Archives of Clinical Hypertension
Alcoholism, Fibroblast Growth Factor 23 and Cardiovascular Risk
Emilio González-Reimers*, Geraldine Quintero-Platt, Candelaria Martín-González, Lucía Romero-Acevedo, Daniel Martínez-Martínez, Melchor Rodríguez-Gaspar and Francisco Santolaria-Fernández
Cite this as
González-Reimers E, Quintero-Platt G, Martín-González C, Romero-Acevedo L, Martínez-Martínez D, et al. (2017) Alcoholism, Fibroblast Growth Factor 23 and Cardiovascular Risk. Arch Clin Hypertens 3(1): 010-015. DOI: 10.17352/ach.000013Background: Bone metabolism is tightly regulated by several hormones that are synthesized in bone cells and that have effects not only on bone but on several distant organs. These hormones are involved in intermediate metabolism and modulate fatty acid transport, playing a role in insulin resistance, liver steatosis, atherosclerosis and cardiovascular risk.
Aims: We review the association of fibroblast growth factor-23 (FGF-23) and α-Klotho with cardiovascular risk, especially in the alcoholic patient.
Results: High levels of FGF-23 are associated with vascular risk, especially with vascular calcifications, hypertension, or left ventricular hypertrophy.
Main Findings: Hyperphosphatemia constitutes a major stimulus for FGF-23 secretion, together with inflammation and reduced iron availability, either by iron deficiency or by iron sequestration in inflammatory diseases. FGF-23 can be expressed by the damaged liver. Alcoholism is a proinflammatory condition, and in alcoholics there is an increased cardiovascular risk. Preliminary data suggest that FGF-23 is raised in alcoholics.
Conclusion: Increased FGF-23 levels have been described in association with hypertension, arterial wall calcification, left ventricular hypertrophy and increased cardiovascular mortality, both in patients with or without chronic kidney disease. Some data suggest that FGF-23 is also related to increased vascular risk in alcoholics.
Brief Summary: FGF-23 is an osteocyte derived molecule related to cardiovascular risk. Hyperphosphatemia is a major trigger mechanism for its secretion, but inflammatory conditions are also associated with increased FGF-23 production. Some data suggest that FGF-23 may be also related with the increased vascular risk observed in chronic alcoholic patients.
Background
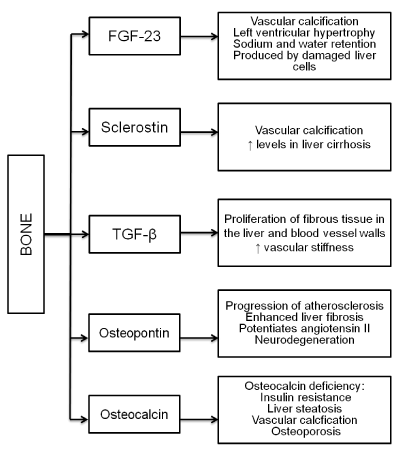
Bone is a metabolically active organ, in which synthesis and resorption are coupled in order to adapt its structure and mechanical properties to load changes and to provide calcium, phosphorus, and alkaline salts to the rest of the body. This process is tightly regulated by several hormones, mainly synthesized by bone cells or by liver, kidneys or parathyroid glands. In contrast with the classic perception of bone only as a reservoir of calcium, phosphorus and alkaline buffers, in the last decades it has become increasingly evident that bone is an endocrine organ, capable of exerting diverse functions in distant structures such as liver, adipocyte, muscle, kidneys, heart or brain [1]. Bone-derived hormones are involved in intermediate metabolism and thus modulate fatty acid transport, playing a role in adiposity, insulin resistance, liver steatosis and/or steatohepatitis, and cardiovascular risk [2,3]. The discovery of the transformation of vascular smooth muscle cells into bone-forming cells, governed by the same hormones that are involved in bone remodeling, has aided in our understanding of all these effects [4]. Indeed, major bone-derived hormones play an outstanding role in atherosclerosis, as depicted in figure 1.
In the past years, an osteocyte derived growth factor, fibroblast growth factor-23 (FGF-23) initially described as a phosphatonin, has been subjected to intensive research, mainly because of its association with hypertension and cardiovascular risk. Very recently, some studies also point out that osteocytes increase FGF-23 synthesis in inflammatory conditions, so it can be considered a marker of inflammation. Also, it has been shown that FGF-23 can be produced by the damaged liver. The purpose of this study is to provide an overview about the association of this molecule with hypertension and cardiovascular risk, and to discuss its potential role in vascular complications related to alcoholism, both because of the smoldering systemic inflammation caused by alcoholism, and because of the eventual liver damage exerted by chronic ethanol consumption.
FGF-23-Α Klotho Axis
In 2000, Yamashita et al isolated the Fibroblast growth factor (FGF)-23, that was expressed in the brain and thymus of adult mice [5]. In the same year a form of autosomal dominant hypophosphatemic rickets (ADHR) was described that was characterized by a mutation in the FGF-23 gene [6].
Later it was shown that osteoblasts and osteocytes are the main source of circulating FGF-23 in vivo. The secretion of FGF-23 by bone is stimulated by vitamin D and by extracellular phosphate, calcium and PTH [7,8].
Serum phosphate levels are finely regulated by hormonal signals. Transepithelial transport of phosphate in the intestine and in the kidney is carried out by sodium/phosphate cotransporters (NaPi-2a, NaPi-2b, NaPi-2c). Several hormonal factors such as parathyroid hormone (PTH), active metabolites of vitamin D and FGF-23 can control the activity of these cotransporters [9].
In 2004 Shimada et al demonstrated that FGF-23 is a potent regulator of vitamin D and phosphate metabolism. After administration of recombinant FGF-23 to parathyroidectomized animals there was a reduction in serum levels of phosphate and calcitriol. The reduction in serum phosphate was associated with a decrease in renal mRNA of the cotransporter NaPi-2a. They found no increase in PTH levels throughout the experiment. FGF-23 also decreased renal mRNA for 1α hydroxylase and increased mRNA for 24-hydroxylase [10]. The direct down-regulation effect of FGF-23 on the membrane expression the sodium-phosphate cotransporter NaPi-2a in the proximal tubule was also reported by Andrukhova et al [11]. The effect on 1α hydroxylase also affects monocytes, impairing immune response [12]. Despite the phosphaturic effect of FGF-23, it inhibits parathyroid hormone (PTH) secretion [13] , although the underlying mechanisms are still unclear [14].
It has been recently shown that another mechanism involved in FGF-23 synthesis is iron deficiency or the so called “functional iron deficiency”, i.e., iron sequestration in inflammatory setting [15], probably via HIF induction of the FGF-23 gene transcription. Interestingly, induction of FGF-23 synthesis by acute inflammation is accompanied by a parallel increase in the cleavaging ability of the osteocyte, leading to increased levels of carboxi- terminal FGF-23 (c-FGF-23), and normal or near normal serum levels of intact FGF-23 (i-FGF-23). By contrast, in chronic inflammation, both products are increased, probably because the cleavage capacity of the osteocyte becomes overwhelmed by continuous FGF-23 overproduction [16,17].
The metabolic effects of FGF-23 require binding of this molecule to a receptor complex that includes FGF receptors (FGFR) and the transmembrane protein α Klotho. There are 4 types of FGFR (FGFR1, 2, 3 y 4) but it is believed that FGF-23 acts through a receptor complex that includes FGFR-1c and α Klotho (although α Klotho can also bind to FGFR3 and 4). Signaling using the FGF-R1 receptor seems to be involved in the regulation of FGF-23 secretion in osteoblasts and osteocytes but the intracellular pathways that regulate this process are still unknown [14].
It seems clear that α Klotho functions as a co-receptor for FGF-23 and increases FGFR’s affinity for FGF-23 20-fold [14]. α Klotho is a transmembrane protein that serves as a coreceptor for FGF-23, strongly increasing the affinity of this molecule for the FGF receptors [14,18]. The main sites for α Klotho expression are choroid plexus in the central nervous system, parathyroid glands, and proximal and distal renal tubules [14].
α Klotho exhibits an extracellular domain that is cleaved, generating the soluble form of α Klotho [19], that can be measured in blood, urine and cerebrospinal fluid. The function of α Klotho is a controversial issue .It was originally described as an anti-aging factor. In 1997, Kuro-o et al described a defect in the expression of the α klotho gene in mice. These mice developed a syndrome similar to aging in humans which included shortened lifespan, infertility, atherosclerosis, skin atrophy, osteoporosis and emphysema [20]. However, after the ablation of vitamin D signaling using mice that lacked a vitamin D receptor (VDR) this phenotype disappeared [21]. Mice with α Klotho and FGF-23 deficits produce excessive amounts of vitamin D because there is no suppressive effect of FGF-23 on the activity of renal 1 α hydroxylase activity. Because of this, it is believed that the early aging phenotype seen in mice without α Klotho and FGF-23 is due to vitamin D intoxication that leads to severe hypercalcemia and hyperphosphatemia with organ dysfunction [14].
Although α Klotho deficiency has been associated with early aging, atherosclerosis, and vascular calcifications, other authors have described a paradoxical increase in α Klotho levels in situations associated with oxidative stress. Nakanishi et al studied α Klotho levels in men and they found higher soluble α Klotho and IL-6 levels in smokers than in subjects who had never smoked. They conclude that the increase in soluble α Klotho levels is a compensatory response to stress and that α Klotho acts as an anti-inflammatory molecule [22]. The recent discovery that inflammation results in an increased FGF-23 secretion adds a new interpretation to raised α Klotho in situations of oxidative stress.
FGF-23 is heavily involved in bone metabolism [9], given its close relationship with PTH, vitamin D, calcium and phosphorus. The relationships between FGF-23/α Klotho levels and bone mineral density (BMD) have been analysed in several studies, but findings show either a weak relationship with BMD [23,24] or no relationship at all [25]. On the contrary, several studies have reported close relationships between FGF-23 and vascular risk, as commented later.
Effects of FGF-23/α Klotho on the cardiovascular system
FGF-23 levels are directly related to the progression of chronic kidney disease, left ventricular hypertrophy, vascular calcification, and increased mortality in patients with chronic renal disease [14]. Gutiérrez et al found that high levels of FGF-23 at the start of hemodialysis were independently associated with mortality [26]. Faul et al found that high FGF-23 levels in patients with chronic kidney disease were associated with a lower ejection fraction, high left ventricular mass index, and a higher prevalence of left ventricular hypertrophy [27]. In addition, it has been described that FGF-23 is an independent risk factor for all-cause mortality and cardiovascular mortality in patients with normal renal function who were subjected to coronary angiography [28]. Other researchers have reported higher FGF-23 levels in patients with an obesity, especially visceral fat accumulation [29], suggesting an indirect relationship with cardiovascular risk in patients with intact renal function. Aditionally, it was shown that FGF-23 is a marker of frailty among community-dwelling individuals [30]. Also, FGF-23 is related with arterial stiffness in diabetic patients, independently on estimated glomerular filtration rate [31]. Therefore, the relationship of FGF with cardiovascular risk extends far beyond chronic kidney failure.
FGF-23 can indirectly promote cardiovascular and renal disease by contributing to sodium and water retention. Andrukhova et al., administered recombinant FGF-23 to mice and they found an increase in the reabsorption of sodium in the distal tubule with subsequent volume expansion, hypertension and cardiac hypertrophy that was dependent on the amount of sodium administered through diet. Chlorothiazide, an inhibitor of sodium reabsorption, prevented volume expansion mediated by FGF-23 and cardiac hypertrophy [11]. However, other authors have failed to find any relation between FGF-23 and volume changes, at least in patients with diabetic nephropathy [32]. FGF-23 levels did not change either after saline fluid overload or after treatment with hydrochlorotiazide, low sodium diet and angiotensin converting enzyme inhibitors. Furthermore, Grabner et al demonstrated that binding of FGF-23 to its receptor FGFR4 in cardiomyocytes induces myocyte hypertrophy (via phospholipase C and calcineurin). An antibody that blocks the binding of FGF-23 to FGFR4 inhibits cardiac hypertrophy induced by FGF-23 in rats with chronic kidney disease [33]. Therefore, cardiac hypertrophy is not necessarily dependent upon the existence of hypertension.
It is not yet known whether FGF-23 can directly affect vascular cells. Scialla et al found no association between FGF-23 levels and vascular calcification in coronary arteries and in the thoracic aorta assessed using computed tomography. They also studied human and mouse vascular smooth muscle and found that FGF-23 had no effect on phosphate induced calcification, not even in the presence of soluble α Klotho [34]. However, Jimbo et al showed that FGF-23 increased phosphate-induced vascular calcification in aortic rings of uremic rats [35]. It has been proposed that FGF-23 and α Klotho are not directly involved in the pathogenesis of vascular calcification; on the contrary, increased levels of both proteins would be due to a compensatory mechanism [36]. FGF-23 would rise in order to counteract increased deposition of calcium phosphate salts.
As was described earlier, an antiaging effect has been described for α Klotho and in this sense it has been proposed that this molecule might have a cardioprotective effect. However, no association was found between soluble α Klotho levels and vascular risk in patients with chronic kidney disease in a recent cohort study [37].
FGF-23 and Alcoholic liver disease
Although FGF-23 has been primarily linked to bone metabolism, recent studies show an association with liver function. Nafidi et al., (2009) reported that partial hepatectomy led to increased FGF-23 levels [38], and later, Raimann et al., (2013) showed that liver tissue could express mRNA for FGF-23 [39]. In the same year Prie et al found that high FGF-23 levels predict the risk of death in patients with end stage liver disease on a liver transplant waiting list [40]. In these patients, high FGF-23 levels were not explained by decreased renal function. They also found in an experimental model that the damaged liver of diethyl-nitrosamine-treated mice expressed FGF-23 mRNA whereas healthy livers of control mice did not [40]. Additionally, in a more recent study by Wasserman et al high FGF-23 levels were described in 2 infants with liver disease due to biliary atresia who had developed a form of hypophosphatemic rickets. They also found that the damaged liver overexpressed FGF-23 [41]. Therefore, altered FGF-23 levels may be associated with liver disease. The link between FGF-23 and the bone-liver-vascular axis has not yet been completely elucidated.
Similar observations have been performed, logically, with α Klotho. In the mentioned study by Raimann et al [39], these authors found that piglets´ liver may express α Klotho, and Chen et al., (2013) also report that human hepatoma cells may express α Klotho. Staining positivity for α Klotho was also associated with the presence of liver cirrhosis [42]. This is in contrast with the observations of Lim et al, who, in 2015, described expression of transmembrane α Klotho in many tissues, but not in the liver. However, they did not study diseased liver [43]. It is possible that, in a way similar to FGF-23, only diseased liver expresses alpha Klotho.
Altered iron homeostasis in liver disease, especially among alcoholics [44], might also explain increased FGF-23 in this disease. As mentioned before, sequestered iron, via HIF, may trigger an excessive production of FGF-23 by the hepatocyte. Indeed, alcoholism is a proinflammatory condition, especially if the liver is involved [45]. Ethanol increases intestinal permeability, leading to increased leakage of Gram negative (and also, to a lesser extent, Gram positive bacteria) from intestine to portal blood [46]. These bacteria activate Kupffer cells [47], leading to increased secretion of proinflammatory cytokines. In addition, the diseased liver, especially if there are portosystemic shunts, as is the case of liver cirrhosis, cannot achieve an adequate defence against the increased portal endotoxemia [48]. Systemic endotoxemia leads to a generalized immune response, and therefore, to a generalized proinflammatory situation [49]. Whether or not this suffices to activate osteocyte-mediated FGF-23 secretion, and/or the damaged liver can also directly synthesize FGF-23 is not known. In a preliminary report we have observed increased FGF-23 levels in a group of alcoholics, especially in cirrhotics [50].
Ethanol consumption may lead to hypertension. It has been known since several decades that the association of ethanol consumption and hypertension adapts to a U-shaped curve [51], although the cutoff point for a “safe” drinking limit regarding hypertension has not been defined. An Australian study showed that daily consumption of 3 beers was associated with a prevalence of hypertension of 10.4% vs only 2.6% among teetotalers [52]. In the same sense, the INTERSALT study revealed that consumption of ethanol in amount greater than 500 ml during one week was associated with an increase of systolic blood pressure of 4.6 mm, and of 3 mm in diastolic one [53]. Indeed, some authors do not find that the relationship between hypertension and ethanol consumption adapts to a J- or U-shaped curve, but to a linear one [54]. Different studies have also pointed out that ethanol consumption –especially in the form of distilled beverages- is associated with left ventricular hypertrophy [55] , a finding that has been interpreted as a compensatory mechanism of the heart exposed to increased afterload in hypertensive patients.
The pathogenesis of the relationship between ethanol and hypertension is unclear. There are many studies relative to the effect of ethanol on nitric oxide synthase, some with contradictory results [56,57] . Raised endothelin-1 levels have been reported in alcoholics [58], but some authors only find these results in advanced (Child C) cirrhotics [59]. Also, increased sympathetic activity has been reported as a causative agent [60,61]. Increased arterial stiffness has been also reported in alcoholics, perhaps in association with increased levels of oxidized LDL, increased expression of adhesion molecules in monocytes [62] and increased atherogenesis [63]. A fascinating hypothesis is that this increased inflammatory status triggers increased secretion of FGF-23 and this in turn causes or contributes to vascular calcification, hypertension and left ventricular hypertrophy in alcoholics. All of this is still speculative, but research in this field is urgently needed.
Conclusion
This review illustrates the emerging role of bone as an endocrine organ. FGF-23, as an osteocyte-derived product is an example of the systemic effects of bone derived molecules that not only have a role in phosphate metabolism but are also related to vascular risk. Although only partially known, this molecule is also related to some a priori unexpected functions, such as hippocampal neurogenesis and learning [64]. Indeed, the fact that chronic inflammation triggers its secretion, together with its protean systemic effects, makes it plausible that it is perhaps involved in several manifestations of common conditions in which a smoldering inflammatory process ensues, as it may happen in the cardiovascular manifestations of alcoholism.
Author Contributions
González-Reimers E, Quintero-Platt G, Martin-Gonzalez C, Rodríguez-Gaspar M, Santolaria-Fernandez F contributed to manuscript conception. González-Reimers E, Quintero-Platt G, Martin-Gonzalez C, Romero-Acevedo L, Martínez-Martínez D,Santolaria-Fernandez F reviewed the literature. González-Reimers E, Quintero-Platt G, Martin-Gonzalez C, Romero-Acevedo L, Santolaria-Fernandez F, Rodríguez-Gaspar M, drafted the manuscript.
- Schwetz V, Pieber T, Obermayer-Pietsch B (2012) The endocrine role of the skeleton: background and clinical evidence. Eur J Endocrinol 166: 959-967. Link: https://goo.gl/ko3DmK
- Ducy P (2011) The role of osteocalcin in the endocrine cross-talk between bone remodelling and energy metabolism. Diabetologia 54: 1291-1297. Link: https://goo.gl/ImFzUz
- Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, et al. (2007) Endocrine regulation of energy metabolism by the skeleton. Cell 130: 456-469. Link: https://goo.gl/GvDvFP
- Anagnostis P, Karagiannis A, Kakafika AI, Tziomalos K, Athyros VG, et al.(2009) Atherosclerosis and osteoporosis: age-dependent degenerative processes or related entities? Osteoporos Int 20: 197-207. Link: https://goo.gl/oCirVA
- Yamashita T, Yoshioka M, Itoh N (2000) Identification of a novel fibroblast growth factor, FGF-23 preferentially expressed in the ventrolateral thalamic nucleus of the brain. Biochem Biophys Res Commun 277: 494-498. Link: https://goo.gl/J9MXoN
- White KE, Evans WE, O´Riordan LH, Speer MC, Econs MJ, et al. (2000) Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat Genet. 26: 345-348. Link: https://goo.gl/pu7fKS
- López I, Rodríguez-Ortiz ME, Almadén Y, Guerrero F, de Oca AM, et al. (2011) Direct and indirect effects of parathyroid hormone on circulating levels of fibroblast growth factor 23 in vivo. Kidney Int 80: 475-82. Link: https://goo.gl/di9shO
- David V, Dai B, Martin A, Huang J, Han X, et al. (2013) Calcium regulates FGF-23 expression in bone. Endocrinology 154: 4469-4482. Link: https://goo.gl/vQKFpO
- Razzaque MS (2009) The FGF23–Klotho axis: endocrine regulation of phosphate homeostasis. Nat Rev Endocrinol 5: 611-619. Link: https://goo.gl/IclIKb
- Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, et al. (2004) FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res 19: 429-435. Link: https://goo.gl/vb2PkY
- Andrukhova O, Slavic S, Smorodchenko A, Zeitz U, Shalhoub V, et al. (2014) FGF23 regulates renal sodium handling and blood pressure. EMBO Mol Med 6: 744-759. Link: https://goo.gl/FWrwsv
- Bacchetta J, Salusky IB, Hewison M (2013) Beyond mineral metabolism, is there an interplay between FGF23 and vitamin D in innate immunity? Pediatr Nephrol 28: 577-82. Link: https://goo.gl/hvQjLo
- Ben-Dov IZ, Galitzer H, Lavi-Moshayoff V, Goetz R, Kuro-o M, et al. (2007) The parathyroid is a target organ for FGF23 in rats. J Clin Invest 117:4003-4008. Link: https://goo.gl/d5gRNq
- Erben RG (2016) Update on FGF23 and Klotho signaling. Mol Cell Endocrinol 432: 56-65. Link: https://goo.gl/scnkM9
- Francis C, David V (2016) Inflammation regulates fibroblast growth factor 23 production. Curr Opin Nephrol Hypertens 25: 325-332. Link: https://goo.gl/yShCZS
- David V, Martin A, Isakova T, Spaulding C, Qi L, et al. (2016) Inflammation and functional iron deficiency regulate fibroblast growth factor 23 production. Kidney Int 89: 135-146. Link: https://goo.gl/YPxA4s
- David V, Francis C, Babitt JL (2017) Ironing out the cross talk between FGF23 and inflammation. Am J Physiol Renal Physiol 312: F1-F8. Link: https://goo.gl/Z8jilh
- Urakawa I, Yamazaki Y, Shimada T, Iijima K, Hasegawa H, et al. (2006) Klotho converts canonical FGF receptor into a specific receptor for FGF23. Nature 444:770-774. Link: https://goo.gl/NQpxwu
- Moe SM (2012) Klotho: a master regulator of cardiovascular disease? Circulation 125: 2181-2183. Link: https://goo.gl/GjFC8d
- Kuro-o M, Matsumura Y, Aizawa H, Kawaguchi H, Suga T, et al. (1997) Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 390: 45-51. Link: https://goo.gl/cidzmK
- Hesse M, Fröhlich LF, Zeitz U, Lanske B, Erben RG (2007) Ablation of vitamin D signaling rescues bone, mineral, and glucose homeostasis in Fgf-23 deficient mice. Matrix Biol. 26: 75-84. Link: https://goo.gl/Hw92lv
- Nakanishi K, Nishida M, Harada M, Ohama T, Kawada N, et al. (2015) Klotho-related Molecules Upregulated by Smoking Habit in Apparently Healthy Men: A Cross-sectional Study. Sci Rep 5: 14230. Link: https://goo.gl/S9tpAV
- Manghat P, Fraser WD, Wierzbicki AS, Fogelman I, Goldsmith DJ, et al. (2010) Fibroblast growth factor-23 is associated with C-reactive protein, serum phosphate and bone mineral density in chronic kidney disease. Osteoporos Int 21: 1853-1861. Link: https://goo.gl/NJoL9Q
- Kanaan N, Claes K, Devogelaer JP, Vanderschueren D, Depresseux G, et al. (2010) Fibroblast growth factor-23 and parathyroid hormone are associated with post-transplant bone mineral density loss. Clin J Am Soc Nephrol 5: 1887-1892. Link: https://goo.gl/7Q276Z
- Park SY, Jeong KH, Moon JY, Lee SH, Ihm CG, et al. (2010) The relationship between circulating fibroblast growth factor 23 and bone metabolism factors in Korean hemodialysis patients. Clin Exp Nephrol 14: 239-243. Link: https://goo.gl/sXekNW
- Gutiérrez OM, Mannstadt M, Isakova T, Rauh-Hain JA, Tamez H, et al. (2008) Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N Engl J Med 359: 584-592. Link: https://goo.gl/W85TzJ
- Faul C, Amaral AP, Oskouei B, Hu MC, Sloan A, et al. (2011) FGF23 induces left ventricular hypertrophy. J Clin Invest 121: 4393-4408. Link: https://goo.gl/Ur4Yys
- Brandenburg VM, Kleber ME, Vervloet MG, Tomaschitz A, Pilz S, et al. (2014) Fibroblast growth factor 23 (FGF23) and mortality: the Ludwigshafen Risk and Cardiovascular Health Study. Atherosclerosis 237: 53-59. Link: https://goo.gl/MRcqNb
- Hu X, Ma X, Luo Y, Xu Y, Xiong Q, et al. (2016) Associations of serum fibroblast growth factor 23 levels with obesity and visceral fat accumulation. Clin Nutr. Link: https://goo.gl/IJM6Q7
- Beben T, Ix JH, Shlipak MG, Sarnak MJ, Fried LF, et al. (2016) Fibroblast Growth Factor-23 and Frailty in Elderly Community-Dwelling Individuals: The Cardiovascular Health Study. J Am Geriatr Soc 64: 270-276. Link: https://goo.gl/ZfPljt
- Llauradó G, Megia A, Cano A, Giménez-Palop O, Simón I, et al. (2015) FGF-23/Vitamin D Axis in Type 1 Diabetes: The Potential Role of Mineral Metabolism in Arterial Stiffness. Link: https://goo.gl/Uj3Kd5
- Humalda JK, Seiler-Muler S, Kwakernaak AJ, Vervloet MG, Navis G, et al. (2016). Response of fibroblast growth factor 23 to volume interventions in arterial hypertension and diabetic nephropathy. Medicine (Baltimore) 95: e5003. Link: https://goo.gl/mM2kLj
- Grabner A, Amaral AP, Schramm K, Singh S, Sloan A, et al. (2015) Activation of Cardiac Fibroblast Growth Factor Receptor 4 Causes Left Ventricular Hypertrophy. Cell Metab. 22: 1020-1032. Link: https://goo.gl/0cS760
- Scialla JJ, Lau WL, Reilly MP, Isakova T, Yang HY, et al (2013) Fibroblast growth factor 23 is not associated with and does not induce arterial calcification. Kidney Int 83:1159-1168. Link: https://goo.gl/1agV4p
- Jimbo R, Kawakami-Mori F, Mu S, Hirohama D, Majtan B, et al.(2014) Fibroblast growth factor 23 accelerates phosphate-induced vascular calcification in the absence of Klotho deficiency. Kidney Int 85: 1103-1111. Link: https://goo.gl/Nf34GB
- Nakahara T, Kawai-Kowase K, Matsui H, Sunaga H, Utsugi T, et al. (2016) Fibroblast growth factor 23 inhibits osteoblastic gene expression and induces osteoprotegerin in vascular smooth muscle cells. Atherosclerosis. 253: 102-110. Link: https://goo.gl/xoAWgW
- Seiler S, Rogacev KS, Roth HJ, Shafein P, Emrich I, et al. (2014) Associations of FGF-23 and sKlotho with Cardiovascular Outcomes among Patients with CKD Stages 2–4. Clin J Am Soc Nephrol 9 :1049-1058. Link: https://goo.gl/uBBPTG
- Nafidi O, Lapointe RW, Lepage R, Kumar R, D'Amour P (2009) Mechanisms of renal phosphate loss in liver resection-associated hypophosphatemia. Ann Surg 249: 824-827. Link: https://goo.gl/BmJwPY
- Raimann A, Ertl DA, Helmreich M, Sagmeister S, Egerbacher M, et al. (2013) Fibroblast growth factor 23 and Klotho are present in the growth plate. Connect Tissue Res. 54:108-117. Link: https://goo.gl/N5jQgS
- Prié D, Forand A, Francoz C, Elie C, Cohen I, et al. (2013) Plasma Fibroblast Growth Factor 23 Concentration Is Increased and Predicts Mortality in Patients on the Liver-Transplant Waiting List. PloS One 8: e66182. Link: https://goo.gl/reSwId
- Wasserman H, Ikomi C, Hafberg ET, Miethke AG, Bove KE, et al. (2016) Two Case Reports of FGF23-Induced Hypophosphatemia in Childhood Biliary Atresia. Pediatrics 138. Link: https://goo.gl/XjrqKi
- Chen L, Liu H, Liu J, Zhu Y, Xu L, et al. (2013) Klotho endows hepatoma cells with resistance to anoikis via VEGFR2/PAK1 activation in hepatocellular carcinoma. PLoS One 8: e58413. Link:
- Lim K, Groen A, Molostvov G, Lu T, Lilley KS, et al. (2015) α-Klotho Expression in Human Tissues. J Clin Endocrinol Metab 100: E1308-1318. Link: https://goo.gl/2TrHKA
- Magdaleno F, Blajszczak CC, Nieto N (2017) Key Events Participating in the Pathogenesis of Alcoholic Liver Disease. Biomolecules 7: E9. Link: https://goo.gl/dEOfg8
- Mookerjee RP, Sen S, Davies NA, Hodges SJ, Williams R, et al. (2003) Tumour necrosis factor alpha is an important mediator of portal and systemic haemodynamic derangements in alcoholic hepatitis. Gut 52: 1182-1187. Link: https://goo.gl/i68Cyh
- Wang HJ, Zakhari S, Jung MK (2010) Alcohol, inflammation, and gut-liver-brain interactions in tissue damage and disease development. World J Gastroenterol 16: 1304-1311. Link: https://goo.gl/XcBoVB
- Nanji AA (2002) Role of Kupffer cells in alcoholic hepatitis. Alcohol 27: 13-15. Link: https://goo.gl/eoqJIs
- Rao R.(2009) Endotoxemia and gut barrier dysfunction in alcoholic liver disease. Hepatology 50: 638-644. Link: https://goo.gl/wxs5vg
- Gao B, Bataller R (2011) Alcoholic liver disease: pathogenesis and new therapeutic targets. Gastroenterology 141: 1572-1585. Link: https://goo.gl/95AKhF
- Quintero-Platt G, González-Reimers E, Rodríguez-Gaspar M, de la Vega-Prieto MJ, Martín-González C, et al. (2016) Cardiovascular disease burden, liver function, and fibroblast growth factor-23 in alcoholic patients. ISBRA-ESBRA CONGRESS. BERLIN 2016
- Klatsky AL, Friedman GD, Siegelaub AB, Gerard MJ (1977) Alcohol Consumption and Blood Pressure Kaiser-Permanente Multiphasic Health Examination Data. New Eng J Med 296: 1194–1200. Link: https://goo.gl/X2LMl9
- Arkwright PD, Beilin LJ, Rouse I, Armstrong BK, Vandongen R (1982) Effects of Alcohol Use and Other Aspects of Lifestyle on Blood Pressure Levels and Prevalence of Hypertension in a Working Population. Circulation 66: 60–66. Link: https://goo.gl/MhXKF5
- Marmot M G, Elliott P, Shipley MJ, Dyer AR, Uueshima H, et al. (1994) Alcohol and Blood Pressure: The INTERSALT Study. BMJ 308: 1263-1267. Link: https://goo.gl/cnXAaB
- Conway DI (2005) Alcohol consumption and the risk for disease. Is there a dose-risk relationship between alcohol and disease? Evid Based Dent 6: 76-77. Link: https://goo.gl/N6Ehah
- Manolio TA, Levy D, Garrison RJ, Castelli WP, Kannel WB (1991) Relation of Alcohol Intake to Left Ventricular Mass: The Framingham Study. J Am College Cardiol 17: 717–721. Link: https://goo.gl/W7Gg5s
- Durante W, Cheng K, Sunahara RK, Schafer AI (1995) Ethanol Potentiates Interleukin-1 β-Stimulated Inducible Nitric Oxide Synthase Expression in Cultured Vascular Smooth Muscle Cells. Biochem J 308: 231–236. Link: https://goo.gl/zOsn4I
- Husain K, Ferder L, Ansari RA, Lalla J (2011) Chronic Ethanol Ingestion Induces Aortic Inflammation/oxidative Endothelial Injury and Hypertension in Rats. Human Exp Toxicol 30: 930–939. Link: https://goo.gl/CS5v8c
- Di Gennaro C, Saccani-Otti G, Pinelli S, Venturi N, Manfredi G, et al.( 2012) Endothelial Dysfunction and High Cardiovascular Risk Profile in Severe Alcoholics Improve Only Partially Following a Medium-Term Alcohol Withdrawal. Alcohol Clin Expl Res 36: 242–250. Link: https://goo.gl/gdIQjQ
- Bruno CM, Neri S, Sciacca C, Caruso L (2000) Plasma Endothelin-1 Levels in Liver Cirrhosis. Int J Clin Lab Res 30: 169–172. Link: https://goo.gl/iuaVgr
- Kloner RA, Rezkalla SH ( 2007) To Drink or Not to Drink? That Is the Question. Circulation 116: 1306–1317. Link: https://goo.gl/YMssxf
- Da Silva AL, Ruginsk SG, Uchoa ET, Crestani CC, Scopinho AA, et al. (2013). Time-Course of Neuroendocrine Changes and Its Correlation with Hypertension Induced by Ethanol Consumption. Alcohol Alcohol 48: 495–504. Link: https://goo.gl/VlmKYj
- Redmond E M, Morrow D, Kundimi S, Miller-Graziano CL, Cullen JP (2009) Acetaldehyde Stimulates Monocyte Adhesion in a P-Selectin- and TNFα-Dependent Manner. Atherosclerosis 204: 372–380. Link: https://goo.gl/TDA6oY
- Zima T, Fialova L, Mestek O, Janebova M , Crkovska J, et al. ( 2001) Oxidative Stress, Metabolism of Ethanol and Alcohol-Related Diseases. J Biomed Sci 8: 59–70. Link: https://goo.gl/pLazU3 Liu P, Chen L, Bai X, Karaplis A, Miao D, et al. (2011) Impairment of spatial learning and memory in transgenic mice overexpressing human fibroblast growth factor-23. Brain Res. 1412: 9-17. Link: https://goo.gl/TiqCFw
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.

 Save to Mendeley
Save to Mendeley
