link: https://goo.gl/zSW3gJ
- Sahoo N, Ray SK (2004) Fluorosis in goats near an aluminium smelter plant in Orissa. Indian J Anim Sci 74: 48–50. Link: https://goo.gl/WPKkDk
- Karram MH, Ibrahim TA (1992) Effect of industrial fluorosis on haemogram of camels. Fluoride 25: 23–36. Link: https://goo.gl/dh5oDh
- Singh JL, Swarup D (1995) Clinical observations and diagnosis of fluorosis in dairy cows and buffaloes: case report. Agri Practice 16: 25–30.
- Chinoy JN (1991) Effects of fluoride on physiology of animals and human beings. Indian J Environ Toxicol 1: 17-32.
- Grobler SR, Dreyer AG, Blignaut RJ (2001) Drinking water in South Africa: Implications for fluoride supplementation. J South Afr Dent Assoc 56: 557–559. Link: https://goo.gl/7xnSHG
- Mothusi B (1998) Psychological effects of dental fluorosis, Fluoride and Fluorosis, The Status of South African Research, Pilanesberg National Park, North West Province, 7, (1995): as cited in Muller WJ, Heath RGM, Villet MH, Finding the optimum: Fluoridation of potable water in South Africa. Water SA, 24: 1–27.
- WHO (2005) World Health Organization, Geneva, Switzerland. Link: https://goo.gl/woF1q5
- Apambire WB, Boyle DR, Michel FA (1997) Geochemistry, genesis, and health implications of fluoriferous ground waters in the upper regions of Ghana. Environ Geol 33: 13–24. Link: https://goo.gl/CeENsA
- Kaimenyi TJ (2004) Oral health in Kenya. Int Dent J 54: 378–382. Link: https://goo.gl/VKAjUS
- Nair KR, Manji F (1982) Endemic fluorosis in deciduous dentition—A study of 1276 children in typically high fluoride area (Kiambu) in Kenya. Odonto-Stomatologie Tropicale 4: 177–184. Link: https://goo.gl/GXKvU4
- Nair KR, Manji F, Gitonga JN (1984) The occurrence and distribution of fluoride in groundwaters in Kenya. East Afr J Med 61: 503–512. Link: https://goo.gl/rhgcUL
- Wongdem JG, Aderinokun GA, Sridhar MK, Selkur S (2000) Prevalence and distribution pattern of enamel fluorosis in Langtang town, Nigeria. Afr J Med Medical Sci 29: 243–246. Link: https://goo.gl/rWqVEw
- Brouwer ID, Dirks OB, De-Bruin A, Hautvast JGAJ (1988) Unsuitability of World Health Organization guidelines for fluoride concentrations in drinking water in Senegal. Lancet 30: 223–225. Link: https://goo.gl/282Cqj
- Mjengera H, Mkongo G (2003) Appropriate deflouridation technology for use in flourotic areas in Tanzania. Physics Chem Earth 28: 1097–1104. Link: https://goo.gl/sWTTM7
- Rwenyonyi CM, Birkeland JM, Haugejorden O, Bjorvatn K (2000) Age as a determinant of severity of dental fluorosis in children residing in areas with 0.5 and 2.5 mg fluoride per liter in drinking water. Clin Oral Invest 4: 157–161. Link: https://goo.gl/fNLxny
- Ibrahim YE, Affan AA, Bjorvatn K (1995) Prevalence of dental fluorosis in Sudanese children from two villages with respectively 0.25 mg/L and 2.56 mg/L F in the drinking water. Int J Paediatr Dent 5: 223–229. Link: https://goo.gl/3K3OQL
- Smith DA, Harris HA, Kirk R (1953) Fluorosis in the Butana, Sudan. J Tropic Med Hyg 56: 57–58. Link: https://goo.gl/wd3zrZ
- Al-Khateeb TL, Al-Marasafi AI, O’Mullane DM (1991) Caries prevalence and treatment need amongst children in an Arabian community. Commun. Dentistry Oral Epidemiol 19: 277–280. Link: https://goo.gl/lJVBxY
- Akpata ES, Fakiha Z, Khan N (1997) Dental fluorosis in 12–15-year-old rural children exposed to fluorides from well drinking water in the Hail region of Saudi Arabia. Comm Dentistry Oral Epidemiol 25: 324–327. Link: https://goo.gl/OAmBxw
- Health Canada (1993) Priority Substances List Assessment Report on Inorganic Fluorides, Canadian Environmental Protection Act, Minister of Supply and Services Canada, Canada Communication Group-Publishing, Ottawa, Canada K1A 0S9, 12–19. Link: https://goo.gl/llzi4a
- Ismail AI, Messer JG (1996) The risk of fluorosis in students exposed to a higher than optimal concentration of fluoride in well water. J Public Health Dent 56: 22–27. Link: https://goo.gl/usvYm7
- Paoloni JD, Fiorentino CE, Sequeira ME (2003) Fluoride contamination of aquifers in the southeast subhumid pampa, Argentina. Environ Toxicol 18: 317–320. Link: https://goo.gl/iIZosO
- Cortes DF, Ellwood RP, O’Mullane DM, de Magalhaes Bastos JR (1996) Drinking water fluoride levels, dental fluorosis and caries experience in Brazil. J Public Health Dentistry 56: 226–228. Link: https://goo.gl/RqfSlb
- Haimanot RT, Fekadu A, Bushra B (1987) Endemic fluorosis in the Ethiopian Rift Valley. Tropic Geogr Med 39: 209–217. Link: https://goo.gl/6NkJq2
- Kloos H, Tekle-Haimanot R, Kloos H, Zein AH (1993) The Ecology of Health and Disease in Ethiopia, Westview Press, Boulder, CO, 445–541.
- Diaz-Barriga F, Navarro-Quezada A, Grijalva MI, Grimaldo M, Loyola-Rodriguez JP et al. (1997) Endemic fluorosis in Mexico. Fluoride 30: 233–239.
- Cohen D, Conrad MH (1998) 65,000 GPD fluoride removal membrane system in Lakeland, California, USA. Desalination 117: 19–35. Link: https://goo.gl/hOkasl
- Reardon JE, Wang Y (2000) A limestone reactor for fluoride removal from wastewaters. Environ Sci Technol 34: 3247–3253. Link: https://goo.gl/fKCdDk
- Heikens A, Sumart S, vanBergen M, Widianarko B, Fokkert L et al. (2005) The impact of the hyperacid Ijen Crater Lake: Risks of excess fluoride to human health. Sci Tot Environ 346: 56–69. Link: https://goo.gl/GJhB3H
- Queste A, Lacombe M, Hellmeier W, Hillermann F, Bortulussi B et al. (2001) High concentrations of fluoride and boron in drinking water wells in the Muenster region—Results of a preliminary investigation. Int J Environ Health 203: 221– 224. Link: https://goo.gl/iZknCh
- Kim K, Jeong YG (2005) Factors influencing natural occurrence of fluoride-rich groundwaters: A case study in the southeastern part of the Korean Peninsula. Chemosphere 58: 1399–1408. Link: https://goo.gl/K2y2CJ
- Bardsen A, Klock KS, Bjorvatn K (1999) Dental fluorosis among persons exposed to high- and low-fluoride drinking water in western Norway. Commun Dentistry Oral Epidemiol 27: 259–267. Link: https://goo.gl/21NdXZ
- Czarnowski W, Wrzesniowska K, Krechniak J (1996) Fluoride in drinking water and human urine in Northern and Central Poland. Sci Total Environ 191: 177–184. Link: https://goo.gl/UyUi99
- Hardisson A, Rodriguez MI, Burgos A, Flores LD, Gutierrez R et al. (2001) Fluoride levels in publically supplied and bottled drinking waters in the island of Tenerife. Spain. Bull Environ Contamin Toxicol 67: 163–170. Link: https://goo.gl/rF0qtH
- Wang GQ, Huang YZ, Xiao BY, Qian XC, Yao H et al. (1997) Toxicity from water containing arsenic and fluoride in Xinjiang. Fluoride 30: 81–84. Link: https://goo.gl/dvIRx4
- Azbar N, Turkman A (2000) Defluoridation in drinking waters. Water Science and Technology. 42: 403–407. Link: https://goo.gl/XQE0vb
- Tsutsui A, Yagi M, Horowitz AM (2000) The prevalence of dental caries and fluorosis in Japanese communities with up to 1.4 ppm of naturally occurring fluoride. J Public Health Dentistry 60: 147–153. Link: https://goo.gl/KEdrCB
- McGrady MG, Ellwood RP, Srisilapanan P, Korwanich N, Worthington HV et al. (2012) Dental fluorosis in populations from Chiang Mai, Thailand with different fluoride exposures – Paper1: assessing fluorosis risk, predictors of fluorosis and the potential role of food preparation. BMC Oral Health 12: 16. Link: https://goo.gl/rFfy3Q
- Dissanayake CB (1996) Water quality and dental health in the Dry Zone of Sri Lanka. Environ Geochem Health 113: 131–140. Link: https://goo.gl/jOsSYw
- Ayoob S, Gupta AK (2006) Fluoride in drinking water: a review on the status and stress effects. Crit Rev Environ Sci Technol 36: 433–487. Link: https://goo.gl/Wkh1Q6
- Susheela AK (2003) A treatise on fluorosis, revised second edition, Fluorosis Research and Rural Development Foundation, New Delhi, India, 13–14. Link: https://goo.gl/OIDxVj
- Jha SK, Nayak AK, Sharma YK (2010) Potential fluoride contamination in the drinking water of Marks Nagar, Unnao district, Uttar Pradesh, India. Environ Geochem Health 32: 217-226. Link: https://goo.gl/YKtBuC
- Shah MT, Danishwar S (2003) Potential fluoride contamination in the drinking water of Naranji area, Northwest Frontier Province, Pakistan. Environ Geochem Health 25: 475–481. Link: https://goo.gl/AOX0v8
- Choubisa SL (2012) Fluoride in drinking water and its toxicosis in tribals, Rajasthan, India. Proc Nat Acad Sci India Sect B Biol Sci 82: 325-330. Link: https://goo.gl/BTXpaO
- Choubisa SL (2015) Industrial fluorosis in domestic goats (Capra hircus), Rajasthan, India. Fluoride 48: 105-112. Link: https://goo.gl/lhOKN6
- Patel PD, Chinoy NJ (1998) Influence of fluoride on biological free radical reactions in ovary of mice and its reversal. Fluoride 31: S27.
- Eraslan G, Kanbur M, Silici S (2007) Evaluation of propolis effects on some biochemical parameters in rats treated with sodium fluoride. Pest Biochem Physiol 88: 273–283. Link: https://goo.gl/SXqLRo
- Bharti VK, Srivastava RS (2009) Fluoride-induced oxidative stress in rat’s brain and its amelioration by buffalo (Bubalus bubalis) pineal proteins and melatonin. Biol Trace Elem Res 130: 131–140. Link: https://goo.gl/BvH6mX
- Garcia-Montalvo EA, Reyes-Perez H, del Razo LM (2009) Fluoride exposure impairs glucose tolerance via decreased insulin expression and oxidative stress. Toxicol 263: 75–83. Link: https://goo.gl/J3vzdx
- Zhang M, Wang AG, He WH (2007) Effects of fluoride on the expression of NCAM, oxidative stress, and apoptosis in primary cultured hippocampal neurons. Toxicol 236: 208–216. Link: https://goo.gl/VmfRKA
- Ghosh J, Das J, Manna P (2008) Cytoprotective effect of arjunolic acid in response to sodium fluoride mediated oxidative stress and cell death via necrotic pathway. Toxicol in vitro 22: 1918–1926. Link: https://goo.gl/rnURqR
- Morgan KT, Ni H, Brown HR (2002) Application of cDNA microarray technology to in vitro toxicology and the selection of genes for a real-time RT-PCR-based screen for oxidative stress in Hep-G2 cells. Toxicol Pathol 30: 435-451. Link: https://goo.gl/371SMA
- Gao Q, Liu YJ, Guan ZZ (2008) Oxidative stress might be a mechanism connected with the decreased alpha 7 nicotinic receptor influenced by high-concentration of fluoride in SHSY5Y neuroblastoma cells. Toxicol In Vitro 22: 837-843. Link: https://goo.gl/qNC3zB
- Sireli M, Bulbul A (2004) The effect of acute fluoride poisoning on nitric oxide and methemoglobin formation in the Guinea pig. Turk J Vet Anim Sci 28: 591–595. Link: https://goo.gl/JOeKTE
- Izquierdo-Vega JA, Sanchez-Gutierrez M, del Razo LM (2008) Decreased in vitro fertility in male rats exposed to fluoride-induced oxidative stress damage and mitochondrial transmembrane potential loss. Toxicol Appl Pharmacol 230: 352–357. Link: https://goo.gl/vOX7PY
- Flora SJ, Mittal M, Mishra D (2009) Co-exposure to arsenic and fluoride on oxidative stress, glutathione linked enzymes, biogenic amines and DNA damage in mouse brain. J Neurol Sci 285: 198–205. Link: https://goo.gl/V9DhQj
- Shanthakumari D, Srinivasalu S, Subramanian S (2004) Effects of fluoride intoxication on lipidperoxidation and antioxidant status in experimental rats. Toxicol 204: 219–228. Link: https://goo.gl/nChBcL
- Chouhan S, Lomash V, Flora SJS (2010) Fluoride-induced changes in haem biosynthesis pathway, neurological variables and tissue histopathology of rats. J Appl Toxicol 30: 63–73. Link: https://goo.gl/f94rRP
- Aydin G, Cic E, Akdogan M, Gokalp O (2003) Histopathological and biochemical changes in lung tissues of rats following administration of fluoride over several generations. J Appl Toxicol 23: 437–446. Link: https://goo.gl/rtCTt3
- Liu G, Chai C, Cui L (2003) Fluoride causing abnormally elevated serum nitric oxide levels in chicks. Environ Toxicol Pharmacol 13: 199–204. Link: https://goo.gl/9U8h3r
- Hassan HA, Yousef MI (2009) Mitigating effects of antioxidant properties of black berry juice on sodium fluoride induced hepatotoxicity and oxidative stress in rats. Food Chem Toxicol 47: 2332–2337. Link: https://goo.gl/IibpcW
- Zhan XA, Wang M, Xu ZR (2006) Effects of fluoride on hepatic antioxidant system and transcription of Cu/Zn SOD gene in young pigs. J Trace Elem Med Biol 20: 83–87. Link: https://goo.gl/0tquA8
- Ghosh D, Das S, Maiti R, Jana D, Das U (2002) Testicular toxicity in sodium fluoride treated rats: association with oxidative stress. Reprod Toxicol 16: 385–390. Link: https://goo.gl/U0aMrL
- Guo XY, Sun GF, Sun YC (2003) Oxidative stress from fluoride induced hepatotoxicity in rats. Fluoride 36: 25–29. Link: https://goo.gl/yVLwIU
- Zhan XA, Xu ZR, Li JX (2005) Effects of fluorosis on lipid peroxidation and antioxidant systems in young pigs. Fluoride 38: 157–161. Link: https://goo.gl/rjuwIF
- Mc-Cord JM, Keele BB, Fridovich I (1984) An enzyme based theory of obligate anaerobiosis, the physiological functions of superoxide dismutase. Proc Natl Acad Sci 68: 1024–1027. Link: https://goo.gl/PyLXOU
- Guney M, Oral B, Demirin H (2007) Protective effects of vitamins C and E against endometrial damage and oxidative stress in fluoride intoxication. Clin Exp Pharmacol Physiol 34: 467–474. Link: https://goo.gl/sjbWZr
- Mittal M, Flora SJS (2007) Vitamin E protects oxidative stress and essential metal imbalance during concomitant exposure to arsenic and fluoride in male mice. Drug Chem Toxicol 30: 263–281. Link: https://goo.gl/VSqhiW
- Chouhan S, Flora SJS (2008) Effects of fluoride on the tissue oxidative stress and apoptosis in rats: biochemical assays supported by IR spectroscopy data. Toxicol 254: 61–67. Link: https://goo.gl/8AXH7T
- Jhala DD, Chinoy NJ, Rao MV (2008) Mitigating effects of some antidotes on fluoride and arsenic induced free radical toxicity in mice ovary. Food Chem Toxicol 46: 1138–1142. https://goo.gl/wXhFT8
- Kanbur M, Eraslan G, Silici S (2009) Effects of sodium fluoride exposure on some biochemical parameters in mice: evaluation of the ameliorative effect of royal jelly applications on these parameters. Food Chem Toxicol 47: 1184–1189. Link: https://goo.gl/kp99Fl
- Basha PM, Madhusudhan N (2010) Pre and post natal exposure of fluoride induced oxidative macromolecular alterations in developing central nervous system of rat and amelioration by antioxidants. Neurochem Res 35: 1017–1028. Link: https://goo.gl/r7ApN0
- Chen T, Cui Y, Gong T (2009) Inhibition of splenocyte proliferation and spleen growth in young chickens fed high fluoride diets. Fluoride 4: 203-209. Link: https://goo.gl/1ntzY3
- Shivarajashankara YM, Shivashankara AR, Gopalakrishna BP (2001) Oxidative stress in children with endemic skeletal fluorosis. Fluoride 34: 103–107. Link: https://goo.gl/3zlHEg
- Xu B, Xu Z, Xia T (2011) Effects of the Fas/Fas-L pathway on fluoride-induced apoptosis in SH-SY5Y cells. Environ Toxicol 26: 86-92. Link: https://goo.gl/EWa7SM
- Salgado-Bustamante M, Ortiz-Perez MD, Calderon-Aranda E (2010) Pattern of expression of apoptosis and inflammatory genes in humans exposed to arsenic and/or fluoride. Sci Total Environ 408: 760–767. Link: https://goo.gl/X9m6K5
- Qiao J, Cui Z, Sun Y, Hu Q, Guan X (2014) Simultaneous removal of arsenate and fluoride from water by Al-Fe (hydr)oxides. Frontiers Environ Sci Eng 8: 169-179. Link: https://goo.gl/MnQCgQ
- Chai L, Wang Y, Zhao N, Yang W, You X (2013) Sulfate-doped Fe3O4/Al2O3 nanoparticles as a novel adsorbent for fluoride removal from drinking water. Water Res 47: 4040-4049. Link: https://goo.gl/Zd3CKL
- Mondal NK, Bhaumik R, Baur T, Das B, Roy P et al. (2012) Studies on Defluoridation of Water by Tea Ash: An Unconventional Biosorbent. Chem Sci Trans 1: 239-256. Link: https://goo.gl/36J8xV
- Chakrabarty S, Patra PK (2013) Effect of sodium fluoride on seed germination, seedling growth and biochemistry of paddy (Oryza sativa L.). Asian J Exp Biol Sci 4: 540-544.
- Thole B, Mtalo F, Masamba W, (2012) Effect of particle size on loading capacity and water quality in water defluoridation with 200°C calcined bauxite, gypsum, magnesite and their composite filter. Afri J Pure Appl Chem 6: 26-34. Link: https://goo.gl/9aJIPJ
- Ghorai S, Pant KK (2005) Equilibrium, kinetics and breakthrough studies for adsorption of fluoride on activated alumina. Sep Purification Technol 42: 265– 271. Link: https://goo.gl/xTfLIc
- Tripathy SS, Bersillon JL, Gopal K (2006) Removal of fluoride from drinking water by adsorption onto alum-impregnated activated alumina. Sep Purific Technol 50: 310–317. Link: https://goo.gl/a523Fm
- Chauhan VS, Dwivedi PK, Iyengar L (2007) Investigations on activated alumina based domestic defluoridation units. J Hazardous Materials 139: 103–107. Link: https://goo.gl/ZRriyI
- Hernandez-Montoya V, Elizalde-Gonzalez MP, Trejo-Vazquez R (2007) Screening of commercial sorbents for removal of fluoride in synthetic and groundwater. Environ Technol 28: 595–607. Link: https://goo.gl/HfqhqK
- Daifullah AAM, Yakout SM, Elreefy SA (2007) Adsorption of fluoride in aqueous solutions using KMnO4-modified activated carbon derived from steam pyrolysis of rice straw. J Hazardous Materials 147: 633–643. Link: https://goo.gl/yDCcbH
- Kumar S, Gupta A, Yadav JP (2007) Fluoride removal by mixtures of activated carbon prepared from Neem (Azadirachta indica) and Kikar (Acacia arabica) leaves. Indian J Chem Technol 14: 355–361. Link: https://goo.gl/A7dkPD
- Bashir MT, Salmiaton A, Nourouzi MM, Azni I, Harun R (2015) Fluoride removal by chemical modification of palm kernel shell-based adsorbent: a novel agricultural waste utilization approach. Asian J Microbiol Biotech Env Sci 17: 533-542. Link: https://goo.gl/w1I91L
- Vardhan C, Karthikeyan J (2011) Removal of fluoride from water using low-cost materials. In: Fifteenth International Water Technology Conference. [Online] Alexandria, Egypt. Link: https://goo.gl/1ghK4y
- Wartelle L, Marshall W (2006) Quaternized agricultural by-products as anion exchange resins. J Environ Manage 78: 157-162. Link: https://goo.gl/2s3mco
- Shen F, Chen X, Ping G, Chen G (2003) Electrochemical removal of fluoride ions from industrial wastewater. Chem Eng Sci 58: 987– 993. Link: https://goo.gl/pV6s1u
- Pinon-Miramontes M, Bautista-Margulis RG, Perez-Hernandez A (2003) Removal of arsenic and fluoride from drinking water with cake alum and a polymeric anionic flocculent. Fluoride 36: 122–128. Link: https://goo.gl/rjlp1o
- Tahaikt M, Achary I, Menkouchi SMA, Amor Z, Taky M et al. (2006) Defluoridation of Moroccan groundwater by electrodialysis: continuous operation. Desalination 189: 215– 220. Link: https://goo.gl/Bc77Ae
- Arora M, Maheshwari RC, Jain SK, Gupta A (2004) Use of membrane technology for potable water production. Desalination. 170: 105–112. Link: https://goo.gl/Azw8Qj
- Hu K, Dickson JM (2006) Nanofiltration membrane performance on fluoride removal from water. Membrane Sci 279: 529–538. Link: https://goo.gl/t2QzrH
- Qianhai Zuo, Xueming Chen, Wei Li, Guohua Chen (2008) Combined electrocoagulation and electroflotation for removal of fluoride from drinking water. J Hazardous Materials 159: 452–457. Link: https://goo.gl/BBSf7i
- Hassan HA, Abdel-Aziz AF (2010) Evaluation of free radical-scavenging and anti-oxidant properties of blackberry against fluoride toxicity in rats. Food Chem Toxicol 48: 1999–2004. Link: https://goo.gl/pXybJx
- Basha PM, Saumya SM (2013) Suppression of Mitochondrial Oxidative Phosphorylation and TCA Enzymes in Discrete Brain Regions of Mice Exposed to High Fluoride: Amelioration by Panax ginseng (Ginseng) and Lagerstroemia speciosa (Banaba) Extracts. Cell Mol Neurobiol 33: 453–464. Link: https://goo.gl/J7vuxH
- Nabavi SF, Habtemariam S, Sureda A, Akbar HM, Daglia M, et al. (2013) In vivo protective effects of gallic acid isolated from peltiphyllum peltatum against sodium fluoride-induced oxidative stress in rat erythrocytes. Arh Hig Rada Toksikol 64: 553-559. Link: https://goo.gl/xBd4BC
- Sinha M, Manna P, Sil PC (2008) Terminalia arjuna Protects Mouse Hearts Against Sodium Fluoride-Induced Oxidative Stress. J Med Food 11: 733–740. Link: https://goo.gl/8aU6DN
- Vasant RA, Narasimhacharya AVRL (2012) Ameliorative effect of tamarind leaf on fluoride-induced metabolic alterations. Environ Health Prev Med 17: 484–493. Link: https://goo.gl/6koPxk
- Chawla SL, Yadav R, Shah D, Rao MV (2008) Protective action of melatonin against fluoride induced hepatotoxicity in adult female mice. Fluoride 41: 44-51. Link: https://goo.gl/QqxdDn
- Jain A, Mehta VK, Chittora R, Mahdi AA, Bhatnagar M (2015) Melatonin ameliorates fluoride induced neurotoxicity in young rats: an in vivo evidence. Asian J Pharm Clin Res 8: 164-167. Link: https://goo.gl/HJElEk
- Bharti VK, Srivastava RS, Kumar H, Bag S, Majumdar AC, et al. (2014) Effects of melatonin and epiphyseal proteins on fluoride-induced adverse changes in antioxidant status of heart, liver, and kidney of rats. Adv Pharmacol Sci 2014:532969. Link: https://goo.gl/IESXbF
- Bharti VK, Srivastava RS (2011a) Effects of epiphyseal proteins and melatonin on the blood biochemical parameters of fluoride-intoxicated rats. Neurophysiol 42: 258-264. Link: https://goo.gl/5pgiO3
- Rao MV, Chawla SL, Patel N (2009) Melatonin reduction of fluoride-induced nephrotoxicity in mice. Fluoride 42: 110–116. Link: https://goo.gl/BtLwwO
- Bharti VK, Srivastava RS (2011b) Effect of pineal proteins and melatonin on certain biochemical parameters of rats exposed to high-fluoride drinking water. Fluoride 44: 30–36. Link: https://goo.gl/kwE7ZU
- Chawla SL, Rao MV (2012) Protective effect of melatonin against fluoride induced oxidative stress in the mouse ovary. Fluoride 45: 125–132. Link: https://goo.gl/7nWbhZ
- Rao MV, Bhatt RN (2012) Melatonin protection against F-induced oxidative stress and testicular dysfunction in rats. Fluoride 45: 116-124. Link: https://goo.gl/NICNbv
- Rao MV, Vyas DD, Meda RB, Chawla SL (2011) In vitro protective role of melatonin against hemolysis induced by sodium fluoride in human red blood cells. Fluoride 44: 77–82. Link: https://goo.gl/b5WUTb
- Bharti VK, Srivastava RS, Anand AK, Kusum K (2012) Buffalo (Bubalus bubalis) epiphyseal proteins give protection from arsenic and fluoride-induced adverse changes in acetylcholinesterase activity in rats. J Biochem Mol Toxicol 26: 10-15. Link: https://goo.gl/Ofll3W
- Bharti VK, Srivastava RS (2011c) Effect of pineal proteins at different dose level on fluoride-induced changes in plasma biochemicals and blood antioxidants enzymes in rats. Biol Trace Elem Res141: 275-282. Link: https://goo.gl/8eDjlV
- Mansour HH, Tawfik SS (2012) Efficacy of lycopene against fluoride toxicity in rats. Pharm Biol 50: 707–711. Link: https://goo.gl/z2hgHD
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
