Archives of Community Medicine and Public Health
Primary undifferentiated pleomorphic sarcoma of the jejunal mesentery with a complicated course: A case report
Jochen Jure* and Marc E Vuylsteke
Cite this as
Jure J, Vuylsteke ME (2022) Primary undifferentiated pleomorphic sarcoma of the jejunal mesentery with a complicated course: A case report. Arch Community Med Public Health 8(2): 052-054. DOI: 10.17352/2455-5479.000173Copyright License
© 2022 Jure J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.A primary Undifferentiated Pleomorphic Sarcoma (UPS) of the mesentery is a rare form of malignancy, most commonly usually affecting the soft tissues of the extremities. Due to their location in the abdominal cavity distant from the intestinal loops they remain asymptomatic for a long time. In this study, an incidental finding on imaging is presented in a 66-year-old woman. CT imaging revealed a large mass in her abdomen, with difficulty pinpointing its exact origin. Exploratory laparotomy was performed to remove the mass in its entirety. Examination of the mass showed findings consistent with an undifferentiated pleomorphic sarcoma. She recovered well after her surgery. However, on follow-up imaging, a solitary metastatic lesion in her lung was detected. The prognosis of UPS is historically dismal with invasive treatment options currently being the preferred methods for management.
Introduction
Undifferentiated Pleomorphic Sarcoma (UPS) is a common malignancy affecting the soft tissue of the extremities in adults. However, intestinal mesentery is very rarely affected and only a few cases have been described. We report the case of an adult woman with an incidental finding of UPS of the jejunal mesentery who rapidly developed solitary lung metastasis.
Case presentation
We present the case of a 66-year-old woman who presented to our hospital in March of 2021 due to intermittent heavy sensations of chest pain in the last three months, mostly occurring at night, located over the anterior aspect of the ribs on her right side. She couldn’t recall any inciting trauma. Further review of systems was unremarkable. She reported no fever, chills, weight loss, fatigue, or change in bowel habits. Her past medical history included hypercholesterolemia and recurring respiratory tract infections associated with IgG2 deficiency. Her medication regimen consisted of a combination preparation of atorvastatin and ezetimibe once daily. She did not smoke, drink alcohol, or use illicit drugs. Vital signs were within normal limits. On physical examination, the pain she reported was reproducible by applying pressure over the right anterior ribs. Head, neck, cardiopulmonary, abdominal, and skin examinations were normal. There were no prominent abdominal masses or organomegaly, and examination of the lymph nodes was unremarkable. Laboratory analysis revealed an inflammatory pattern. Erythrocyte sedimentation rate was elevated at 118mm (normal range, <30mm) and CRP at 41.8mg/l (normal range, <5 mg/l). Serum iron level was 42µg/dl (normal range, 50 to 150), the ferritin level was 367µg/l (normal range, 13 to 150). Liver function tests were mildly elevated, alanine transaminase was 64U/l (normal range, 10 to 35) and aspartate transaminase was 33 (normal range, 10 to 35), alkaline phosphatase was 271U/l (normal range, 35 to 105) and gamma-glutamyltransferase was 175 U/l (normal range, < 35). Further complete blood count, serum electrolyte levels, renal function tests, thyroid function tests, and uric acid levels were within normal limits. On serum protein electrophoresis a discrete band of gamma globulins was noted. Rheumatological and autoimmunity panels were negative. Ultrasound examination could not visualize any abnormalities at the abdominal wall or rib cage but did reveal a liver hemangioma, for which further investigation with Computed Tomography (CT) scanning was planned. Due to the clinical picture and inflammatory markers the diagnosis of costochondritis was made.
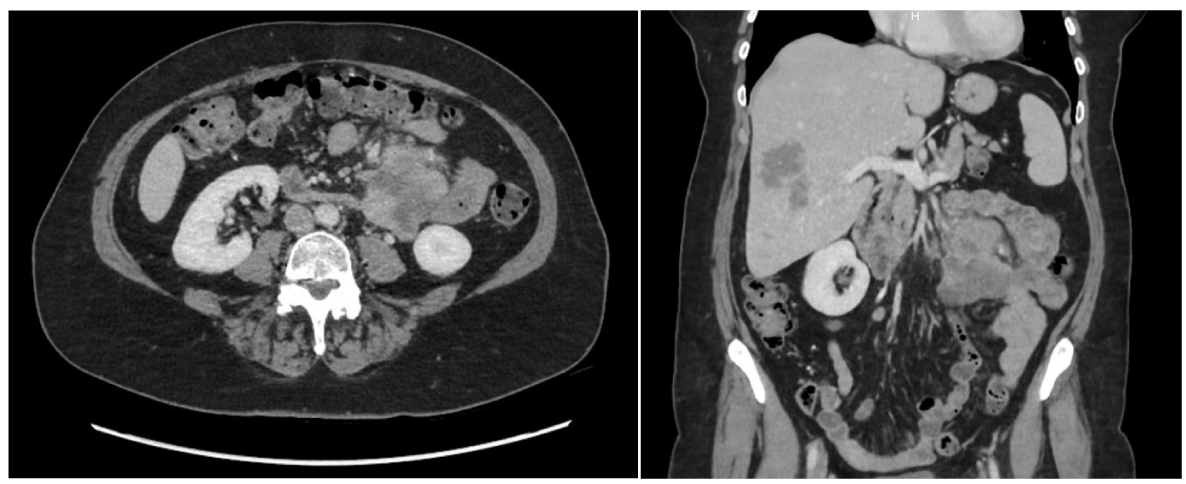
CT scanning of the abdomen confirmed the presence of multiple liver hemangiomas, consistent and comparable with previous imaging performed in 2007. A new finding however was the presence of a hypodense lobulated mass with enhancing walls in the left upper quadrant measuring 5cm by 6.4cm by 6.9cm, surrounded by several prominent lymph nodes (Figure 1). It was unclear whether the mass originated from the small intestine or not. No other abnormalities were detected. The differential diagnosis included intestinal adenocarcinoma, gastrointestinal stromal tumor, or carcinoid tumor.
Exploratory laparotomy was indicated and revealed an encapsulated, irregular mass of 10cm by 7cm by 6cm in size, arising from the mesentery of the first jejunal loop. There was no adherence of the tumor to the intestinal wall. The entirety of the mass with its surrounding mesentery and 28cm of jejunum was resected. An end-to-end anastomosis connecting the distal duodenum and proximal jejunum was created. There were no ascites or other visible metastatic nodes in the pelvis, liver, or omentum.
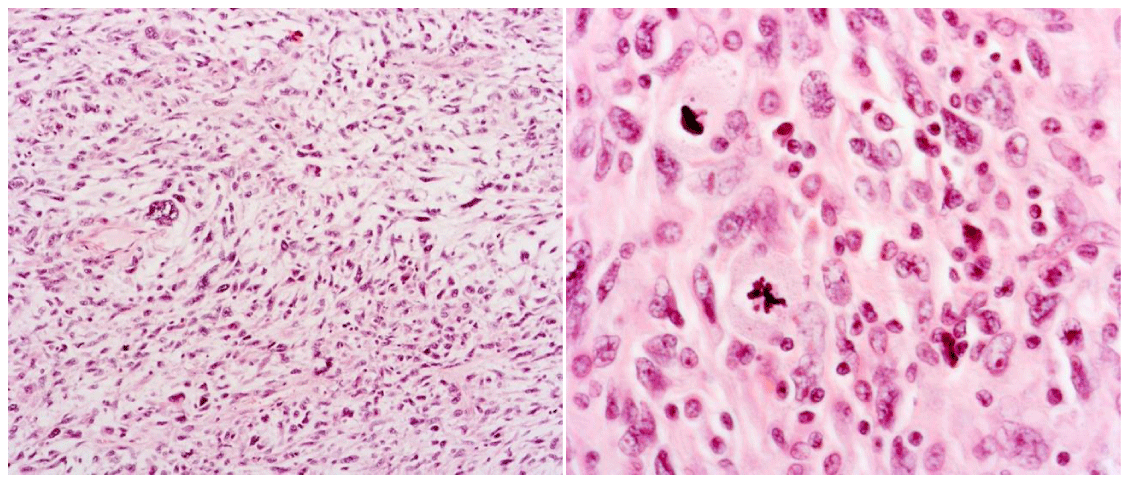
Initial microscopic examination of the specimen in the mesentery revealed a tumor consisting of epithelioid cells displaying an abundant eosinophilic cytoplasm. The cell nuclei were enlarged, and some cells had multiple nuclei (Figure 2). There was marked mitosis observable, along with some atypical mitotic figures. Local necrosis was present, as well as local inflammatory infiltrates, rich in lymphocytes and eosinophilic polynuclear cells. Local biopsies of the intestinal wall itself were unremarkable. The eleven lymph nodes that were resected along with the mesentery all showed no presence of a tumor. Further immunohistochemical staining was performed and was unable to show any differentiation. The tumor cells stained positive for vimentin. There was no expression of S100, SOX10, CD45, EMA, CK20, SMA, cytokeratin 7, desmin, MDM2, CD3, CD5, CD20, CD117, CD34, CD21. There was retained SMARCB1 expression. These results were most consistent with the diagnosis of an Undifferentiated Pleomorphic Sarcoma (UPS), grade III, according to the Federation Nationale des Centres de Lutte Contre le Cancer (FNCLCC).
Further investigations at the time of surgery consisted of a colonoscopy, mammography, dermatological evaluation, and CT scanning of the thorax. All of which were unremarkable. Follow-up imaging was scheduled every three months.
CT imaging of the thorax performed at the first follow-up visit three months after initial discharge showed the presence of a peripheral poly lobular lung nodule in the left lower lobe, measuring 14.6mm by 10mm (Figure 3). No lymphadenopathies or other remarkable findings were noted. As this finding was most consistent with a solitary metastasis in an easily accessible location, Video-Assisted Thoracoscopic Surgery (VATS) was proposed with diagnostic and therapeutic intent. During the procedure, the lesion was macroscopically visible, and a wedge excision was performed. Gross pathological examination showed the presence of a white round tumoral lesion measuring 15mm in diameter. The microscopic investigation confirmed the presence of a metastatic lesion consistent with undifferentiated pleomorphic sarcoma that was completely removed. Further follow-up is identical and consists of imaging studies every three months.
Discussion
While the mesentery of the small bowel is a relatively rare location of origin for a primary malignancy, it does provide plenty of pathways for the spread of more distant malignancies. It is the main provider of blood and lymphatic flow to the intestine and is susceptible to metastatic spreading, by its location and connection to the peritoneal spaces and retroperitoneum. Tumors arising from the mesentery can present with a variety of symptoms depending on their location. When they are of sufficient size or in close enough proximity to the bowels, they can cause symptoms ranging from vague abdominal pain, presence of an abdominal mass, weight loss, or a change in bowel habits, or can go as far as to cause bowel obstruction or even perforation. On the other hand, they can be asymptomatic, as was the case in the patient described in this report [1].
With the mesentery already being a rather rare location for a primary malignancy, the multitude of different cellular lines present makes for a wide array of varying possibilities where malignant growth can originate from. Besides vascular and lymphatic structures, the mesentery is home to a large amount of connective tissue and fatty tissue as well (Coffey 2016). The most common primary mesenteric masses are lymphatic in origin, with the most common solid mesenteric mass being lymphoma. Other possibilities that should be included in the differential diagnosis are among other things cysts, desmoid tumors, gastrointestinal stromal tumors, or sclerosing mesenteritis [2].
The mass that was found in our case report was identified as an Undifferentiated Pleomorphic Sarcoma (UPS). This term includes malignancies that were described as malignant fibrous histiocytomas (MFH) in the past. It is used to describe tumors in which all recognizable lines of differentiation have been excluded. They are typically high grade, show a wide range of morphological features, and are often associated with a poor prognosis [3]. They are a rare entity affecting both genders, typically in the sixth and seventh decades of life. The primary location of origin is mostly the deep soft tissues of the extremities, preferably the lower limb, and to a lesser extent the trunk and subcutaneous tissues [4]. They tend to grow rapidly and have a metastatic rate of approximately 50%, with 5% of patients already showing distant metastasis at the time of presentation [5]. The diagnosis is one of exclusion and is made after extensive microscopic sampling and immunohistochemical staining. Often there is a very heterogeneous pattern of staining in the case of Cytokeratin. microscopically, showing a fascicular, storiform, or sheet-like appearance, and they are often composed of an admixture of spindle cells and pleomorphic, sometimes multinucleated cells, with eosinophilic or basophilic cytoplasm. Immunohistochemical staining for S100 and SOX10 is done to exclude metastatic melanoma, and for SMA and desmin to exclude pleomorphic myogenic sarcomas. Usually, these lesions show positive for vimentin, as was the case in our report, although none of these findings is truly specific [5,6].
As mentioned before, a UPS arising from the mesentery is a very rare lesion. While a tumor originating in the soft tissues of the extremities may rapidly become symptomatic, the location of the mesentery and its distance to the bowel can make for a long asymptomatic course, even with its potential for rapid growth. In our case report, the tumor had already reached a significant size and was still an incidental finding. The treatment of any UPS should include complete surgical resection with negative resection margins. While radiotherapy has been proven to reduce the rate of local recurrence and metastasis in UPS of the extremities, its role is unclear in other locations. There have been studies that investigated the usage of chemotherapy in UPS, more specifically the use of doxorubicin and ifosfamide, but their role has not yet been properly defined and usage of them is usually reserved for unresectable tumors or significant metastatic disease. The prognosis of UPS, in general, is rather poor, with a 5-year survival rate after surgery of 67.2%, while the 5-year survival rate of abdominal UPS is 14%. The survival rates for UPS arising from the mesentery are unclear owing to the very low numbers reported in the literature, although the aggressive potential of the tumor is shown in our case with a lung metastasis arising within three months after complete surgical resection. We approached this solitary lung metastasis based on the management of other metastatic pulmonary diseases and resected the affected part of the lung [7-9].
Conclusion
We present a case of a rare primary UPS of the jejunal mesentery that was treated by complete ileal resection. Within three months the mass had metastasized to the lung and was consequently treated by VATS through which a wedge excision was performed of the affected location.
- Sheth S, Horton KM, Garland MR, Fishman EK (2000) Mesenteric neoplasms: CT appearances of primary and secondary tumors and differential diagnosis. Radiogr Rev Publ Radiol Soc North Am Inc 23: 456-457.
- Dufay C, Abdelli A, Le Pennec V, Chiche L (2012) Mesenteric tumors: diagnosis and treatment. J Visc Surg 149: e239-e251. Link: https://bit.ly/3iW8jf3
- Jo VY, Fletcher CDM (2014) WHO classification of soft tissue tumours: an update based on the 2013 (4th) edition. Pathology. 46: 95-104.Link: https://bit.ly/3wXizfy
- Nascimento AF, Raut CP (2008) Diagnosis and management of pleomorphic sarcomas (so-called “MFH”) in adults. J Surg Oncol 97: 330-339.Link: https://bit.ly/3uIf0qN
- Hornick JL (2018) Subclassification of pleomorphic sarcomas: How and why should we care? Ann Diagn Pathol 37: 118-124. Link: https://bit.ly/3wS8uAo
- Dei Tos AP (2006) Classification of pleomorphic sarcomas: where are we now? Histopathology 48: 51–62. Link: https://bit.ly/375b6Qo
- Vodanovich DA, Spelman T, May D, Slavin J, Choong PFM (2019) Predicting the prognosis of undifferentiated pleomorphic soft tissue sarcoma: a 20-year experience of 266 cases. ANZ J Surg 89: 1045–1050. Link: https://bit.ly/35zCBS0
- Lee JH, Kang DB, Park WC (2019) Primary Undifferentiated Pleomorphic Sarcoma of the Colon Mesentery. Ann Coloproctol 35: 152-154. Link: https://bit.ly/3DrRceJ
- Nakayama Y, Minagawa N, Torigoe T, Yamaguchi K (2011) Malignant fibrous histiocytoma originating from the mesorectum: a case report. World J Surg Oncol 9: 15. Link: https://bit.ly/35x7ars
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
